

Chinese Journal of Organic Chemistry >
Synthesis and Biological Activity of Novel Phosphoramide with Hydantoin
Received date: 2013-05-14
Revised date: 2013-06-11
Online published: 2013-06-21
Supported by
Project supported by the National Key Technologies R&D Program (No. 2011BAE06B03), the National Natural Science Foundation of China (No. 20772150) and the National Key Laboratory of Elemento-Organic Chemistry in Nankai University (No. 0902, 201003).
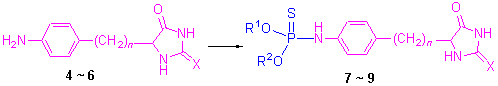
Based on the synthesis of phosphates and phosphorylamidates with hydantoin, thirty and eight new O,O-dialkyl-(aryl)-5-[4-aminophenyl(benzyl)]-2,4-imidazolidinedione thiophosphoramide derivatives with hydantoin were synthesized by the reaction of O,O-dialkyl(aryl) thiophosphoryl chloride with 5-(4-aminophenyl)-and 5-(4-aminobenzyl)-2,4-imidazolidine-dione intermediates, and their structures were confirmed by 1H NMR, 31P NMR, IR, elemental analysis and X-ray diffraction. 9b: C14H20N3O3PS2·1/3H2O, Mr=379.43, triclinic, space group P-1, a=0.82092(5) nm, b=1.79810(14) nm, c=2.0153(2) nm, α=72.289(8)°, β=79.355(7)°, γ=77.288(6)°, V=2.7421(4) nm3, Dc=1.379 g/cm3, Z=6, F(000)=1196, μ(Mo Kα)=0.397 mm-1, S=1.031, final R=0.0426, wR=0.0797. The preliminary bioassay showed that O,O-dimethyl-5-(4-amino-phenyl)-2,4-imidazolidinedione thiophosphoramide (7a), O-ethyl-O-phenyl-5-(4-aminobenzyl)-2,4-imidazolidine-dione thio-phosphoramide (9n) and O,O-dimethyl-5-(3-aminophenyl)-2-thio-2,4-imidazolidinedione thiophosphoramide (11a)have inhibitory rates 75.4%, 80.5% and 81.7% against Brasica campestris at the concentration of 100 μg/mL, while eleven compounds showed excellent insecticidal activities against Myzus Persicae with the LD50 data in the range of 182.41~368.52 μg/mL, respectively.
Wang Jinmin , Xu Zhihong , Han Jintao , Dong Hongbo , Liu Bin , Wang Mingan . Synthesis and Biological Activity of Novel Phosphoramide with Hydantoin[J]. Chinese Journal of Organic Chemistry, 2013 , 33(10) : 2186 -2195 . DOI: 10.6023/cjoc201305023
/
| 〈 |
|
〉 |