

Research Progress of Aqueous Pollutants Removal by Sulfidated Nanoscale Zero-valent Iron
Received date: 2017-02-08
Online published: 2017-04-01
Supported by
Project supported by the National Natural Science Foundation of China (Nos. 51579096, 51222805, 51521006, 51508175) and the National Program for Support of Top-Notch Young Professionals of China (2012).
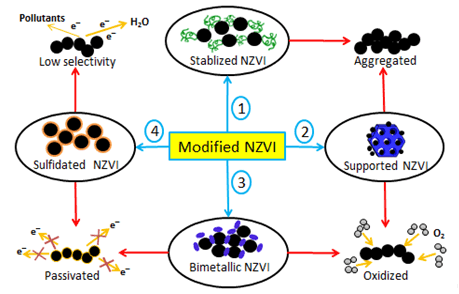
Nanoscale zero-valent iron (NZVI), an environmental remediation agent derived from wide range of raw materials, has been extensively applied in the field of remedying polluted water environment such as groundwater and wastewater. Although NZVI possesses some advantages such as excellent reaction activity, low cost and low toxicity, the limitation of in-situ remediation and storage concerning this kind of material has not been completely overcome yet. Among methods to improve the practical application of NZVI in water environment, sulfidation has become a research hotspot over recent decade. This means that the focus of modifying NZVI has shifted from reaction activity to electron selectivity. Most of the preparation methods of sulfidated NZVI belong to the chemical approach. These sulfidated materials have been heavily used to degrade organic pollutants and remove heavy metals in water to test their practical reactivity. Reaction mechanisms of pollutants and sulfidated NZVI in different environmental systems have also been extensively investigated. Hereinto, according to the species of organic pollutants and the reaction conditions, these reaction mechanisms can be roughly divided into three categories, including adsorption, reduction, and oxidation. In recent years, it is noted that sulfidated NZVI has made great progress to enhance the reaction activity and electrons selectivity, though it still has some limitations in the practical application. It is necessary to thoroughly review recent research progress about the reaction activities of sulfidated NZVI and its reaction mechanisms with pollutants in water, because it can clearly figure out new directions towards future development of sulfidated NZVI application. Due to the superior properties of sulfidated zero-valent iron, this material and relevant iron sulfide-based materials are going to belong to the most important functional materials in the field of environmental remediation with promising development prospect.
Tang Jing , Tang Lin , Feng Haopeng , Dong Haoran , Zhang Yi , Liu Sishi , Zeng Guangming . Research Progress of Aqueous Pollutants Removal by Sulfidated Nanoscale Zero-valent Iron[J]. Acta Chimica Sinica, 2017 , 75(6) : 575 -582 . DOI: 10.6023/A17020045
[1] Zhang, Y.; Zeng, G.; Tang, L.; Huang, D.; Jiang, X.; Chen, Y. Biosens. Bioelectron. 2007, 22, 2121.
[2] Tang, L.; Zeng, G.; Shen, G.; Li, Y.; Zhang, Y.; Huang, D. Environ. Sci. Technol. 2008, 42, 1207.
[3] Tang, L.; Fang, Y.; Pang, Y.; Zeng, G.; Wang, J.; Zhou, Y.; Deng, Y.; Yang, G.; Cai, Y.; Chen, J. Chem. Eng. J. 2014, 254, 302.
[4] Tang, L.; Yang, G.; Zeng, G.; Ca, Y.; Li, S.; Zhou, Y.; Pang, Y.; Liu, Y.; Zhang, Y.; Luna, B. Chem. Eng. J. 2014, 239, 114.
[5] Li, J.; Qin, H.; Zhang, X.; Guan, X. Acta Chim. Sinica 2017, DOI:10.6023/A17010007. (李锦祥, 秦荷杰, 张雪莹, 关小红, 化学学报, 2017, DOI:10.6023/A17010007.)
[6] Chen, H.; Huang, S.; Zhang, Z.; Liu, Y.; Wang, X. Acta Chim. Sinica 2017, DOI:10.6023/A17010039. (陈海军, 黄舒怡, 张志宾, 刘云海, 王祥科, 化学学报, 2017, DOI:10. 6023/A17010039.)
[7] Zhou, Q.; Li, J.; Wang, M.; Zhao, D. Crit. Rev. Env. Sci. Tec. 2016, 46, 783.
