

Copper-Catalyzed Decarboxylative Iodination Reaction in the Gas Phase
Received date: 2018-02-26
Online published: 2018-04-28
Supported by
Project supported by the National Natural Science Foundation of China (No. 21532005), the National Key Basic Research Program of China (No. 2016YEF0200503), and the National Scientific Instrumentation Grant Program of China (Nos. 2012YQ12004907, 2011YQ09000501).
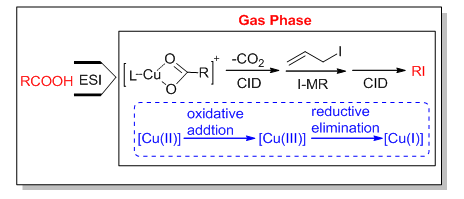
Organocopper complexes play the key role in Cu-catalyzed organic reaction. This manuscript offered a method to synthesize ligand-ligated organocopper complexes. Copper acetate was used as the catalyst and 2-(aminomethyl)pyridine (2-AMP) as the ligand to react with benzoic acid to generate the organocopper complex. This complex (A1) was easily transferred from solution to gas phase via electrospray ionization mass spectrometry (ESI-MS). Firstly, the collision-induced dissociation (CID) experiment of complex ion A1 was carried out in the ion-trap analyzer to investigate the gas-phase reactivity of it (the single isotope ion with 63Cu was isolated and used in MS/MS and next ion-molecule reaction). The decarboxylation reaction was taken place upon CID to generate the fragment ion B1. Next, the ion-molecule reaction (I-MR) of B1 was introduced after ion B1 was isolated, while allyl iodide was used as the neutral reagent. The iodine group transfer product ion C1 was obtained from the ion-molecule reation. The valence state of the central metal Cu changed from +2 in B1 to +3 in C1 during this process. Then ion A3 was dissociated to form the Cu(I) complex D1 with a neutral loss of iodobenzene upon CID. During these steps, the reagent benzoic acid reacted with allyl iodide in the gas phase with Cu2+ as catalyst and 2-AMP as ligand to produce iodobenzene, thus the copper-catalyzed decarboxylative iodination reaction was created in the gas phase. From the result, the mechanism of decarboxylative iodination reaction was speculated and carefully studied. Meanwhile, this reaction was also suitable for different carboxylic acids and bidentate nitrogen ligands. The aim of this manuscript is to study the reactive copper complexes in isolated environment and solvent free-condition. The gas phase mass spectrometric results supported the proposed mechanism. This method not only detected the gas-phase reactivities of a series of organocopper complexes, but also provided significant information of the mechanism of copper-catalyzed decarboxylative iodination reaction in the condensed phase.
Yin Xinchi , Jiang You , Chu Shiying , Weng Guofeng , Fang Xiang , Pan Yuanjiang . Copper-Catalyzed Decarboxylative Iodination Reaction in the Gas Phase[J]. Acta Chimica Sinica, 2018 , 76(6) : 436 -439 . DOI: 10.6023/A18020080
[1] Shepard, A. F.; Winslow, N. R.; Johnson, J. R. J. Am. Chem. Soc. 1930, 52, 2083.
[2] Hubacher, M. H. Anal. Chem. 1949, 21, 945.
[3] (a) Chamchaang, W.; Chantarasiri, N.; Chaona, S.; Thebtaranonth, C.; Thebtaranonth, Y. Tetrahedron 1984, 40, 1727.
(b) Lu, P. F.; Sanchez, C.; Cornella, J.; Larrosa, I. Org. Lett. 2009, 11, 5710.
(c) Cornella, J.; Sanchez, C.; Banawa, D.; Larrosa, I. Chem. Commun. 2009, 7176.
[4] (a) Li, Z.; Zheng, J.; Hu, W.; Li, J.; Wu, W.; Jiang, H. Org. Chem. Front. 2017, 4, 1363.
(b) Yuan, J. W.; Yang, L. R.; Mao, P.; Qu, L. B. Org. Chem. Front. 2017, 4, 545.
[5] Guan, B; Xu, X.; Wang, H.; Li, X. Chin. J. Org. Chem. 2016, 36, 1564(in Chinese). (关保川, 许孝良, 王红, 李小年, 有机化学, 2016, 36, 1564)
[6] (a) Frisch, A. C.; Beller, M. Angew. Chem. Int. Ed. 2005, 44, 674.
(b) Kambe, N.; Iwasaki, T.; Terao, J. Chem. Soc. Rev. 2011, 40, 4937.
