Chinese Journal of Organic Chemistry ›› 2019, Vol. 39 ›› Issue (11): 3223-3229.DOI: 10.6023/cjoc201904051 Previous Articles Next Articles
王毛锐, 吴雨峥, 姚健, 邓黎, 潘英明, 黄克斌*( ), 唐海涛*(
), 唐海涛*( )
)
收稿日期:2019-04-22
发布日期:2019-07-03
通讯作者:
黄克斌,唐海涛
E-mail:kbhuang@mailbox.gxnu.edu.cn;httang@gxnu.edu.cn
基金资助:
Wang Maorui, Wu Yuzheng, Yao Jian, Deng Li, Pan Yingming, Huang Kebin*( ), Tang Haitao*(
), Tang Haitao*( )
)
Received:2019-04-22
Published:2019-07-03
Contact:
Huang Kebin,Tang Haitao
E-mail:kbhuang@mailbox.gxnu.edu.cn;httang@gxnu.edu.cn
Supported by:Share
Wang Maorui, Wu Yuzheng, Yao Jian, Deng Li, Pan Yingming, Huang Kebin, Tang Haitao. Triethylamine Promoted the C-C Bond Cleavage of α-Halo Ketones: α-Acetoxyaryl Ketone Synthesis[J]. Chinese Journal of Organic Chemistry, 2019, 39(11): 3223-3229.
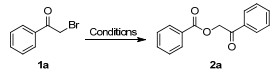 | ||||
| Entry | Catalyst | Base (equiv.) | Solvent | Yieldb/% |
| 1 | Pd(OAc)2 | Et3N (0.5) | MeCN | 44c |
| 2 | — | Et3N (0.5) | MeCN | 46 |
| 3 | — | Et3N (1) | MeCN | 55 |
| 4 | — | Et3N (2) | MeCN | 66 |
| 5 | — | Et3N (3) | MeCN | 60 |
| 6 | — | Et2NH (2) | MeCN | 45 |
| 7 | — | (CH3)3CNH2 (2) | MeCN | Trace |
| 8 | — | DBU (2) | MeCN | NR |
| 9 | — | NaOH (2) | MeCN | NR |
| 10 | — | Cs2CO3 (2) | MeCN | Trace |
| 11 | — | Et3N (2) | MeCN/H2Od | 43 |
| 12 | — | Et3N (2) | 1, 4-Dioxane | NR |
| 13 | — | Et3N (2) | DMF | Trace |
| 14 | — | — | N(Et)3 | 23 |
| 15 | — | Et3N (2) | MeCN | 89e |
 | ||||
| Entry | Catalyst | Base (equiv.) | Solvent | Yieldb/% |
| 1 | Pd(OAc)2 | Et3N (0.5) | MeCN | 44c |
| 2 | — | Et3N (0.5) | MeCN | 46 |
| 3 | — | Et3N (1) | MeCN | 55 |
| 4 | — | Et3N (2) | MeCN | 66 |
| 5 | — | Et3N (3) | MeCN | 60 |
| 6 | — | Et2NH (2) | MeCN | 45 |
| 7 | — | (CH3)3CNH2 (2) | MeCN | Trace |
| 8 | — | DBU (2) | MeCN | NR |
| 9 | — | NaOH (2) | MeCN | NR |
| 10 | — | Cs2CO3 (2) | MeCN | Trace |
| 11 | — | Et3N (2) | MeCN/H2Od | 43 |
| 12 | — | Et3N (2) | 1, 4-Dioxane | NR |
| 13 | — | Et3N (2) | DMF | Trace |
| 14 | — | — | N(Et)3 | 23 |
| 15 | — | Et3N (2) | MeCN | 89e |
| [1] |
(a) Souillart, L.; Cramer, N. Chem. Rev. 2015, 115, 9410.
doi: 10.1021/acs.chemrev.5b00138 |
|
(b) Chen, F.; Wang, T.; Jiao, N. Chem. Rev. 2014, 114, 8613.
