

Recent Advances in Visible Light Induced Radical 1,2-Functionalization of Alkynes
Received date: 2024-03-18
Online published: 2024-04-29
Supported by
National Natural Science Foundation of China(22071062); National Natural Science Foundation of China(22271096); Fundamental Research Funds for the Central Universities, SCUT(2022ZYGXZR107)
As one of the most basic functional groups in organic molecules, the carbon-carbon triple bond containing two π-bonds harbor great potential for diverse organic transformations. Among them, the functionalization reaction based on the addition of free radicals to alkynes is an effective way to synthesize diverse functionalized olefins, ketones, and even alkane molecules, as well as an important tool for constructing multiple chemical bonds simultaneously. In the last decade, the application of visible light-induced radical reaction strategy for alkyne functionalization has received widespread attention due to it align with the concepts of green chemistry and sustainable development. Nowadays, photoinduced radical functionalization of alkynes has been developed unprecedentically, and many new methods have emerged. Indeed, light-induced radical functionalization reactions of alkynes provide a relatively green approach to the diverse transformations of alkynes. These conversions include hydrofunctionalization, oxidative functionalization, difunctionalization, and so on. These reactions feature mild conditions and easy operation, and do not even require the addition of metals and photosensitizers. Importantly, they provide an efficient way for the construction of valuable skeletons. In particular, the difunctionalization reaction enables the synthesis of high-added-value challenging molecules from simple feedstocks. In these reactions, the alkenyl radicals formed by radical addition to alkynes are the key intermediates. Thus, the type of radical precursor and the further transformation of the alkenyl radical determine the type of chemical bond constructed and the structure of the final product. This paper mainly reviews the new advances in photoinduced radical 1,2-functionalization reactions of alkynes in the past five years, classifying them in terms of the types of chemical bonds constructed, and providing a systematic summary of the types of radicals and reactions. The mechanism and universality of the reactions, as well as its advantages and disadvantages were discussed, and the challenges and opportunities facing the field were outlooked as well.
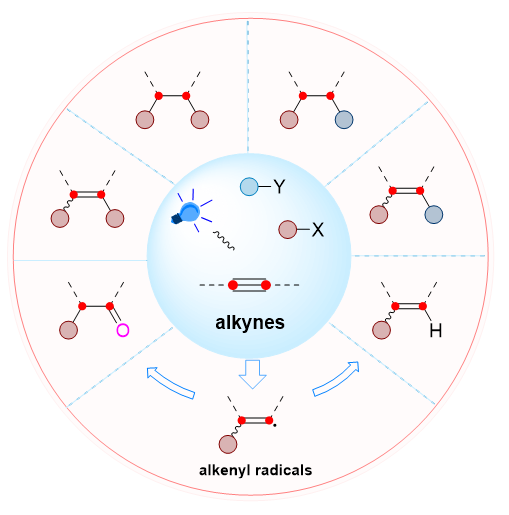
Key words: visible light; alkynes; radical reaction; functionalization; photocatalysis
Kangkui Li , Xianyang Long , Yue Huang , Shifa Zhu . Recent Advances in Visible Light Induced Radical 1,2-Functionalization of Alkynes[J]. Acta Chimica Sinica, 2024 , 82(6) : 658 -676 . DOI: 10.6023/A24030090
