

Chinese Journal of Organic Chemistry >
Synthesis and Antidiabetic Activity of Novel Molecules Containing p-Aminobenzoic Acid and Benzenesulfonamide Moiety
Received date: 2012-04-13
Revised date: 2012-05-31
Online published: 2012-10-27
Supported by
Project supported by the Scientific and Technological Project in Chongqing City (Nos. 2011AB5001, 2011AC1053, 2011AC5107).
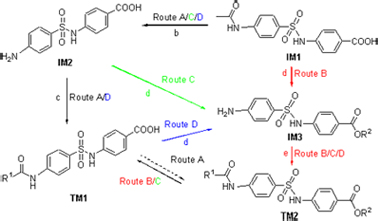
A new class of target molecules containing p-aminobenzoic acid and benzenesulfonamide moiety is reported, based on the structural features of a series of analogues with strong biological activities previously synthesized by the group. By taking 4 discrete synthetic routes, the practical procedures and facile preparative routes for both the intermediates of IM1~IM3 and the target molecules of TM1 and TM2 were established. A total of 26 designed compounds were synthesized smoothly using the established synthetic approaches under mild reaction conditions, with low cost and high yields (64%~95%). The chemical structures of 21 new compounds were confirmed by 1H NMR, 13C NMR and HRMS techniques. The bioassay test demonstrates weak antidiabetic activity for all the target molecules. This study has further expanded the application of alcohol/SOCl2 system in the deacylation of N-arylacetamides and chloro-N-arylacetamides as well as esterification of carboxy group concomitantly, which is supportive to the structure optimization of novel molecules containing p-aminobenzoic acid and benzenesulfonamide moiety.
Yang Long , Yan Jufang , Fan Li , Chen Xin , Shangguan Ruiyan , Wang Linfa , Yang Dacheng . Synthesis and Antidiabetic Activity of Novel Molecules Containing p-Aminobenzoic Acid and Benzenesulfonamide Moiety[J]. Chinese Journal of Organic Chemistry, 2012 , 32(10) : 1908 -1918 . DOI: 10.6023/cjoc201204016
[1] Xu, Y. P.; Etgen, G. J.; Broderick, C. L.; Canada, E.; Gonzalez, I.; Lamar, J.; Montrose-Rafizadeh, C.; Oldham, B. A.; Osborne, J. J., Xie, C. Y.; Shi, Q.; Winneroski, L. L.; York, J.; Yumibe, N.; Zink, R.; Mantlo, N. J. Med. Chem. 2006, 49, 5649.
[2] Yoshida, K.; Hishida, A.; Iida, O.; Hosokawa, K.; Kawabata, J. J Agric. Food Chem. 2008, 56, 4367.
[3] Mohler, M. L.; He Y. L.; Wu Z. Z.; Hwang, D. J.; Miller, D. D. Med. Res. Rev. 2009, 29, 125.
[4] Kim, S. J.; Jung, M. H.; Yoo, K. H.; Cho, J. H.; Oh, C. H. Bioorg. Med. Chem. Lett. 2008, 18, 5815.
[5] Temperini, C.; Cecchi, A.; Scozzafava, A.; Supuran, C. T. Bioorg. Med. Chem. Lett. 2008, 18, 2567.
[6] Rothschild, C. M.; Hines, M. T.; Breuhaus, B.; Gay, J.; Sellon, D. C. J. Vet. Intern. Med. 2004, 18, 370.
[7] Tanaka, H.; Ohshima, N.; Hidaka, H. Mol. Pharmacol. 1999, 55, 356.
[8] Yang, H. T.; Endoh, M. Eur. J. Pharmacol. 1996, 312, 281.
[9] Hao, G. F.; Yang, G. F. Chin. J. Org. Chem. 2008, 28, 1545 (in Chinese).
(郝格非, 杨光富, 有机化学, 2008, 28, 1545.)