doi: 10.1021/acs.chemrev.5b00138 |
|
|
(c) Wang, T.; Jiao, N. Acc. Chem. Res. 2014, 47, 1137.
doi: 10.1021/acs.chemrev.5b00138 |
|
|
(d) Murakami, M.; Ishida, N. J. Am. Chem. Soc. 2016, 138, 13759.
doi: 10.1021/acs.chemrev.5b00138 |
|
|
(e) Tiwari, B.; Zhang, J.; Chi, Y. R. Angew. Chem. Int. Ed. 2012, 51, 1911.
doi: 10.1021/acs.chemrev.5b00138 |
|
|
(f) Sai, M.; Yorimitsu, H.; Oshima, K. Angew. Chem. Int. Ed. 2011, 50, 3294.
doi: 10.1021/acs.chemrev.5b00138 |
|
|
(g) Chen, W.-L.; Wu, S.-Y.; Mo, X.˗L.; Wei, L.˗X.; Liang, C.; Mo, D.˗L. Org. Lett. 2018, 20, 3527.
doi: 10.1021/acs.chemrev.5b00138 |
|
| [2] |
(a) Tobisu, M.; Chatani, N. Chem. Soc. Rev. 2008, 37, 300.
doi: 10.1039/B702940N |
|
(b) Qin, C.; Shen, T.; Tang, C.; Jiao, N. Angew. Chem. Int. Ed. 2012, 51, 6971.
doi: 10.1039/B702940N |
|
|
(c) Qin, C.; Zhou, W.; Chen, F.; Ou, Y.; Jiao, N. Angew. Chem. Int. Ed. 2011, 50, 12595.
doi: 10.1039/B702940N |
|
|
(d) Klein, J. E. M. N.; Plietker, B. Org. Biomol. Chem. 2013, 11, 1271.
doi: 10.1039/B702940N |
|
|
(e) Kulinkovich, O. G. Chem. Rev. 2003, 103, 2597.
doi: 10.1039/B702940N |
|
|
(f) Bart, S. C.; Chirik, P. J. J. Am. Chem. Soc. 2003, 125, 886.
doi: 10.1039/B702940N |
|
|
(g) Seiser, T.; Cramer, N. J. Am. Chem. Soc. 2010, 132, 5340.
doi: 10.1039/B702940N |
|
|
(h) Seiser, T.; Saget, T.; Tran, D. N.; Cramer, N. Angew. Chem. Int. Ed. 2011, 50, 7740.
doi: 10.1039/B702940N |
|
|
(i) Jun, C-H. Chem. Soc. Rev., 2004, 33, 610.
doi: 10.1039/B702940N |
|
| [3] |
(a) Kumar, K. A. A.; Venkateswarlu, V.; Vishwakarma, R. A.; Sawant, S. D. Synthesis 2015, 47, 3161.
doi: 10.1055/s-00000084 |
|
(b) Zhang, J.; Jiang, J.; Li, Y.; Zhao, Y.; Wan, X. Org. Lett. 2013, 15, 3222.
doi: 10.1055/s-00000084 |
|
| [4] |
Liu L.; Ishida N.; Murakami M. Angew. Chem. Int. Ed. 2012, 51 2485.
doi: 10.1002/anie.201108446 |
| [5] |
(a) Wang, M.; Lu, J.; Zhang, X.; Li, L.; Li, H.; Luo, N.; Wang, F. ACS Catal. 2016, 6, 6086.
doi: 10.1021/acscatal.6b02049 |
|
(b) Liu, H.; Wang, M. Li, H.; Luo, N.; Xu, S.; Wang, F. J. Catal. 2017, 346, 170.
doi: 10.1021/acscatal.6b02049 |
|
|
(c) Wang, M.; Lu, J.; Li, L.; Li, H.; Liu, H.; Wang, F. J. Catal. 2017, 348, 160.