| [1] | (a) Gleiter, R.; Werz, D. B. Chem. Rev. 2010, 110, 4447. |
| [1] | (b) Brand, J. P.; Waser, J. Chem. Soc. Rev. 2012, 41, 4165. |
| [1] | (c) Bhunia, S.; Ghosh, P.; Patra, S. R. Adv. Synth. Catal. 2020, 362, 3664. |
| [1] | (d) Heravi, M. M.; Dehghani, M.; Zadsirjan, V.; Ghanbarian, M. Curr. Org. Synth. 2019, 16, 205. |
| [1] | (e) Chen, L.; Chen, K.; Zhu, S. Chem 2018, 4, 1208. |
| [1] | (f) Patel, M.; Saunthwal, R. K.; Verma, A. K. Acc. Chem. Res. 2017, 50, 240. |
| [1] | (g) Alabugin, I. V.; Gonzalez-Rodriguez, E. Acc. Chem. Res. 2018, 51, 1206. |
| [1] | (h) Fang, G.; Bi, X. Chem. Soc. Rev. 2015, 44, 8124. |
| [1] | (i) Khan, R.; Chen, J.; Fan, B. Adv. Synth. Catal. 2020, 362, 1564. |
| [2] | (a) Wille, U. Chem. Rev. 2013, 113, 813. |
| [2] | (b) Hu, C.; Mena, J.; Alabugin, I. V. Nat. Rev. Chem. 2023, 7, 405. |
| [2] | (c) Ren, X.; Lu, Z. Chin. J. Catal. 2019, 40, 1003. |
| [2] | (d) Chalotra, N.; Kumar, J.; Naqvi, T.; Shah, B. A. Chem. Commun. 2021, 57, 11285. |
| [2] | (e) Bag, D.; Kour, H.; Sawant, S. D. Org. Biomol. Chem. 2020, 18, 8278. |
| [3] | (a) Zhang, Y.; Cai, Z.; Warratz, S.; Ma, C.; Ackermann, L. Sci. China Chem. 2023, 66, 703. |
| [3] | (b) Deng, W.; Li, Y.; Li, Y.-G.; Bao, H. Synthesis 2018, 50, 2974. |
| [3] | (c) Ma, J.; Li, J.; Meng, Q.; Zeng, X. Chin. J. Org. Chem. 2023, 43, 2040. (in Chinese) |
| [3] | (马佳敏, 李姣兄, 孟千森, 曾祥华, 有机化学, 2023, 43, 2040.) |
| [4] | (a) Romero, N. A.; Nicewicz, D. A. Chem. Rev. 2016, 116, 10075. |
| [4] | (b) Prier, C. K.; Rankic, D. A.; MacMillan, D. W. C. Chem. Rev. 2013, 113, 5322. |
| [4] | (c) Milligan, J. A.; Phelan, J. P.; Badir, S. O.; Molander, G. A. Angew. Chem. Int. Ed. 2019, 58, 6152. |
| [4] | (d) Gao, P.-P.; Xiao, W.-J.; Chen, J.-R. Chin. J. Org. Chem. 2022, 42, 3923. (in Chinese) |
| [4] | (高盼盼, 肖文精, 陈加荣, 有机化学, 2022, 42, 3923.) |
| [4] | (e) Wang, H.; Wu, P.; Zhao, X.; Zeng, J.; Wan, Q. Acta Chim. Sinica 2019, 77, 231. (in Chinese) |
| [4] | (王浩, 吴品儒, 赵祥, 曾静, 万谦, 化学学报, 2019, 77, 231.) |
| [4] | (f) Laishram, R. D.; Chen, J.; Fan, B. Chem. Rec. 2021, 21, 69. |
| [4] | (g) Zhang, J.; Chen, Y. Acta Chim. Sinica 2017, 75, 41. (in Chinese) |
| [4] | (张晶, 陈以昀, 化学学报, 2017, 75, 41.) |
| [4] | (h) Wang, D.; Zhang, L.; Luo, S. Acta Chim. Sinica 2017, 75, 22. (in Chinese) |
| [4] | (王德红, 张龙, 罗三中, 化学学报, 2017, 75, 22.) |
| [5] | Giese, B.; Lachhein, S. Angew. Chem., Int. Ed. Engl. 1982, 21, 768. |
| [6] | (a) Zheng, H.; Su, J.; Zhou, Y.; Zhu, G. Chin. J. Org. Chem. 2022, 42, 4060. (in Chinese) |
| [6] | (郑汉良, 苏静雯, 周雨露, 朱钢国, 有机化学, 2022, 42, 4060.) |
| [6] | (b) Liao, J.; Yang, X.; Ouyang, L.; Lai, Y.; Huang, J.; Luo, R. Org. Chem. Front. 2021, 8, 1345. |
| [6] | (c) Zhou, Y.; Gu, Z.; Hong, Y.; Chen, H.; Luo, J.; Zheng, H.; Zhu, G. Org. Chem. Front. 2024, 11, 1232. |
