

Chinese Journal of Organic Chemistry >
Progresses in Syntheses and Applications of Bis-1,2,3-triazoles
Received date: 2013-07-15
Revised date: 2013-08-30
Online published: 2013-09-06
Supported by
Project supported by the National Natural Science Foundation of China (No. 20772035), the 3rd Talents Special Funds of Guangdong Higher Education (No. Guangdong-Finance-Education [2011]431) and the Natural Science Foundation of Guangdong Province (No. S2011010001556).
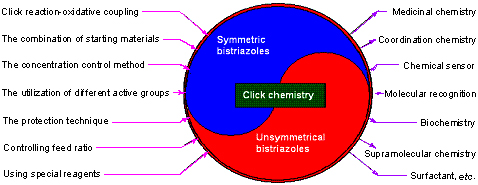
As a kind of important N-heterocyclic compounds, bis-1,2,3-triazoles have drawn more and more interest of chemists in different research territories. In order to synthesize symmetric and unsymmetrical bis-1,2,3-triazoles, many synthetic strategies, methods and techniques, such as click reaction-oxidative coupling, the combination of starting materials, the concentration control method, the utilization of different active groups, the protection technique, the control of feed ratio, the use of special reagents, and so on, have been developed recently. Classified as different substrates and synthetic approaches, these research progresses are summarized. At the same time, the new progresses in the applications of bis-1,2,3-triazoles in different fields, including medicinal chemistry, coordination chemistry, chemical sensor, molecular recognition, biochemistry, supramolecular chemistry and surfactant are also mentioned in this review. It is pointed out that how to cleverly design and efficiently synthesize the novel bis-1,2,3-triazoles for some special purpose via the simple general methods is still a great challenge.
Key words: bis-1,2,3-triazoles; synthesis; click reaction; azides; alkynes; progress
Huo Jingpei , Wei Xinping , Mo Guangzhen , Peng Pai , Zhong Mingli , Chen Renhong , Wang Zhaoyang . Progresses in Syntheses and Applications of Bis-1,2,3-triazoles[J]. Chinese Journal of Organic Chemistry, 2014 , 34(1) : 92 -106 . DOI: 10.6023/cjoc201307021
[1] Meldal, M.; Tornoe, C. W. Chem. Rev. 2008, 108, 2952.
[2] Bonger, K. M.; Hoogendoorn, S.; van Koppen, C. J.; Timmers, C. M.; van der Marel, G. A.; Overkleeft, H. S. ACS Med. Chem. Lett. 2011, 2, 85.
[3] Chan, T. R.; Hilgraf, R.; Sharpless, K. B.; Fokin, V. V. Org. Lett. 2004, 6, 2853.
[4] Wang, Q.; Chan, T. R.; Hilgraf, R.; Fokin, V. V.; Sharpless, K. B.; Finn, M. G. J. Am. Chem. Soc. 2003, 125, 3192.
[5] Service, R. F. Science 2008, 320, 868.
[6] Wu, P.; Feldman, A. K.; Nugent, A. K.; Hawker, C. J.; Scheel, A.; Voit, B.; Pyun, J.; Frechet, J. M. J.; Sharpless, K. B.; Fokin, V. V. Angew. Chem., Int. Ed. 2004, 43, 3928.
[7] Iha, R. K.; Wooley, K. L.; Nystrom, A. M.; Burke, D. J.; Kade, M. J.; Hawker, C. J. Chem. Rev. 2009, 109, 5620.
[8] Xiong, X.-Q.; Chen, H.-X. Chin. J. Org. Chem. 2013, 33, 1437 (in Chinese).
(熊兴泉, 陈会新, 有机化学, 2013, 33, 1437.)
[9] Sokolova, N. V.; Nenajdenko, V. G. RSC Adv. 2013, 3, 16212.
[10] Rostovtsev, V. V.; Green, L. G.; Fokin, V. V.; Sharpless, K. B. Angew. Chem., Int. Ed. 2002, 41, 2596.
[11] Angell, Y.; Burgess, K. Angew. Chem., Int. Ed. 2007, 46, 3649.
[12] Kwon, M.; Jang, Y.; Yoon, S.; Yang, D.; Jeon, H. B. Tetrahedron Lett. 2012, 53, 1606.
[13] Gonzalez, J.; Perez, V. M.; Jimenez, D. O.; Lopez-Valdez G.; Corona, D.; Cuevas-Yanez, E. Tetrahedron Lett. 2011, 52, 3514.
[14] Goyard, D.; Praly, J.-P.; Vidal, S. Carbohydr. Res. 2012, 362, 79.
