

Chinese Journal of Organic Chemistry >
Synthesis and Cytotoxicity of N-Benzylidene Hesperitin Hydrazones and Analogues
Received date: 2013-04-18
Revised date: 2013-05-22
Online published: 2013-05-31
Supported by
Project supported by the National Natural Science Foundation of China (No. 81060261) and the Natural Science Foundation of Guangxi Province (No. 2011jjD20002)
Hesperitin hydrazone (2) was generated by the reaction of natural hesperitin (1) as raw material with hydrazine hydrate. Eight N-benzylidene hesperitin hydrazones 3~10 and six analogues 11~16 were synthesized by the reaction of 2 with various aldehydes, respectively. The structures of all reaction products were confirmed by NMR, IR and HR-MS techniques. And the cytotoxity against human cancer cell SGC-7901 of the synthesized compounds was also evaluated. The results showed 5, 7, 10 and 16 exhibiting distinct cytotoxity against SGC-7901.
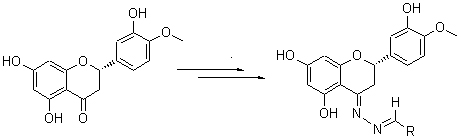
Key words: hesperitin; hesperitin hydrazones; N-benzylidene; cytotoxicity
Liu Zhiping , Wei Wanxing , Gan Chunfang , Huang Yanmin , Liu Sheng , Zhou Min , Cui Jianguo . Synthesis and Cytotoxicity of N-Benzylidene Hesperitin Hydrazones and Analogues[J]. Chinese Journal of Organic Chemistry, 2013 , 33(9) : 1988 -1993 . DOI: 10.6023/cjoc201304028
/
| 〈 |
|
〉 |