

Chinese Journal of Organic Chemistry >
Synthesis and Antifungal Activity of 2-Carbalkoxyalkyl/(n-butyl)-4-aryl-1,5-benzothiazepines
Received date: 2013-10-23
Revised date: 2013-01-18
Online published: 2013-02-22
Supported by
Project supported by the National Natural Science Foundation of China (No. 20972040), the Research Project of Education Department of Hebei Province of China (No. Q2012001), and the Scientific Research Fund of Hebei College of Industry and Technology (No. Qz1102).
A series of novel 2-substituted-1,5-benzothiazepines were synthesized successfully and their structures were determined by elemental analysis, IR, MS/HRMS and 1H NMR. The synthetic conditions of 2-substituted-4-aryl-1,5-benzothiazepines were investigated. The antifungal activities of these 2-substituted-1,5-benzothiazepine derivatives were tested and the results confirmed that the 2-ethyl carboxylate was necessary for antifungal activity in 1,5-benzothiazepine compounds 1.
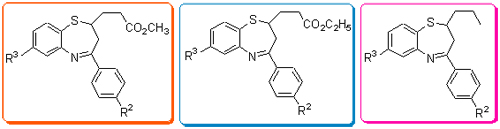
Li Wenhong , Liu Xiying , Zhang Bo , Li Yuan . Synthesis and Antifungal Activity of 2-Carbalkoxyalkyl/(n-butyl)-4-aryl-1,5-benzothiazepines[J]. Chinese Journal of Organic Chemistry, 2013 , 33(07) : 1503 -1508 . DOI: 10.6023/cjoc201210044
/
| 〈 |
|
〉 |