

Palladium-Catalyzed Ring-Opening Reaction of Oxa/Azabenzonorbornadienes with Aryl Acetylenes
Received date: 2013-05-20
Online published: 2013-06-27
Supported by
Project supported by the National Natural Science Foundation of China (No. 21162040, 20902080, 21362043) and the Applied Basic Research Program of Yunnan Province (No. 2012FB170).
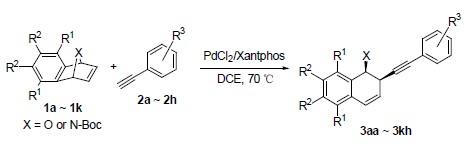
The transition-metal catalyzed ring opening reaction of oxa/azabenzonorbornadienes with carbo-nucleophiles has played an important role in the synthesis of tetrahydronaphthalene structures, which widely exist in natural products and bioactive molecules. Caused by the easy manipulation of reaction procedures and the less limits of suitable substrates, the ring opening reaction of oxa/azabenzonorbornadienes with terminal alkynes as nucleophiles has more potential in organic synthesis than with the organometal nucleophiles, such as zinc reagents, lithium reagents, Grignard reagents, aluminium reagents, organoboronic acids and zirconium reagents. However, only limited progress had been achieved for the ring opening reaction of oxa/azabenzonorbornadienes with terminal alkynes. Up to now, only a Ni(dppe)Cl2/ZnCl2/Zn catalytic system and a chiral rhodium catalyst had been developed for this reaction. Unfortunately, the former formed a relatively complicated reaction system, and suffered from the absence of chiral control ability. The latter was generated by composing of[Rh(OAc)(C2H4)2]2 with (R)-DTBM-segphos and could result in high enantioselectivity. But the scope of suitable substrates for it was too limited, and only sterically bulky acetylenes were found useful. Developing new and efficient catalysts for this reaction is still interesting and desirable. As the continuation of our study on transition-metal catalyzed reactions between norbornadienes and terminal alkynes, we surprisingly found that the complex of PdCl2 and Xantphos could catalyze the ring opening reaction of oxabenzonorbornadiene 1a with p-methoxyphenylacetylene 2a smoothly. The optimization of reaction conditions showed that 1,2-dichloroethane was the only effective solvent and 70 ℃ was the proper reaction temperature. The investigation on the scope of suitable substrates showed that this new developed PdCl2/Xantphos catalyst had good tolerance for various functional groups. Under the promotion of this Pd catalyst, a series of oxa/azabenzonorbornadienes could react with various aryl acetylenes, having different substituents on the phenyl rings, smoothly to generate the ring opening products in good yields. Further efforts to design and synthesize new Pd catalysts to realize the asymmetric version of the ring opening reaction of oxa/azabenzonorbornadienes with terminal alkynes are still in progress.
Chen Hualei , Li Sifeng , Xu Jianbin , Yang Qingjing , Liu Shanshan , Zhou Yongyun , Huang Chao , Fan Baomin . Palladium-Catalyzed Ring-Opening Reaction of Oxa/Azabenzonorbornadienes with Aryl Acetylenes[J]. Acta Chimica Sinica, 2013 , 71(9) : 1243 -1247 . DOI: 10.6023/A13050543
/
| 〈 |
|
〉 |