

Chinese Journal of Organic Chemistry >
Synthesis and Liquid Crystal Property of 4,5-Disubstituted Phenanthrene and 9,10-Dihydrophenanthrene Derivatives
Received date: 2013-06-20
Revised date: 2013-07-15
Online published: 2013-07-26
Supported by
Project supported by the National Natural Science Foundation of China (No. 21032004), the Specialized Research Fund for the Doctoral Program of Higher Education of China (No. 20110002110051) and the National Electronics and Information Industry Development Fund.
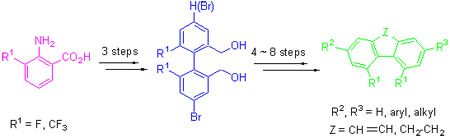
Using 2-amino-3-substituted benzoic acid as starting material, 4, 5-disubstituted phenanthrene derivatives were synthesized via sequential reactions of diazotization-coupling reaction, esterification, reduction, oxidation and cyclization, and using reduced biphenyl intermediates as starting materials. 4, 5-Disubstituted 9, 10-dihydrophenanthrene derivatives were syn-thesized via sequential reactions of bromination, grinard reaction, using 4, 5-disubstituted phenanthrene and 4, 5-disubstituted 9, 10-dihydrophenanthrene derivatives as starting materials. Liquid crystal monomer compounds were synthesized via sequential reactions of halogenations and coupling reaction, and the chemical structures of products were identified by 1H NMR, 13C NMR spectra.
Cao Jianhua , Li Min , Sui Yan , Hua Ruimao . Synthesis and Liquid Crystal Property of 4,5-Disubstituted Phenanthrene and 9,10-Dihydrophenanthrene Derivatives[J]. Chinese Journal of Organic Chemistry, 2013 , 33(11) : 2349 -2356 . DOI: 10.6023/cjoc201306029
[1] Xu,H.Ph.D.Dissertation,Jilin University,Changchun,2009 (in Chinese).(许海,博士论文,吉林大学,长春,2009.)
[2] Dubois,F.; Gingras,M.Tetrahedron Lett.1998,39,5039.
[3] Kharasch,M.S.; Nudenberg,W.; Fields,E.K.J.Am.Chem.Soc.1944,66,1276.
[4] Campeau,L.C.; Parisien,M.; Jean,A.; Fagnou,K.J.Am.Chem.Soc.2006,128,581.
[5] Brown,C.; Sikkel,B.J.; Carvalho,C.F.; Sargent,M.V.J.Chem.Soc.,Perk.Trans.1 1982,3007.
[6] Shi,M.; Xu,B.J.Org.Chem.2002,67,294.
[7] Kuninobu,Y.; Tatsuzaki,T.; Matsuki,T.; Takai,K.J.Org.Chem.2011,76,7005.
[8] Some,S.; Dutta,B.; Ray,J.K.Tetrahedron Lett.2006,47,1221.
[9] Bacon,L.J.Chem.Soc.1958,1375.
[10] Cosmo,R.; Sternhell,S.Aust.J.Chem.1987,40,35.
[11] Yosuke,N.; Takahiro,F.; Sachiko,S.; Jun,N.Chem.Lett.1999,10,1039.
[12] Tian,H.; Shi,J.; Dong,S.; Yan,D.; Wang,L.-X.; Geng,Y.; Wang,F.S.Chem.Commun.2006,33,3498.
[13] Cosmo,R.; Sternhell,S.Australian J.Chem.1987,40,2137.
[14] Witting,G.; Zimmermann,H.Chem.Ber.1953,86,629.
[15] Newman,M.S.; Lilje,K.C.J.Org.Chem.1979,44,4944.
[16] Ramurthy,S.; Lin,X.; Subramanian,S.; Rico,A.C.; Wang,X.M.; Jain,R.; Murray,J.M.; Bashman,S.E.; Warne,R.L.; Shu,W.; Zhou,Y.; Dove,J.; Aikawa,M.; Amiri,P.; Wang,W.; Jensen,J.M.; Wagman,A.S.; Pfister,K.B.; Ng,S.C.WO 2007117607,2007 [Chem.Abstr.2007,147,469365].
[17] Ligtenbarg,A.G.J.; Beuken,E.K.; Meetsma,A.; Veldman,N.; Smeets,W.J.J.J.Chem.Soc.1998,2,263.
[18] Dameron,A.A.; Ciszek,J.W.; Tour,J.M.; Weisc,P.S.J.Phys.Chem.B 2004,108,16761.
[19] Ciszek,J.W.; Tour,J.M.Tetrahedron Lett.2004,45,2801.
[20] Kawamura,M.; Funahashi,M.US 20070029927,2007 [Chem.Abstr.2007,146,216108].
[21] Helms,A.; Heiler,D.; Mclendon,G.J.Am.Chem.Soc.1992,114,6227.
[22] Vonlanthen,D.; Rotzler,J.; Neuburger,M.; Mayor,M.Eur.J.Org.Chem.2010,75,120.
/
| 〈 |
|
〉 |