

A TS Search for Stable Configurations of Double Icosahedral Agn (n=19, 23, 24, 25) Clusters Linked by Sharing Atoms
Received date: 2013-05-02
Online published: 2013-06-27
Supported by
Project supported by the National Natural Science Foundation of China (Grant No. 51071065) and the Research Fund for the Doctoral Program of Higher Education of China (Grant No. 20100161110001).
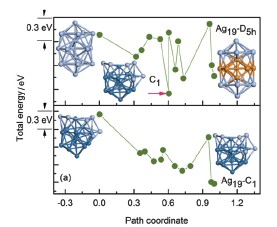
A DMol3 molecular orbital package based on DFT theory is adopted to calculate the energetics and electronic structures of icosahedral Ag13 and double icosahedral Agn (n=19, 23, 24, 25) clusters. Ab initio MD simulation for Ag13 cluster at 300 K is also carried out in the micro-canonical ensemble (NVE). During optimization and total energy calculation, electronic exchange-correlation energy functions represented in reciprocal space with the PBE type under GGA approximation are used. A double-numerical basis set together with d-polarization functions (DNP) is chosen to describe the electronic wave functions of Ag atoms. And only core electrons described by the DFT Semi-core Pseudopots (DSPP) are calculated. All atomic positions in Agn (n=13, 19, 23, 24, 25) clusters are relaxed by geometry optimization under RMS force of 0.002 Ha/Å and RMS displacement of 0.005 Å. The calculation of total energy and electronic structure is followed by the geometry optimization with self-consistent field (SCF) tolerance of 1×10-5 Ha. Further, using linear synchronous transit (LST) and quadratic synchronous transit (QST) methods based on the minimum energy path (MEP) with the aid of the climbing image nudged elastic band (NEB) method, a new transition state (TS) search technique for stable configurations of clusters is developed, in which the stable configuration is independent on selected initial configurations. As this technique being applied to search the stable configurations of Agn (n=7, 13, 19, 23, 25) clusters, it is confirmed the low energy stable configuration of the isolated icosahedron Ag13-Ih is indeed a plate-shape layered structure Ag13-C2 including a pentagonal bipyramid segment. The ground state of the double icosahedra linked with intercross sharing atoms Ag19-D5h is also demonstrated to be a spherical geometry Ag19-C1 consisting of 6 atoms around a deformed Ag13 icosahedron. And similarly to Agn (n=17~22) clusters, all stable configurations of Ag23-D3h, Ag24-D2h, Ag25-D5d clusters have a deformed Ag13 icosahedral nucleus as reported in literatures.
Key words: Agn cluster; stable configuration; TS search; DMol3
Jiang Yuanqi , Peng Ping , Wen Dadong , Han Shaochang . A TS Search for Stable Configurations of Double Icosahedral Agn (n=19, 23, 24, 25) Clusters Linked by Sharing Atoms[J]. Acta Chimica Sinica, 2013 , 71(10) : 1429 -1434 . DOI: 10.6023/A13050470
[1] Koretsky, G. M.; Knickelbein, M. B. J. Chem. Phys. 1997, 107, 10555.
[2] Tang, D.-Y.; Hu, J.-P.; Zhang, Y.-Q.; Hu, C.-W. Acta Chim. Sinica 2009, 67, 1859. (唐典勇, 胡建平, 张元勤, 胡常伟, 化学学报, 2009, 67, 1859.)
[3] Peyser, L. A.; Vinson, A. E.; Bartko, A. P.; Dickson, R. M. Science. 2001, 291, 103.
[4] Long, J.; Qiu, Y.-X.; Wang, S.-G. Acta Chim. Sinica 2008, 66, 1771. (龙娟, 仇毅翔, 王曙光, 化学学报, 2008, 66, 1771.)
[5] Mendez-Villuendas, E.; Bowles, R. K. Phys. Rev. Lett. 2007, 98, 185503.
[6] Liu, J.; Zhao, J. Z.; Hu, Z. Q. Appl. Phys. Lett. 2006, 89, 031903.
[7] Han, Z.; Zhang, D.-J.; Liu, C.-B. Acta Chim. Sinica 2009, 67, 387. (韩哲, 张冬菊, 刘成卜, 化学学报, 2009, 67, 387.)