[10] Seri, K.; Kazuko, S.; Katsumi, K.; Yorishige, I.; Hiroyuki, A. Eur. J. Pharmacol. 2000, 389, 253.
[11] Basset, G. J. C.; Quinlivan, E. P.; Ravanel, S.; Rébeillé, F.; Nichols, B. P.; Shinozaki, K.; Seki, M.; Adams-Phillips, L. C.; Giovannoni, J. J.; Gregory III, J. F.; Hanson, A. D. Proc. Natl. Acad. Sci. U. S. A. 2004, 101, 1496.
[12] Pawelczak, K.; Jones, T. R.; Kempny, M.; Jackman, A. L.; Newell, D. R.; Krzyzanowski, L.; Rzeszotarska, B. J. Med. Chem. 1989, 32, 160.
[13] Lahue, B. R.; Wan, Z. K.; Snyder, J. K. J. Org. Chem. 2003, 68, 4345.
[14] Ohkanda, J.; Buckner, F. S.; Lockman, J. W.; Yokoyama, K.; Carrico, D.; Eastman, R.; Luca-Fradley, K. D.; Davies, W.; Croft, S. L.; Voorhis, W. C. V.; Gelb, M. H.; Sebti, S. M.; Hamilton, A. D. J. Med. Chem. 2004, 47, 432.
[15] Sun, J.; Yang, Y. S.; Li, W.; Zhang, Y. B.; Wang, X. L.; Tang, J. F.; Zhu, H. L. Bioorg. Med. Chem. Lett. 2011, 21, 6116.
[16] Prabhu, P. P.; Shastry, C. S.; Pande, S.; Pai, A. J. Pharm. Res. 2011, 4, 2209.
[17] Qiu, J. Y.; Xu, B.; Huang, Z. M.; Pan, W. D.; Cao, P. X.; Liu, C. X.; Hao, X. J.; Song, B. A.; Liang, G. Y. Bioorg. Med. Chem. 2011, 19, 5352.
[18] Chen, X. F.; Wu, Y. B.; Jin, J.; Wang, R. Z.; Wang, C.; Liu, J. Acta Pharm. Sin. 2010, 45, 263 (in Chinese).
(陈晓芳, 武燕彬, 金洁, 王瑞贞, 王翀, 刘浚, 药学学报, 2010, 45, 263.)
[19] Mulongo, G.; Mbabazi, J.; Odongkara, B.; Twinomuhwezi, H.; Mpango, G. B. Res. J. Chem. Sci. 2011, 1, 102.
[20] Pfefferkorn, J. A.; Song, Y. T.; Sun, K. L.; Miller, S. R.; Trivedi, B. K.; Choi, C.; Sorenson, R. J.; Bratton, L. D.; Unangst, P. C.; Larsen, S. D.; Poel, T. J.; Cheng, X. M.; Lee, C.; Erasga, N.; Auerbach, B.; Askew, V.; Dillon, L.; Hanselman, J. C.; Lin, Z. W.; Lu, G.; Robertson, A.; Olsen, K.; Mertz, T.; Sekerke, C.; Pavlovsky, A.; Harris, M. S.; Bainbridge, G.; Caspers, N.; Chen, H. F.; Eberstadt, M. Bioorg. Med. Chem. Lett. 2007, 17, 4538.
[21] Dhananjeyan, M. R.; Trendel, J. A.; Bykowski, C.; Sarver, J. G.; Ando, H.; Erhardt, P. W. J. Chromatogr., B 2008, 867, 247.
[22] Yang, D. C.; Yan, J. F.; Xu, J.; Ye, F.; Zhou, Z. W.; Zhang, W. Y.; Fan, L.; Chen, X. Acta Pharm. Sin. 2010, 45, 66 (in Chinese).
(杨大成, 晏菊芳, 许荩, 叶飞, 周祖文, 张蔚瑜, 范莉, 陈欣, 药学学报, 2010, 45, 66.)
