

Preparation and Electrochemical Performance of Carbon Nanoballs from Petroleum Asphalt
Received date: 2012-11-30
Online published: 2013-03-21
Supported by
Project supported by the Xinjiang Autonomous Region of high-tech projects (No. 201016118) and the Xinjiang Autonomous Region of major projects: (No. 201130113-1).
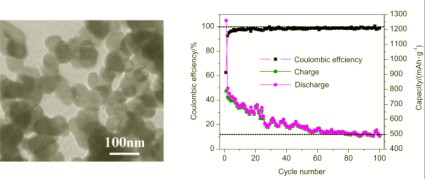
In this work, a simple low-temperature method has been employed to synthesize carbon nanoballs (CNBs). In a typical procedure, 1 g of petroleum asphalt in an alumina boat was directly heated to 450 ℃ at a rate of 10 ℃/min in air in a conventional box furnace to obtain the CNBs. Subsequently, the synthesized CNBs were treated in tubular furnace at 1500 ℃ under nitrogen atmosphere for 3 h and the obtained products were denoted as H-CNBs. The as-synthesized CNBs and H-CNBs were characterized by scanning electron microscopy, transmission electron microscopy, X-ray diffraction, Raman spectroscopy, and infrared spectroscopy. It was found that the diameters of CNBs and H-CNBs were in the range of 50~80 nm, and both of them showed a relatively low degree of graphitization. Compared with the CNBs, the H-CNBs showed a slightly higher degree of graphitization than the CNBs. A proposed formation mechanism of the CNBs is suggested to be as follows. Firstly, petroleum asphalt decomposed and formed a large amount of hydrocarbons at 220~500 ℃, such as benzene, methane, acetylene and so on. Carbon atoms from thermal cracking products of hydrocarbons deposited on the substrate in the absence of O2. Carbon atoms developed to CNBs as the temperature increased and the reaction time prolonged. The CNBs and H-CNBs were used as anode material in lithium ion batteries. The electrochemical performance was studied by galvanostatic charge/discharge at ambient temperature. The CNBs as anode materials exhibited a high initial capacity of 1260 mAh/g at a current of 1 C, which stabilized at around 500 mAh/g after 100 cycles. In contrast, the H-CNBs showed an initial capacity of 413 mAh/g under the same electrochemical conditions, which stabilized at 200 mAh/g after about 20 cycles with slight increase upon further cycling. Coulombic efficiency of as high as 98% was recorded after initial cycling for 10 cycles and 30 cycles for CNBs and H-CNBs, respectively. After cycling at different currents from 0.1 C, to 1 C, 5 C, and 10 C, the capacity of CNBs was almost fully recovered to the initial capacity at 0.1 C.
Key words: petroleum asphalt; carbon nanoballs; electrode material; specific capacity
Pan Hailing , Li Li , Liu Jianghua , Jia Dianzeng . Preparation and Electrochemical Performance of Carbon Nanoballs from Petroleum Asphalt[J]. Acta Chimica Sinica, 2013 , 71(05) : 787 -792 . DOI: 10.6023/A12110989
/
| 〈 |
|
〉 |