

Chinese Journal of Organic Chemistry >
Synthesis and Anti-tumor Activities of Resveratrol Derivatives on Cervical Cancer HeLa Cells
Received date: 2013-03-23
Revised date: 2013-04-26
Online published: 2013-05-06
Supported by
Project Supported by the Innovation Program of Shanghai Municipal Education Commission (No.12ZZ188), the Science and Technology Commission of Shanghai Municipal (No.11430502500), the Professor of Special Appointment (Eastern Scholar) at Shanghai Institutions of Higher Learning (No.405ZK110060002).
Nine novel resveratrol derivatives were synthesized from resveratrol, chloroacetyl chloride, p-toluenesulfonyl chloride and p-methyl benzoyl chloride.These structures were confirmed by IR, 1H NMR, 13C NMR and HR-MS techniques.The target compounds were evaluated for their anti-tumor activities against cervical cancer HeLa cells by 3-(4,5-dimethylthi- azol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) method, and the results indicated that compounds 4a, 6a, 7b, 8b and 11c of all the derivatives displayed better activities than resveratrol against HeLa cells, the compounds 6a and 8b displayed excellent inhibition effect, with IC50 values of 22.7 and 18.0 μmol/L, respectively, even higher than that of resveratrol (IC50=114 μmol/L).The inhibitory ratio of compounds 6a and 8b were 95.5% and 87.7% against HeLa cells at the concentration of 150 μmol/L.
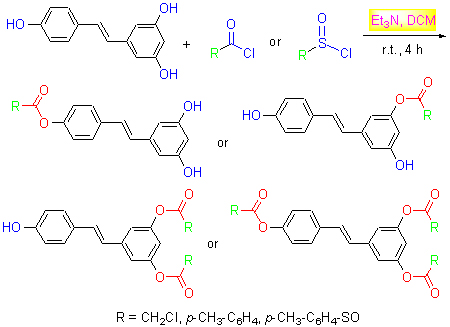
Key words: resveratrol; derivative; synthesis; HeLa cell; anti-tumor activities
Du Cheng , Ren Yujie , Wang Qingwei , Jin Lu . Synthesis and Anti-tumor Activities of Resveratrol Derivatives on Cervical Cancer HeLa Cells[J]. Chinese Journal of Organic Chemistry, 2013 , 33(06) : 1279 -1283 . DOI: 10.6023/cjoc201303035
/
| 〈 |
|
〉 |