

Deprotonation Kinetics of 1-Methylguanine After One-Electron Oxidation
Received date: 2014-09-28
Online published: 2014-10-22
Supported by
Project supported by the National Natural Science Foundation of China (No. 21333012), the National Basic Research Program of China (No. 2013CB834602), and the Chinese Academy of Sciences (No. XDB12020200).
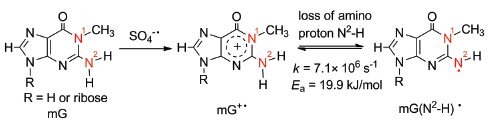
Among the four natural DNA bases, guanine (G) is the most sensitive to oxidation due to its lowest oxidation potential. When G base is oxidized to guanine cation radical (G+·), it will deprotonate from both the imino proton N1-H and the amino proton N2-H. According to the pKa values for N1-H and N2-H deprotonation, the main deprotonation site in G base is N1-H which would interfere with the N2-H deprotonation, making the kinetics of N2-H deprotonation difficult to be measured. Herein, the N2-H deprotonation kinetics is investigated using 1-methylguanosine (mG), where N1-H is substituted by methyl group to avoid the N1-H deprotonation and N9-H is substituted by ribose to ensure enough solubility of methylguanine in water, by nanosecond transient absorption (ns-TA) spectroscopy. By 355 nm photolysis of Na2S2O8, the highly oxidizing radical SO4-· is generated, which will oxidize mG to mG+· instantaneously. The time-resolved absorption spectra obtained for reaction of mG with SO4-· exhibits transient absorptions for mG(N2-H)· featured by absorption band at 600 nm, indicating that the mG+· deprotonation product is mG(N2-H)· and the deprotonation site is therefore validated to be N2-H. The mG concentration dependence of mG(N2-H)· formation rate constant is assessed through changing the mG concentration from 0.25 mmol·L-1 to 5 mmol·L-1. The concentration dependence experiment reveals that the rate-limiting step to form mG(N2-H)· is the bimolecular reaction of mG with SO4-· when mG concentration is lower than 2 mmol·L-1 and the bimolecular reaction rate constant to form mG+· is (3.7±0.1)×109 L·mol-1·s-1; when mG concentration is above 2 mmol·L-1, the rate-limiting step to form mG(N2-H)· is the first-order mG+· deprotonation and the N2-H deprotonation rate constant is (7.1±0.2)×106 s-1. Furthermore, the N2-H deprotonation rate constant is measured at different temperatures varying from 278 K to 298 K. According to Arrhenius equation, the activation energy barrier for the N2-H deprotonation is determined to be 19.9±1.0 kJ· mol-1. These results can provide valuable kinetic information on the oxidative damage of DNA.
Wu Lidan , Jie Jialong , Liu Kunhui , Su Hongmei . Deprotonation Kinetics of 1-Methylguanine After One-Electron Oxidation[J]. Acta Chimica Sinica, 2014 , 72(11) : 1182 -1186 . DOI: 10.6023/A14090666
[1] Lapi, A.; Pratviel, G.; Meunier, B. Met.-Based Drugs 2001, 8, 47.
[2] Zhang, Q. H.; Wang, Y.; Liu, C.; Yang, Z. Z. Acta Chim. Sinica 2014, 72, 956. (张千慧, 王阳, 刘翠, 杨忠志, 化学学报, 2014, 72, 956.)
[3] Yin, W. F.; Ou, Z. Z.; Gao, Y. Y.; Hao, P.; Guo, C. L.; Wang, Z. L. Acta Chim. Sinica 2010, 68, 1343. (殷卫峰, 欧植泽, 高云燕, 郝平, 郭创龙, 王中丽, 化学学报, 2010, 68, 1343.)
[4] Wang, X. X.; Gu, Y.; Chen, D. X.; Fang, Y. F.; Huang, Y. P. Acta Chim. Sinica 2010, 68, 2463. (王晓星, 顾彦, 陈登霞, 方艳芬, 黄应平, 化学学报, 2010, 68, 2463.)