| [7] | Ren, Y.-Y.; Zheng, X.; Zhang, X. Synlett 2018, 29, 1028. |
| [8] | Meyer, C. F.; Hell, S. M.; Misale, A.; Trabanco, A. A.; Gouverneur, V. Angew. Chem. Int. Ed. 2019, 58, 8829. |
| [9] | Li, K.; Zhang, X.; Chen, J.; Gao, Y.; Yang, C.; Zhang, K.; Zhou, Y.; Fan, B. Org. Lett. 2019, 21, 9914. |
| [10] | Deng, H.-P.; Fan, X.-Z.; Chen, Z.-H.; Xu, Q.-H.; Wu, J. J. Am. Chem. Soc. 2017, 139, 13579. |
| [11] | Song, Z.-Q.; Liu, Z.; Gan, Q.-C.; Lei, T.; Tung, C.-H.; Wu, L.-Z. Org. Lett. 2020, 22, 832. |
| [12] | Liu, F.; Zhang, K.; Zhao, X.-F.; Meng, Q.-X.; Zhao, T.-S.; Tian, W.-F.; He, Y.-Q. Tetrahedron Lett. 2023, 115, 154321. |
| [13] | Yue, H.; Zhu, C.; Kancherla, R.; Liu, F.; Rueping, M. Angew. Chem. Int. Ed. 2020, 59, 5738. |
| [14] | Lai, S.-Z.; Yang, Y.-M.; Xu, H.; Tang, Z.-Y.; Luo, Z. J. Org. Chem. 2020, 85, 15638. |
| [15] | Wang, H.; Li, Y.; Tang, Z.; Wang, S.; Zhang, H.; Cong, H.; Lei, A. ACS Catal. 2018, 8, 10599. |
| [16] | Huang, T.; Saga, Y.; Guo, H.; Yoshimura, A.; Ogawa, A.; Han, L.-B. J. Org. Chem. 2018, 83, 8743. |
| [17] | Ikeshita, D.; Masuda, Y.; Ishida, N.; Murakami, M. Org. Lett. 2022, 24, 2504. |
| [18] | Kaur, S.; Zhao, G.; Busch, E.; Wang, T. Org. Biomol. Chem. 2019, 17, 1955. |
| [19] | Burykina, J. V.; Shlapakov, N. S.; Gordeev, E. G.; K?nig, B.; Ananikov, V. P. Chem. Sci. 2020, 11, 10061. |
| [20] | Wu, X.; Gao, B. Org. Lett. 2023, 25, 8722. |
| [21] | Zhong, M.; Gagné, Y.; Hope, T. O.; Pannecoucke, X.; Frenette, M.; Jubault, P.; Poisson, T. Angew. Chem. Int. Ed. 2021, 60, 14498. |
| [22] | Tang, H.; Zhang, M.; Zhang, Y.; Luo, P.; Ravelli, D.; Wu, J. J. Am. Chem. Soc. 2023, 145, 5846. |
| [23] | Huo, L.; Li, X.; Zhao, Y.; Li, L.; Chu, L. J. Am. Chem. Soc. 2023, 145, 9876. |
| [24] | Malpani, Y. R.; Biswas, B. K.; Han, H. S.; Jung, Y.-S.; Han, S. B. Org. Lett. 2018, 20, 1693. |
| [25] | Lv, Y.; Liu, Q.; Liu, F.; Yue, H.; Li, J.-S.; Wei, W. Tetrahedron Lett. 2020, 61, 151335. |
| [26] | Hosseini-Sarvari, M.; Valikhani, A. New J. Chem. 2021, 45, 12464. |
| [27] | Song, T.; Zhang, Y.; Wang, C.; Li, Y.; Yang, Y. Chin. J. Chem. 2022, 40, 2618. |
| [28] | Liu, L.; Liu, M.; Liu, B.; Wang, Q.; Li, Y.; Feng, K.; Qiu, R.; Zhou, Y. Tetrahedron Lett. 2023, 133, 154823. |
| [29] | (a) Badir, S. O.; Molander, G. A. Chem 2020, 6, 1327. |
| [29] | (b) Zhu, C.; Yue, H.; Chu, L.; Rueping, M. Chem. Sci. 2020, 11, 4051. |
| [29] | (c) Zhu, S.; Zhao, X.; Li, H.; Chu, L. Chem. Soc. Rev. 2021, 50, 10836. |
| [29] | (d) Dong, Y.-J.; Zhu, B.; Geng, Y.; Zhao, Z.-W.; Su, Z.-M.; Guan, W. CCS Chem. 2021, 4, 1429. |
| [29] | (e) Xu, L.; Wang, F.; Chen, F.; Zhu, S.; Chu, L. Chin. J. Org. Chem. 2022, 42, 1. (in Chinese) |
| [29] | (徐磊, 王方, 陈凡, 朱圣卿, 储玲玲, 有机化学, 2022, 42, 1.) |
| [30] | Guo, L.; Song, F.; Zhu, S.; Li, H.; Chu, L. Nat. Commun. 2018, 9, 4543. |