doi: 10.1021/acscatal.6b02049 |
|
|
(d) Anjum, A.; Srinivas, P. Chem. Lett. 2001, 9, 900.
doi: 10.1021/acscatal.6b02049 |
|
|
(e) Baruah, S.; Borthakur, S.; Gogoi, S. Chem. Commun. 2017, 53, 9133.
doi: 10.1021/acscatal.6b02049 |
|
|
(f) Nakamura, R.; Obora, Y.; Ishii, Y. Adv. Synth. Catal. 2009, 351, 1677.
doi: 10.1021/acscatal.6b02049 |
|
|
(g) Minisci, F.; Recupero, F.; Pedulli, G. F.; Lucarini, M. J. Mol. Catal. A 2003, 204~205, 63.
doi: 10.1021/acscatal.6b02049 |
|
|
(h) Minisci, F.; Recupero, F.; Fontana, F.; Bjϕrsvik, H˗R.; Liguori, L. Synlett. 2002, 610.
doi: 10.1021/acscatal.6b02049 |
|
|
(i) Sathyanarayana, P.; Ravi, O.; Muktapuram, P. R.; Bathula, S. R. Org. Biomol. Chem. 2015. 13, 9681.
doi: 10.1021/acscatal.6b02049 |
|
| [6] |
(a) Xu, L.; Wang, S.; Chen, B.; Li, M.; Hu, X.; Hu, B.; Jin, L.; Sun, N.; Shen, Z. Synlett. 2018, 1505.
doi: 10.1080/00397910500503611 |
|
(b) Shaikh, T. M. A.; Sudalai, A. Eur. J. Org. Chem. 2008, 4877.
doi: 10.1080/00397910500503611 |
|
|
(c) Santaniello, E.; Manzocchi, A.; Farachi, C. Synthesis 1980, 563.
doi: 10.1080/00397910500503611 |
|
|
(d) Lee, J. C.; Lee, J. M. Synth. Commun. 2006, 36, 1071.
doi: 10.1080/00397910500503611 |
|
|
(e) Lee, J. C.; Choi, J. H.; Lee, Y. C. Synlett. 2001, 1563.
doi: 10.1080/00397910500503611 |
|
| [7] |
(a) Liu, H.; Li, H.; Lu, J.; Zeng, S.; Wang, M.; Luo, N.; Xu, S.; Wang, F. ACS Catal. 2018, 8, 4761.
doi: 10.1021/acscatal.8b00022 |
|
(b) Hirashima, S˗i.; Nobuta, T.; Tada, N.; Itoh, A. Synlett 2009, 2017.
doi: 10.1021/acscatal.8b00022 |
|
|
(c) Yamaguchi, T.; Matsusaki, Y.; Tada, N.; Miura, T.; Itoh, A. Photochem. Photobiol. Sci. 2013, 12, 417.
doi: 10.1021/acscatal.8b00022 |
|
| [8] |
(a) Sathyanarayana, P.; Upare, A.; Ravi, O.; Muktapuram, P. R.; Bathula, S. R. RSC Adv. 2016, 6, 22749.
doi: 10.1039/C6RA02962K |
|
(b) Angeles, N. A.; Villavicencio, F.; Guadarrama, C.; Corona, D.; Cuevas˗Yañez, E. J. Braz. Chem. Soc. 2010, 21, 905.
doi: 10.1039/C6RA02962K |
|
|
(c) Cao, L.; Ding, J.; Gao, M.; Wang, Z.; Li, J.; Wu, A. Org. Lett. 2009, 11, 3810.
doi: 10.1039/C6RA02962K |
|
|
(d) Rajendar, K.; Kant, R.; Narender, T. Adv. Synth. Catal. 2013, 355, 3591.
doi: 10.1039/C6RA02962K |
|
| [9] |
(a) Caputo, D. F. J.; Arroniz, C.; Dürr, A. B.; Mousseau, J. J.; Stepan, A. F.; Mansfield, S. J.; Anderson, E. A. Chem. Sci. 2018, 9, 5295.