[23] Zhang, K.; Yan, J. F.; Tang, X. M.; Liu, H. P.; Fan, L.; Zhou, G. M.; Yang, D. C. Acta Pharm. Sin. 2011, 46, 412 (in Chinese).
(张坤, 晏菊芳, 唐雪梅, 刘红萍, 范莉, 周光明, 杨大成, 药学学报, 2011, 46, 412.)
[24] Tang, X. M.; Yan, J. F.; Zhang, Y. X.; Zhang, W. Y.; Su, X. Y.; Chen, X.; Zhou, Z. W.; Yang, D. C. Chin. J. Org. Chem. 2009, 29, 1790 (in Chinese).
(唐雪梅, 晏菊芳, 张映霞, 张蔚瑜, 苏小燕, 陈欣, 周祖文, 杨大成, 有机化学, 2009, 29, 1790.)
[25] Song, X. L.; Yan, J. F.; Fan, L.; Chen, X.; Xu, J.; Zhou, Z. W.; Yang, D. C. Chin. J. Org. Chem. 2009, 29, 606 (in Chinese).
(宋小礼, 晏菊芳, 范莉, 陈欣, 许荩, 周祖文, 杨大成, 有机化学, 2009, 29, 606.)
[26] Tang, X. M.; Fan, L.; Yu, H. X.; Liao, Y. H.; Yang, D. C. Chin. J. Org. Chem. 2009, 29, 595 (in Chinese).
(唐雪梅, 范莉, 于红霞, 廖玉华, 杨大成, 有机化学, 2009, 29, 595.)
[27] Novacek, A.; Sedlackova, V.; Korner, J.; Danek, J. CS 266792, 1990 [Chem. Abstr. 1991, 114, 246957].
[28] Rajagopalan, S. Proc.-Indian Acad. Sci., Sect. A 1943, 18A, 108.
[29] Yang, D. C.; Wang, L. F.; Fan, L.; Wang, G. B.; Shang-Guan, R. Y.; Sun, J.; Li, C. Z.; Yang, L. CN 102153481, 2011 [Chem. Abstr. 2011, 155, 352231].
[30] Wang, G. B.; Wang, L. F.; Li, C. Z.; Sun, J.; Zhou, G. M.; Yang, D. C. Res. Chem. Intermed. 2012, 38, 77.
[31] Meshram, G. A.; Patil, V. D. Tetrahedron Lett. 2009, 50, 1117.
[32] Bahrami, K.; Khodaei, M. M.; Soheilizad, M. J. Org. Chem. 2009, 74, 9287.
[33] Palakurthy, N. B.; Mandal, B. Tetrahedron Lett. 2011, 52, 7132.
[34] Yu, X. F.; Lu, X. Y. Adv. Synth. Catal. 2011, 353, 2805.
[35] Luo, X. X.; Bai, H.; Xue, X. Y.; Hou, Z.; Zhou, Y.; Sang, G. J.; Meng, J. R. CN 101857562, 2010 [Chem. Abstr. 2010, 153, 554743].
[36] Pastor-Navarro, N.; García-Bover, C.; Maquieira, á.; Puchades, R. Anal. Bioanal. Chem. 2004, 379, 1088.
[37] Mirza, S. M.; Mustafa, G.; Khan, I. U.; Zia-Ur-Rehman, M.; Shafiq, M. Acta Crystallogr., Sect. E 2010, E67, o25.
[38] Chitranshi, P.; Xue, L. Bioorg. Med. Chem. Lett. 2011, 21, 6357.
[39] Motoshima, K.; Ishikawa, M.; Hashimoto, Y.; Sugita, K. Bioorg. Med. Chem. 2011, 19, 3156.
[40] Ismail, N. S. M.; Hattori, M. Bioorg. Med. Chem. 2011, 19, 374.