| [31] | Qin, J.; Zhang, Z.; Lu, Y.; Zhu, S.; Chu, L. Chem. Sci. 2023, 14, 12143. |
| [32] | Yang, B.; Li, S.-J.; Wang, Y.; Lan, Y.; Zhu, S. Nat. Commun. 2021, 12, 5257. |
| [33] | Yang, B.; Lu, S.; Wang, Y.; Zhu, S. Nat. Commun. 2022, 13, 1858. |
| [34] | Zhang, Y.; Han, Y.; Zhu, S.; Qing, F.-L.; Xue, X.-S.; Chu, L. Angew. Chem. Int. Ed. 2022, 61, e202210838. |
| [35] | Wang, J.; Wu, X.; Cao, Z.; Zhang, X.; Wang, X.; Li, J.; Zhu, C. Adv. Sci. 2024, 2309022. |
| [36] | Lee, W.; Lee, Y.; Yoo, M.; Han, S. B.; Kim, H. J. Org. Chem. Front. 2020, 7, 3209. |
| [37] | Dong, X.; Jiang, W.; Hua, D.; Wang, X.; Xu, L.; Wu, X. Chem. Sci. 2021, 12, 11762. |
| [38] | Pan, C.; Meng, Y.; Deng, Y.; Zhou, B.; Chen, J.; He, Z.; Sun, W.; Khan, R.; Fan, B. Chin. J. Chem. 2022, 40, 2040. |
| [39] | Wang, Y.; Cao, Z.; He, Q.; Huang, X.; Liu, J.; Neumann, H.; Chen, G.; Beller, M. Chem. Sci. 2023, 14, 1732. |
| [40] | Miao, M.; Zhu, L.; Zhao, H.; Song, L.; Yan, S.-S.; Liao, L.-L.; Ye, J.-H.; Lan, Y.; Yu, D.-G. Sci. China Chem. 2023, 66, 1457. |
| [41] | Masuda, Y.; Ikeshita, D.; Higashida, K.; Yoshida, M.; Ishida, N.; Murakami, M.; Sawamura, M. J. Am. Chem. Soc. 2023, 145, 19060. |
| [42] | Zhu, C.; Yue, H.; Maity, B.; Atodiresei, I.; Cavallo, L.; Rueping, M. Nat. Catal. 2019, 2, 678. |
| [43] | Long, T.; Zhu, C.; Li, L.; Shao, L.; Zhu, S.; Rueping, M.; Chu, L. Nat. Commun. 2023, 14, 55. |
| [44] | Xu, L.; Zhu, S.; Huo, L.; Chen, F.; Yu, W.; Chu, L. Org. Chem. Front. 2021, 8, 2924. |
| [45] | Li, K.; Deng, J.; Long, X.; Zhu, S. Green Chem. 2023, 25, 7253. |
| [46] | Lü, S.; Wang, Z.; Zhu, S. Nat. Commun. 2022, 13, 5001. |
| [47] | Lü, S.; Wang, Z.; Gao, X.; Chen, K.; Zhu, S. Angew. Chem. Int. Ed. 2023, 62, e202300268. |
| [48] | Yang, B.; Li, K.; Wang, Y.; Zhu, S. Sci. China Chem. 2024, 67, 936. |
| [49] | Chakrasali, P.; Kim, K.; Jung, Y.-S.; Kim, H.; Han, S. B. Org. Lett. 2018, 20, 7509. |
| [50] | Li, H.; Cheng, Z.; Tung, C.-H.; Xu, Z. ACS Catal. 2018, 8, 8237. |
| [51] | Sahoo, A. K.; Dahiya, A.; Das, B.; Behera, A.; Patel, B. K. J. Org. Chem. 2021, 86, 11968. |
| [52] | Nie, X.; Xu, T.; Hong, Y.; Zhang, H.; Mao, C.; Liao, S. Angew. Chem. Int. Ed. 2021, 60, 22035. |
| [53] | Kumar, S.; Kumar, J.; Naqvi, T.; Raheem, S.; Rizvi, M. A.; Shah, B. A. ChemPhotoChem 2022, 6, e202200110. |
| [54] | Abramov, V. A.; Topchiy, M. A.; Rasskazova, M. A.; Drokin, E. A.; Sterligov, G. K.; Shurupova, O. V.; Malysheva, A. S.; Rzhevskiy, S. A.; Beletskaya, I. P.; Asachenko, A. F. Org. Biomol. Chem. 2023, 21, 3844. |
| [55] | Ren, X.; Ke, Q.; Zhou, Y.; Jiao, J.; Li, G.; Cao, S.; Wang, X.; Gao, Q.; Wang, X. Angew. Chem. Int. Ed. 2023, 62, e202302199. |
| [56] | Xie, S.-L.; Yan, J.-Z.; Xie, M.-J.; Li, X.; Zhou, F.; Zheng, M.-Q.; Wang, X.-L.; Feng, J.; Zhang, Y.; Duan, Y.-N.; Niu, Y.-D.; Li, D.; Xia, H.-D. Green Chem. 2024, 26, 323. |
/
| 〈 |
|
〉 |