[5] Lu, Y. M.; Ou, Z. B.; Hu, W.; Le, X. Y. Acta Chim. Sinica 2012, 70, 973. (卢艳梅, 区志镔, 胡伟, 乐学义, 化学学报, 2012, 70, 973.)
[6] Buxton, G. V.; Greenstock, C. L.; Helman, W. P.; Ross, A. B. J. Phys. Chem. Ref. Data 1988, 17, 513.
[7] Misiaszek, R.; Crean, C.; Joffe, A.; Geacintov, N. E.; Shafirovich, V. J. Biol. Chem. 2004, 279, 32106.
[8] Jaruga, P.; Dizdaroglu, M. Nucleic Acids Res. 1996, 24, 1389.
[9] Steenken, S.; Jovanovic, S. V.; Bietti, M.; Bernhard, K. J. Am. Chem. Soc. 2000, 122, 2373.
[10] Nguyen, K. V.; Burrows, C. J. Acc. Chem. Res. 2012, 45, 2151.
[11] Candeias, L.; Steenken, S. J. Am. Chem. Soc. 1989, 111, 1094.
[12] Candeias, L.; Steenken, S. J. Am. Chem. Soc. 1992, 114, 699.
[13] Kobayashi, K.; Tagawa, S. J. Am. Chem. Soc. 2003, 125, 10213.
[14] Mundy, C. J.; Colvin, M. E.; Quong, A. A. J. Phys. Chem. A 2002, 106, 10063.
[15] Wetmore, S. D.; Boyd, R. J.; Eriksson, L. A. J. Phys. Chem. B 1998, 102, 9332.
[16] Gervasio, F. L.; Laio, A.; Iannuzzi, M.; Parrinello, M. Chem.-Eur. J. 2004, 10, 4846.
[17] Adhikary, A.; Kumar, A.; Becker, D.; Sevilla, M. D. J. Phys. Chem. B 2006, 110, 24171.
[18] Adhikary, A.; Kumar, A.; Munafo, S. A.; Khanduri, D.; Sevilla, M. D. Phys. Chem. Chem. Phys. 2010, 12, 5353.
[19] Close, D. M.; Sagstuen, E.; Nelson, W. H. J. Chem. Phys. 1985, 82, 4386.
[20] Hole, E. O.; Sagstuen, E.; Nelson, W. H.; Close, D. M. Radiat. Res. 1992, 129, 1.
[21] Kobayashi, K.; Yamagami, R.; Tagawa, S. J. Phys. Chem. B 2008, 112, 10752.
[22] Kumar, A.; Sevilla, M. D. J. Phys. Chem. B 2009, 113, 11359.
[23] Liu, N.; Ban, F. Q.; Boyd, R. J. J. Phys. Chem. A 2006, 110, 9908.
[24] Ceron-Carrasco, J. P.; Requena, A.; Perpete, E. A.; Michaux, C.; Jacquemin, D. J. Phys. Chem. B 2010, 114, 13439.
[25] Huie, R. E.; Clifton, C. L.; Neta, P. Radiat. Phys. Chem. 1991, 38, 477.
[26] Rokhlenko, Y.; Geacintov, N. E.; Shafirovich, V. J. Am. Chem. Soc. 2012, 134, 4955.
[27] Mcelroy, W. J. J. Phys. Chem. 1990, 94, 2435.
[28] Chatgilialoglu, C.; Caminal, C.; Guerra, M.; Mulazzani, Q. G. Angew. Chem., Int. Ed. 2005, 44, 6030.
[29] Chatgilialoglu, C.; D'Angelantonio, M.; Guerra, M.; Kaloudis, P.; Mulazzani, Q. G. Angew. Chem., Int. Ed. 2009, 48, 2214.
/
| 〈 |
|
〉 |