[8] Liang, Z.; Wang, Y. Environ. Prot. 2002, 4, 15. (梁震, 王焰新, 环境保护, 2002, 4, 15.)
[9] Jiemvarangkul, P.; Zhang, W.; Lien, H. L. Chem. Eng. J. 2011, 170, 482.
[10] Phenrat, T.; Saleh, N.; Sirk, K.; Tilton, R. D.; Lowry, G. V. Environ. Sci. Technol. 2007, 41, 284.
[11] Shi, L.; Zhang, X.; Chen, Z. Water Res. 2011, 45, 886.
[12] Greenlee, L. F.; Torrey, J. D.; Amaro, R. L.; Shaw, J. M. Environ. Sci. Technol. 2012, 46, 12913.
[13] Fan, D.; O'Brien Johnson, G.; Tratnyek, P. G.; Johnson, R. L. Environ. Sci. Technol. 2016, 50, 9558.
[14] Fan, D.; O'Carroll, D. M.; Elliott, D. W.; Xiong, Z.; Tratnyek, P. G.; Johnson, R. L.; Garcia, A. N. Remediat. J. 2016, 26, 27.
[15] Ma, X.; Gurung, A.; Deng, Y. Sci. Total Environ. 2013, 443, 844.
[16] El-Temsah, Y. S.; Joner, E. J. Chemosphere 2012, 89, 76.
[17] He, F.; Zhao, D.; Liu, J.; Roberts, C. B. Ind. Eng. Chem. Res. 2007, 46, 29.
[18] Liu, Z.; Zhang, F.; Hoekman, S. K.; Liu, T.; Gai, C.; Peng, N. ACS Sustain. Chem. Eng. 2016, 4, 3261.
[19] Shi, L. N.; Zhang, X.; Chen, Z. L. Water Res. 2011, 45, 886.
[20] Chen, Z.; Wang, T.; Jin, X.; Chen, Z.; Megharaj, M.; Naidu, R. J. Colloid Interface Sci. 2013, 398, 59.
[21] Wu, Y.; Yang, M.; Hu, S.; Wang, L.; Yao, H. Toxicol. Environ. Chem. 2014, 96, 227.
[22] Krasae, N.; Wantala, K.; Tantriratna, P.; Grisdanurak, N. App. Env. Res. 2014, 36, 15.
[23] Ryu, A.; Jeong, S. W.; Jang, A.; Choi, H. Appl. Catal. B-Environ. 2011, 105, 128.
[24] Xie, Y.; Fang, Z.; Cheng, W.; Tsang, P. E.; Zhao, D. Sci. Total Environ. 2014, 485, 363.
[25] Shih, Y. H.; Chen, M. Y.; Su, Y. F. Appl. Catal. B-Environ. 2011 105, 24.
[26] Kim, E. J.; Kim, J. H.; Azad, A. M.; Chang, Y. S. ACS Appl. Mater. Interfaces 2011, 3, 1457.
[27] Park, S. W.; Kim, S. K.; Kim, J. B.; Choi, S. W.; Inyang, H. I.; Tokunaga, S. Water Air Soil Pollut:Focus. 2006, 6, 97.
[28] He, F. In Iron Environmental Chemistry and Pollution Control Technology Seminar, Shanghai, 2016, p. 41. (何锋, 铁环境化学及污染控制技术研讨会, 上海, 2016, p. 41.)
[29] Yanlai, H.; Weile, Y. Environ. Sci. Technol. 2016, 50, 12992.
[30] Greenwood, N. N.; Earnshaw, A. Chemistry of the Elements, 2nd ed., Elsevier Butterworth Heinemann, 1997.
[31] Kappes, M.; Frankel, G. S.; Sridhar, N.; Carranza, R. M. J. Electrochem. Soc. 2012, 159, C195.
[32] Macdonald, D. D.; Roberts, B.; Hyne, J. B. Corros. Sci. 1978, 18, 411.
[33] Schmitt, G. Corrosion 1991, 47, 285.
[34] Rajajayavel, S. R. C.; Ghoshal, S. Water Res. 2015, 78, 144.
[35] Fan, D.; Anitori, R. P.; Tebo, B. M.; Tratnyek, P. G. Environ. Sci. Technol. 2013, 47, 5302.
