

Chinese Journal of Organic Chemistry >
Synthesis and Crystal Structure of 12-(2-Chlorophenyl)-7,11,13,16-tetraoxaspiro[5.2.5.2]cetane
Received date: 2013-03-02
Revised date: 2013-04-19
Online published: 2013-04-24
Supported by
Project supported by the Construct Program of the Key Discipline in Hunan Province (No. 2011-76), the Science and Technology Planning Project of Hunan Province (No. 2013FJ3004), the Scientific Research Fund of Hunan Provincial Education Department (Nos. 13A030, 013B029), and the Science and Technology Innovative Research Team in Higher Educational Institutions of Hunan Province (No. 2012-318)
12-(2-Chlorophenyl)-7,11,13,16-tetraoxaspiro[5.2.5.2]cetane (2) was prepared by cyclohexanone and 5,5-bis(hydroxymethyl)-2-(2-chlorophenyl)-1,3-dioxane (1) which synthesized from the reaction of the o-chlorobenzaldehyde and pentaerythriol, or prepared by o-chlorobenzaldehyde and 9,9-bis(hydroxymethyl)-7,11-dioxaspiro[5,5]undecane (3) which synthesized from the reaction of the cyclohexanone and pentaerythriol. Compounds 1 and 2 were characterized by X-ray diffraction, elemental analysis, 1H NMR. Single crystal X-ray diffraction analyses reveal that compound 1 belongs to monoclinic system with space group P2(1)/n with a=0.56768(15) nm, b=0.9807(3) nm, c=2.1785(6) nm, a=90°, b=90.204(4)°, g=90°, V= 1.2128(5) nm3, Z=4, Dc=1.417 Mg/m3, μ=0.315 mm-1, F(000)=544. Compound 2 also belongs to monoclinic system with space group C2/c with a=1.5894(5) nm, b=1.5704(4) nm, c=2.8369(9) nm, a=90°, b=101.103(5)°, g=90°, V=6.948(4) nm3, Z=16, Dc=1.296 Mg/m3, μ=0.237 mm-1, F(000)=2880.
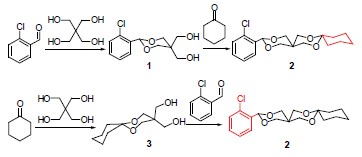
Key words: o-chlorobenzaldehyde; pentaerythriol; crystal structure; 1H NMR
Yuan Lin , Ou Guangchuan , Shi Jianghua , Zhang Min , Yuan Xianyou . Synthesis and Crystal Structure of 12-(2-Chlorophenyl)-7,11,13,16-tetraoxaspiro[5.2.5.2]cetane[J]. Chinese Journal of Organic Chemistry, 2013 , 33(08) : 1817 -1821 . DOI: 10.6023/cjoc201303003
/
| 〈 |
|
〉 |