doi: 10.1039/C8SC01355A |
|
(b) Ram, R. N.; Soni, V. K. J. Org. Chem. 2015, 80, 8922.
doi: 10.1039/C8SC01355A |
|
|
(c) Luo, J.; Zhang, X.; Zhang, J. ACS Catal. 2015, 5, 2250.
doi: 10.1039/C8SC01355A |
|
|
(d) Xu, G˗C.; Yu, H˗L.; Zhang, X˗Y.; Xu, J˗H. ACS Catal. 2012, 2, 2566.
doi: 10.1039/C8SC01355A |
|
|
(e) Neumann, M.; Füldner, S.; König, B.; Zeitler, K. Angew. Chem. Int. Ed. 2011, 50, 951.
doi: 10.1039/C8SC01355A |
|
|
(f) Jiang, H.; Bak, J. R.; López˗Delgado, F. J.; Jørgensen, K. A. Green Chem. 2013, 15, 3355.
doi: 10.1039/C8SC01355A |
|
|
(g) Zhou, F.; Hu, X.; Gao, M.; Cheng, T.; Liu, G. Green Chem. 2016, 18, 5651.
doi: 10.1039/C8SC01355A |
|
|
(h) Toma, T.; Shimokawa, J.; Fukuyama, T. Org. Lett. 2007, 9, 3195.
doi: 10.1039/C8SC01355A |
|
| [10] |
Yu S.; Liu S.; Lan Y.; Wan B.; Li X. J. Am. Chem. Soc. 2015, 137 1623.
doi: 10.1021/ja511796h |
| [11] |
(a) Tong, W.; Li, W.˗H.; He, Y.; Mo, Z.˗Y.; Tang, H.˗T.; Wang, H.˗S.; Pan, Y.˗M. Org. Lett. 2018, 20, 2494.
doi: 10.1021/acs.orglett.8b00886 |
|
(b) He, Y.; Wang, Y.; Liang, X.; Huang, B.; Wang, H.; Pan, Y.˗M.; Org. Lett. 2018, 20, 7117.
doi: 10.1021/acs.orglett.8b00886 |
|
|
(c) Huang, X.˗Y.; Ding, R.; Mo, Z.˗Y.; Xu, Y.˗L.; Tang, H.˗T.; Wang, H.˗S.; Chen, Y.˗Y.; Pan, Y.˗M. Org. Lett. 2018, 20, 4819.
doi: 10.1021/acs.orglett.8b00886 |
|
|
(d) Zhang, B.; Studer, A. Chem. Soc. Rev. 2015, 44, 3505.
doi: 10.1021/acs.orglett.8b00886 |
|
|
(e) Wang, X.; Xiong, W.; Huang, Y.; Zhu, J.; Hu, Q.; Wu, W.; Jiang, H. Org. Lett. 2017, 19, 5818.
doi: 10.1021/acs.orglett.8b00886 |
|
|
(f) Hu, W.; Li, J.; Xu, Y.; Li, J.; Wu, W.; Liu, H.; Jiang, H. Org. Lett. 2017, 19, 678.
doi: 10.1021/acs.orglett.8b00886 |
|
|
(g) Peng, J.; Gao, Y.; Hu, W.; Gao, Y.; Hu, M.; Wu, W.; Ren, Y.; Jiang, H. Org. Lett. 2016, 18, 5924.
doi: 10.1021/acs.orglett.8b00886 |
|
| [12] |
(a) Tan, L.; Chen, C.; Liu, W. Beilstein J. Org. Chem. 2017, 13, 1079.
doi: 10.3762/bjoc.13.107 |
|
(b) Jia, W˗G.; Zhang, H.; Li, D˗D.; Yan, L˗Q. RSC Adv. 2016, 6, 27590.
doi: 10.3762/bjoc.13.107 |
| No related articles found! |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||