[8] De Heer, W. A. Rev. Mod. Phys. 1993, 65, 611.
[9] Doye, J. P. K.; Wales, D. J. New. J. Chem. 1998, 22, 733.
[10] Piotrowski, M. J.; Piquini, P.; Da Silva, J. L. F. Phys. Rev. B 2010, 81, 155446.
[11] Oviedo, J.; Palmer, R. E. J. Chem. Phys. 2002, 117, 9548.
[12] Tian, D. X.; Zhang, H. L.; Zhao, J. J. Solid State Commun. 2007, 144, 174.
[13] Zhang, H. L.; Tian, D. X. Comput. Mater. Sci. 2008, 42, 462.
[14] Yang, M.; Jackson, K. A.; Jellinek, J. J. Chem. Phys. 2006, 125, 144308.
[15] Baishya, K.; Idrobo, J. C.; Ö?üt, S.; Yang, M. L.; Jackson, K.; Jellinek, J. Phys. Rev. B 2008, 78, 075439.
[16] Harb, M.; Rabilloud, F.; Simon, D.; Rydlo, A.; Lecoultre, S. J. Chem. Phys. 2008, 129, 194108.
[17] Li, G. F.; Peng, P.; Zhou, D. W.; Han, S. C. Comput. Mater. Sci. 2009, 47, 302.
[18] Henkelman, G.; Jónsson, H. J. Chem. Phys. 2000, 113, 9978.
[19] Henkelman, G.; Uberuaga, B. P.; Jónsson, H. J. Chem. Phys. 2000, 113, 9901.
[20] Shechtman, D.; Blech, I.; Gratias, D.; Cahn, J. W. Phys. Rev. Lett. 1984, 53, 1951.
[21] Reichert, H.; Klein, O.; Dosch, H.; Denk, M.; Honkimaki, V.; Lippmann, T.; Reiter, G. Nature 2000, 408, 839.
[22] Shen, Y. T.; Kim, T. H.; Gangopadhyay, A. K.; Kelton, K. F. Phys. Rev. Lett. 2009, 102, 057801.
[23] Li, F.; Liu, X. J.; Hou, H. Y.; Chen, G.; Chen, G. L. J. Appl. Phys. 2011, 110, 013519.
[24] Lee, M.; Lee, C. M.; Lee, K. R.; Ma, E.; Lee, J. C. Acta Mater. 2011, 59, 159.
[25] Kelton, K. F.; Lee, G. W.; Gangopadhyay, A. K.; Hyers, R. W.; Rathz, T. J.; Rogers, J. R.; Robinson, M. B.; Robinson, D. S. Phys. Rev. Lett. 2003, 90, 195504.
[26] Pang, H.; Jin, Z. H.; Lu, K. Phys. Rev. B 2003, 67, 094113.
[27] Delley, B. J. Chem. Phys. 1990, 92, 508.
[28] Delley, B. J. Chem. Phys. 2000, 113, 7756.
[29] Halgren, T. A.; Lipscomb, W. N. Chem. Phys. Lett. 1977, 49, 225.
[30] Kramer, H. G.; Beutel, V.; Weyers, K.; Demtroder, W. Chem. Phys. Lett. 1992, 193, 331.
[31] Morse, M. D. Chem. Rev. 1986, 86, 1049.
[32] Harris, I. A.; Kidwell, R. S.; Northby, J. A. Phys. Rev. Lett. 1984, 53, 2390.
[33] Echt, O.; Sattler, K.; Recknagel, E. Phys. Rev. Lett. 1981, 47, 1121.
[34] Tevekeliyska, V.; Dong, Y.; Springborg, M.; Grigoryan, V. G. Eur. Phys. J. D 2007, 43, 19.
[35] Doye, J. P. K.; Wales, D. J.; Zetterling, F. H. M.; Dzugutov, M. J. Chem. Phys. 2003, 118, 2792.
[36] Nagel, S. R.; Tauc, J. Phys. Rev. Lett. 1975, 35, 380.
[37] Zhao, Y.; Li, S.; Xu, W. G.; Li, Q. S. J. Phys. Chem. A 2004, 108, 4887.
/
| 〈 |
|
〉 |