(c) Tasker, S. Z.; Standley, E. A.; Jamison, T. F. Nature 2014, 509, 299.
[7] Eisch, J. J. Organometallics 2002, 21, 5439.
[8] (a) Goossen, L. J.; Collet, F.; Goossen, K. Isr. J. Chem. 2010, 50, 617.
(b) Rodriguez, N.; Goossen, L. J. Chem. Soc. Rev. 2011, 40, 5030.
(c) Shang, R.; Liu, L. Sci. China Chem. 2011, 54, 1670.
(d) Cornella, J.; Larrosa, I. Synthesis 2012, 653.
(e) Dzik, W. I.; Lange, P. P.; Goossen, L. J. Chem. Sci. 2012, 3, 2671.
[9] (a) Yamanaka, M.; Kato, S.; Nakamura, E. J. Am. Chem. Soc. 2004, 126, 6287.
(b) Yamanaka, M.; Nakamura, E. J. Am. Chem. Soc. 2005, 127, 4697.
(c) Candish, L.; Standley, E. A.; Gomez-Suarez, A.; Mukherjee, S.; Glorius, F. Chem. Eur. J. 2016, 22, 9971.
(d) Ni, S.; Sha, W.; Zhang, L.; Xie, C.; Mei, H.; Han, J.; Pan, Y. Org. Lett. 2016, 18, 712.
(e) Perry, G. J. P.; Quibell, J. M.; Panigrahi, A.; Larrosa, I. J. Am. Chem. Soc. 2017, 139, 11527.
[10] Nakamura, E.; Mori, S. Angew. Chem. Int. Ed. 2000, 39, 3750.
[11] (a) Bertz, S. H.; Cope, S.; Murphy, M.; Ogle, C. A.; Taylor, B. J. J. Am. Chem. Soc. 2007, 129, 7208.
(b) Hu, H.; Snyder, J. P. J. Am. Chem. Soc. 2007, 129, 7210.
[12] (a) Schroder, D. Acc. Chem. Res. 2012, 45, 1521.
(b) Coelho, F.; Eberlin, M. N. Angew. Chem. Int. Ed. 2011, 50, 5261.
(c) Gronert, S. Chem. Rev. 2001, 101, 329.
(d) Gronert, S. Mass Spectrom. Rev. 2005, 24, 100.
(e) Rijs, N.; Khairallah, G. N.; Waters, T. O'Hair, R. A. J. J. Am. Chem. Soc. 2008, 130, 1069.
[13] (a) Meyer, M. M.; Khairallah, G. N.; Kass, S. R.; O'Hair, R. A. J. Angew. Chem. Int. Ed. 2009, 48, 2934.
(b) Schlangen, M.; Schroder, D.; Schwarz, H. Angew. Chem. Int. Ed. 2007, 46, 1641.
(c) Schwarz, H. Angew. Chem. Int. Ed. 2011, 50, 10096.
[14] (a) Rijs, N.; Yates, B. F.; O'Hair, R. A. J. Chem. Eur. J. 2010, 16, 2674.
(b) Rijs, N.; Yoshikai, N.; Nakamura, E.; O'Hair, R. A. J. J. Am. Chem. Soc. 2012, 134, 2569.
(c) Rijs, N.; O'Hair, R. A. J. Organometallics 2012, 31, 8012.
(d) Sharif, H. A.; Vikse, K. L.; Khairallah, G. N.; O'Hair, R. A. J. Organometallics 2013, 32, 5416.
(e) Rijs, N.; Gonzalez-Navarrete, P.; Schlangen, M.; Schwarz, H. J. Am. Chem. Soc. 2016, 138, 3125.
(f) Geng, C.; Li, J.; Weiske, T.; Schlangen, M.; Shaik, S.; Schwarz, H. J. Am. Chem. Soc. 2017, 139, 1684.
[15] Zhang, X.; Bai, X.; Fang, L.; Jiang, K.; Li, Z. J. Am. Soc. Mass Spectrom. 2016, 27, 940.
[16] Jiang, X.; Huang, H.; Chai, Y.; Lohr, T. L.; Yu, S.; Lai, W.; Pan, Y.; Delferro, M.; Marks, T. J. Nat. Chem. 2017, 9, 188.
[17] Chai, Y.; Shen, S.; Weng, G.; Pan, Y. Chem. Commun. 2014, 50, 11668.
/
| 〈 |
|
〉 |