[41] Hutchinson, J. H.; Li, Y. W.; Arruda, J. M.; Baccei, C.; Bain, G.; Chapman, C.; Correa, L.; Darlington, J.; King, C. D.; Lee, C.; Lorrain, D.; Prodanovich, P.; Rong, H. J.; Santini, A.; Stock, N.; Prasit, P.; Evans, J. F. J. Med. Chem. 2009, 52, 5803.
[42] Suryadevara, P. K.; Olepu, S.; Lockman, J. W.; Ohkanda, J.; Karimi, M.; Verlinde, C. L. M. J.; Kraus, J. M.; Schoepe, J.; Voorhis, W. C. V.; Hamilton, A. D.; Buckner, F. S.; Gelb, M. H. J. Med. Chem. 2009, 52, 3703.
[43] Takaya, J.; Miyashita, Y.; Kusama, H.; Iwasawa, N. Tetrahedron 2011, 67, 4455.
[44] Rewcastle, G. W.; Gamage, S. A.; Flanagan, J. U.; Frederick, R.; Denny, W. A.; Baguley, B. C.; Kestell, P.; Singh, R.; Kendall, J. D.; Marshall, E. S.; Lill, C. L.; Lee, W. J.; Kolekar, S.; Buchanan, C. M.; Jamieson, S. M. F.; Shepherd, P. R. J. Med. Chem. 2011, 54, 7105.
[45] Brenna, E.; Fuganti, C.; Gatti, F. G.; Parmeggiani, F. Tetrahedron: Asymmetry 2009, 20, 2594.
[46] Sliman, F.; Desmaele, D. Synthesis 2010, 619.
[47] Shi, G. Q.; Dropinski, J. F.; McKeever, B. M.; Xu, S. H.; Becker, J. W.; Berger, J. P.; MacNaul, K. L.; Elbrecht, A.; Zhou, G. C.; Doebber, T. W.; Wang, P. R.; Chao, Y. S.; Forrest, M.; Heck, J. V.; Moller, D. E.; Jones, A. B. J. Med. Chem. 2005, 48, 4457.
[48] Kumar, A.; Maurya, R. A.; Sharma, S.; Ahmad, P.; Singh, A. B.; Tamrakar, A. K.; Srivastava, A. K. Bioorg. Med. Chem. 2009, 17, 5285.
[49] Yang, D. C.; Yan, J. F.; Wang, L. F.; Ye, F.; Fan, L.; Mou, X.; Li, T. J.; Liu, H. L.; Chen, W. D.; Xu, J.; Song, X. L. CN 101735286, 2010 [Chem. Abstr. 2010, 153, 116535].
[50] Corradi, V.; Mancini, M.; Santucci, M. A.; Carlomagno, T.; Sanfelice, D.; Mori, M.; Vignaroli, G.; Falchi, F.; Manetti, F.; Radi, M.; Botta, M. Bioorg. Med. Chem. Lett. 2011, 21, 6867.
[51] Yao, Z. Y.; Yang, D. C.; Fan, L. Chin. J. Med. Chem. 2003, 13, 16 (in Chinese).
(姚志勇, 杨大成, 范莉, 中国药物化学杂志, 2003, 13, 16.)
[52] Gupta, P.; Paul, S. Green Chem. 2011, 13, 2365.
[53] Rajput, A. P.; Gore, R. P. Der Pharma Chem. 2011, 3, 409.
[54] Balaskar, R. S.; Gavade, S. N.; Mane, M. S.; Shingare, M. S.; Mane, D. V. Green Chem. Lett. Rev. 2011, 4, 91.
[55] Chou, C. T.; Yellol, G. S.; Chang, W. J.; Sun, M. L.; Sun, C. M. Tetrahedron 2011, 67, 2110.
[56] Berger, J.; Moller, D. E. Annu. Rev. Med. 2002, 53, 409. Krentz, A. J.; Bailey, C. J. Drugs 2005, 65, 385.
/
| 〈 |
|
〉 |