[36] Tang, J.; Tang, L.; Feng, H.; Zeng, G.; Dong, H.; Zhang, C.; Huang, B.; Deng Y.; Wang, J.; Zhou, Y. J. Hazard. Mater. 2016, 320, 581.
[37] Butler, E. C.; Hayes, K. F. Environ. Sci. Technol. 2001, 35, 3884.
[38] Zou, Y.; Wang, X.; Khan, A.; Wang, P.; Liu, Y.; Alsaedi, A.; Hayat, T.; Wang, X. Environ. Sci. Technol. 2016, 50, 7290.
[39] Su, Y.; Adeleye, A. S.; Keller, A. A.; Huang, Y.; Dai, C.; Zhou, X.; Zhang, Y. Water Res. 2015, 74, 47.
[40] Xu, C.; Zhang, B.; Wang, Y.; Shao, Q.; Zhou, W.; Fan, D.; Bandstra, J. Z.; Shi, Z.; Tratnyek, P. G. Environ. Sci. Technol. 2016, 50, 11879.
[41] Li, D.; Mao, Z.; Zhong, Y.; Huang, W.; Wu, Y.; Peng, P. Water Res. 2016, 103, 1.
[42] Du, J.; Bao, J.; Lu, C.; Werner, D. Water Res. 2016, 102, 73.
[43] Fan, D.; Anitori, R. P.; Tebo, B. M.; Tratnyek, P. G. Environ. Sci. Technol. 2014, 48, 7409.
[44] Tang, L.; Tang, J.; Zeng, G.; Yang, G.; Xie, X.; Zhou, Y.; Pang, Y.; Fang, Y.; Wang, J.; Xiong, W. Appl. Surf. Sci. 2015, 333, 220.
[45] Ling, X.; Li, J.; Zhu, W.; Zhu, Y.; Sun, X.; Shen, J.; Han, W.; Wang, L. Chemosphere 2012, 87, 655.
[46] Zhuang, Y.; Ahn, S.; Luthy, R. G. Environ. Sci. Technol. 2010, 44, 8236.
[47] Ramos, M. A.; Yan, W.; Li, X. Q.; Koel, B. E.; Zhang, W. X. J. Phys. Chem. C 2009, 113, 14591.
[48] Ai, Z.; Gao, Z.; Zhang, L.; He, W.; Yin, J. J. Environ. Sci. Technol. 2013, 47, 5344.
[49] Wang, L.; Cao, M.; Ai, Z.; Zhang, L. Environ. Sci. Technol. 2014, 48, 3354.
[50] Liu, W.; Ai, Z.; Cao, M.; Zhang, L. Appl. Catal. B-Environ. 2014, 150~151, 1.
[51] Xiong, Z.; Lai, B.; Yang, P.; Zhou, Y.; Wang, J.; Fang, S. J. Hazard. Mater. 2015, 297, 261.
[52] Keenan, C. R.; Sedlak, D. L. Environ. Sci. Technol. 2008, 42, 1262.
[53] Song, S.; Su, Y.; Adeyemi, S. A.; Zhang, Y. Appl. Catal. B-Environ. 2017, 201, 211.
[54] Su, Y.; Adeleye, A. S.; Huang, Y.; Zhou, X.; Keller, A. A.; Zhang, Y. Sci. Rep. 2016, 6, 24358.
[55] Yang, X.; David, M. C. Environ. Sci. Technol. 2010, 44, 8649.
[56] Ariel, N. G.; Hardiljeet, K. B.; Denis, M. O. Environ. Sci. Technol. 2016, 50, 5243.
[57] David, T.; Dimin, F.; Paul, G. T.; Eun-Ju, K.; Yoon-Seok, C. Environ. Sci. Technol. 2012, 46, 12484.
[58] Kim, E. J.; Murugesan, K.; Kim, J. H.; Tratnyek, P. G.; Chang, Y. S. Ind. Eng. Chem. Res. 2013, 52, 9343.
[59] Eun-Ju, K.; Jae-Hwan, K.; Yoon-Seok, C.; David, T.; Paul, G. T. Environ. Sci. Technol. 2014, 48, 4002.
[60] Adeleye, A. S.; Stevenson, L. M.; Su, Y.; Nisbet, R. M.; Zhang, Y.; Keller, A. A. Environ. Sci. Technol. 2016, 50, 5597.
/
| 〈 |
|
〉 |