[15] Jur?ek, O.; Cametti, M.; Pontini, M.; Kolehmainena, E.; Rissanen, K. Org. Biomol. Chem. 2013, 11, 4585.
[16] Lei, M.; Song, W.-Z.; Zhan, Z.-J.; Chen, X.-W.; Wang, Y.-G. Chin. J. Org. Chem. 2010, 30, 1854 (in Chinese).
(雷鸣, 宋汪泽, 詹祖金, 陈小微, 王彦广, 有机化学, 2010, 30, 1854.)
[17] Wang, Z.-X.; Zhao, Z.-G. J. Heterocycl. Chem. 2007, 44, 89.
[18] Huang, Y.-E.; Gard, G.-L.; Shreeve, J.-M. Tetrahedron Lett. 2010, 51, 6951.
[19] Thirumurugan, P.; Matosiuk, D.; Jozwiak, K. Chem. Rev. 2013, 113, 4905.
[20] Oladeinde, O. A.; Hong, S. Y.; Holland, R. J.; Maciag, A. E.; Keefer, L. K.; Saavedra, J. E.; Nandurdikar, R. S. Org. Lett. 2010, 12, 4256.
[21] Gaur, M.; Goel, M.; Sridhar, L.; Ashok, T. D. S.; Prabhakar, S.; Dureja, P.; Raghunathan, P.; Eswaran, S. V. Monatsh. Chem. 2012, 143, 283.
[22] Huigens, R. W.; Rogers, S. A.; Steinhauer, A. T.; Melander, C. Org. Biomol. Chem. 2009, 7, 794.
[23] Karuturi, R.; Al-Horani, R. A.; Mehta, S. C.; Gailani, D.; Desai, U. R. J. Med. Chem. 2013, 56, 2415.
[24] Jurášeka, M.; D?ubákb, P.; Sedlákc, D.; Dvo?ákováa, H.; Hajdúchb, M.; Bart?někc, P.; Drašar, P. Steroids 2013, 78, 356.
[25] Camp, C.; Dorbes, S.; Picard, C.; Benoist, E. Tetrahedron Lett. 2008, 49, 1979.
[26] Hung, H.-C.; Cheng, C.-W.; Ho, I.-T.; Chung, W.-S. Tetrahedron Lett. 2009, 50, 302.
[27] Hung, H.-C.; Cheng, C.-W.; Wang, Y.-Y.; Chen, Y.-J.; Chung, W.-S. Eur. J. Org. Chem. 2009, 6360.
[28] Huang, H.-J.; Fang, H.-Y.; Chir, J.-L.; Wu, A.-T. Luminescence 2011, 26, 518.
[29] Li, C.; Tang, J.; Xie, J. Tetrahedron 2009, 65, 7935.
[30] Romero, T.; Orenes, R. A.; Tárraga, A.; Molina, P. Organo-metallics 2013, 32, 5740.
[31] Wei, P.-F.; Yan, X.-Z.; Li, J.-Y.; Ma, Y.-J.; Yao, Y.; Huang, F.-H. Tetrahedron 2012, 68, 9179.
[32] Ol'shevskaya, V. A.; Makarenkov, A. V.; Kononova, E. G.; Petrovskii, P. V.; Verbitskiy, E. V.; Rusinov, G. L.; Charushin, V. N.; Hey-Hawkins, E.; Kalinin, V. N. Polyhedron 2012, 42, 302.
[33] Zhang, X. J.; Li, H. Y.; You, L. F.; Tang, Y.; Hsung, R. P. Adv. Synth. Catal. 2006, 348, 2437.
[34] Sarmiento-Sánchez, J. I.; Ochoa-Teran, A.; Rivero, I. A. ARKIVOC 2011, 177.
[35] Scott, S. O.; Gavey, E. L.; Lind, S. J.; Gordon, K. C.; Crowley, J. D. Dalton Trans. 2011, 40, 12117.
[36] Guisado-Barrios, G.; Bouffard, J.; Donnadieu, B.; Bertrand, G. Organometallics 2011, 30, 6017.
[37] Schweinfurth, D.; Büttner, N.; Hohloch, S.; Deibel, N.; Klein, J.; Sarkar, B. Organometallics 2013, 32, 5834.
[38] Singh, N.; Metla, B. P. R.; Elias, A. J. J. Organomet. Chem. 2012, 717, 99.
[39] Li, Y.-J.; Huffman, J. C.; Flood, A. H. Chem. Commun. 2007, 2692.
[40] Fletcher, J. T.; Bumgarner, B. J.; Engels, N. D.; Skoglund, D. A. Organometallics 2008, 27, 5430.
[41] