

Chinese Journal of Organic Chemistry >
Synthesis and Antitumor Activity of O,O'-Dialkyl-α-phenyl-α-(substituted benzoylanoxy)-methylphosphonate
Received date: 2012-11-20
Revised date: 2012-12-20
Online published: 2013-01-07
Supported by
Project supported by the National Natural Science Foundation of China (No. 81160385) and the Science and Technology Department of Guizhou Province (No. LKZ[2010]47).
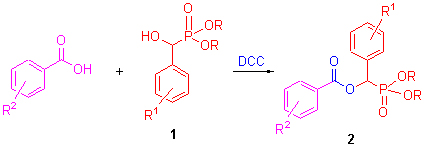
In search of more effective anticancer agents, the title compounds were prepared via the esterification reaction of α-hydroxyphosphonate and substituted bezoic acid with N,N'-dicyclohexylcarbodiimide (DCC) as condensing agent. Twelve of them were reported for the first time. Their structures were clearly established by elemental analysis, IR, 1H NMR and 13C NMR spectra. The antitumor activities of target compounds were tested for the first time. The result indicated that they exhibited certain antitumor activities, and compounds 2i, 2k, 2m showed more potent activities against SGC-7901 with IC50 values of 18.8, 18.5 and 24.5 μmol/L, against A-549 with IC50 values of 24.1, 29.0 and 20.7 μmol/L. Meanwhile, preliminary structure-activity relationships of the title compounds were studied.
Key words: α-hydroxyphosphonate; synthesis; esterification; antitumor activity
Yang Jiaqiang , Gu Qing , Shu Bo , Song Baoan , Liu Jingzi . Synthesis and Antitumor Activity of O,O'-Dialkyl-α-phenyl-α-(substituted benzoylanoxy)-methylphosphonate[J]. Chinese Journal of Organic Chemistry, 2013 , 33(05) : 1113 -1118 . DOI: 10.6023/cjoc201211038
/
| 〈 |
|
〉 |