

Copper Complex Based Chemosensor which Could Colorimetrically Detect CN- in Water with Specific Selectivity and High Sensitivity
Received date: 2013-06-09
Online published: 2013-07-22
Supported by
Project supported by the National Natural Science Foundation of China (Nos. 21064006, 21161018 and 21262032), the Natural Science Foundation of Gansu Province (No. 1010RJZA018) and the Program for Changjiang Scholars and Innovative Research Team in University of Ministry of Education of China (IRT1177).
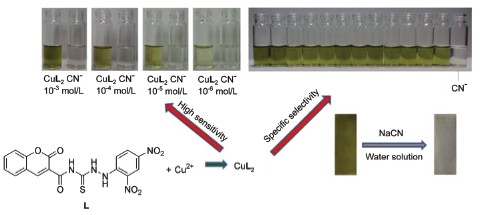
Owing to CN- could firmly bind copper(Ⅱ) via coordinate bonds, a competitive coordination mechanism based colorimetric chemosensor CuL2 was designed and synthesized. We carried out a series of CN- recognition research based on this work. As a result, chemosensor CuL2 could colorimetrically detect CN- in water solution with specific selectivity and high sensitivity. CuL2 can be utilized as a new colorimetric chemosensor for detection of cyanide at neutral pH in aqueous condition. Upon the addition of cyanide water solution to the DMSO/H2O (V:V=3:1) HEPES buffered solution (pH=7.0) of chemosensor CuL2, the green color of chemosensor CuL2 faded to colorless immediately, in the corresponding UV-vis spectra, the absorption at 446 nm disappeared. The detection limits were 8.0×10-6 and 4.0×10-7 mol/L of CN- using the visual color changes and UV-vis changes respectively, which is lower than the WHO guideline of 1.9 μmol·L-1. Other anions such as F-, Cl-, Br-, I-, AcO-, HSO4-, ClO4-, N3-, SO42-, NO3- and SCN- had no influence on the probing behavior of CuL2 toward cyanide. The CuL2 complex showed a highly selective and sensitive response toward cyanide by the complexation between the Cu2+ and CN-. The process can be monitored by UV-vis spectral changes as well as a visual color change. The good selectivity and high sensitivity of the complex CuL2 toward cyanide make CuL2 a promising candidate in practical applications as a good cyanide probe. Considering the simplicity of the analysis and low cost of the starting materials in the present system, this new method may be provided an useful potential candidate of colorimetric chemosensor for determination of cyanide in a variety of biological and environmental systems. Test strips based on CuL2 were fabricated, when different anion solutions were added to the test strips respectively, only the addition of CN- could induced an obviously color changes, the detection limits of the test strips for CN- is 8.0×10-5 mol/L. This test strips could act as a convenient and efficient CN- test kit.
Key words: cyanide ion; colorimetric recognition; complex; thiourea; copper(Ⅱ) ion
Lin Qi , Zhu Xin , Chen Pei , Fu Yongpeng , Zhang Youming , Wei Taibao . Copper Complex Based Chemosensor which Could Colorimetrically Detect CN- in Water with Specific Selectivity and High Sensitivity[J]. Acta Chimica Sinica, 2013 , 71(11) : 1516 -1520 . DOI: 10.6023/A13060614
/
| 〈 |
|
〉 |