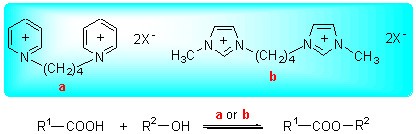
A series of functional binuclear ionic liquids based on bis-(3-methyl-1-imidazole)butylidene double P-toluene sulfonic acid salt (Im-PTSA), bis-(3-methyl-1-imidazole)butylidene double bisulfate (Im-HSO4), bis-(1-pyridine)butylidene double p-toluene sulfonic acid salt (Py-PTSA), bis-(1-pyridine)butylidene double bisulfate (Py-HSO4) were synthesized by a two-step proceeding and their structures were characterized by FT-IR and 1H NMR spectra. Their thermal stabilities were characterized by TG. In addition, the acidity and solubility of functional binuclear ionic liquids were also studied. The catalytic activity of the binuclear ionic liquids for the esterification of succinic acid with ethanol was measured. The results show that under the optimized conditions of n(succinic acid):n(ethanol)=1:3, catalyst used dosage 1.90% (wt), 70 ℃ and 2.5 h, the yield of diethyl succinate reached 93.6% and the selectivity was near up to 100%. Im-PTSA was reused at least 8 times without significant decrease in activity after drying under vacuum. Austenitic stainless steel 316L was used for conducting the corrosion test under the above esterificaion condition, the corrosion rates of the steel plates dipped in the systems with these ionic liquids were less than one tenth of that with sulfuric acid. Fischer esterification of monocarboxylic acids and dicarboxylic acids with different alcohols was observed on using Im-PTSA as catalyst which gave high product yield and selectivity. Use of such a reaction catalyst should be appreciated for its convenient separation.
[1] Joseph, T.; Sahoo, S.; Halligudi, S. B. J. Mol. Catal. A: Chem. 2005, 234, 107.
[2] Xie, C.; Li, H.; Li, L.; Yu, S.; Liu, F. J. Hazard. Mater. 2008, 151, 847.
[3] Li, X.; Eli, W. J. Mol. Catal. A: Chem. 2008, 279, 159.
[4] Li, X.; Lin, Q.; Ma, L. Ultrason. Sonochem. 2010, 17, 752.
[5] Sunitha, S.; Kanjilal, S.; Reddy, P. S.; Prasad, R. B. N. Tetrahedron Lett. 2007, 48, 6962.
[6] Janus, E.; Goc-Maciejewska, I.; Lozynski, M.; Pernak, J. Tetrahedron Lett. 2006, 47, 4079.
[7] Durand, J.; Teuma, E.; Gómez, M. C. R. Chim. 2007, 10, 152.
[8] Cai, S.; Wang, S. Chin. J. Chem. Eng. 2011, 19, 57.
[9] Zhao, Y.; Li, Z.; Xia, C. Chin. J. Catal. 2011, 32, 440.
[10] Deng, Y.; Shi, F.; Beng, J.; Qiao, K. J. Mol. Catal. A: Chem. 2001, 165, 33.
[11] Fraga-Dubreuil, J.; Bourahla, K.; Rahmouni, M.; Bazureau, J. P.; Hamelin, J. Catal. Commun. 2002, 3, 185.
[12] Cole, A. C.; Jensen, J. L. J. Am. Chem. Soc. 2002, 124, 5962.
[13] Kou, Y.; Yang, Y.-L. Petrochem. Technol. 2004, 33, 297 (in Chinese).
(寇元, 杨雅立, 石油化工, 2004, 33, 297.)
[14] Zhang, S.-J.; Lv, X.-M. Ionic Liquid—From Basic Research to Industrial Applications, Science Press, Beijing, 2006, p. 71 (in Chinese).
(张锁江, 吕兴梅, 离子液体——从基础研究到工业应用, 科学出版社, 北京, 2006, p. 71.)
[15] Lv, X.-M. M.S. Thesis, Nanjing University, Nanjing, 2011 (in Chinese).
(吕学铭, 硕士论文, 南京大学, 南京, 2011.)
[16] Bermúdez, M. D.; Jiménez, A. E.; Martínez-Nicolás, G. Appl. Surf. Sci. 2007, 253, 7295.
[17] Ganeshpure, P. A.; George, G.; Das, J. J. Mol. Catal. A: Chem. 2008, 279, 182.
[18] Liang, J.-H.; Xu, Y.; Ren, X.-Q.; Jiang, M.; Li, Z.-J. Acta PetroleI Sin. 2011, 27, 482 (in Chinese).
(梁金花, 徐玥, 任晓乾, 姜岷, 李振江, 石油学报, 2011, 27, 482.)
[19] Huang, B.-H.; Li, Z.-J.; Shi, N.; Xu, X.-L.; Zhang, K.; Fang, Y.-X. Chin. J. Org. Chem. 2009, 29, 770 (in Chinese).
(黄宝华, 黎子进, 史娜, 徐效陵, 张焜, 方岩雄, 有机化学, 2009, 29, 770.)


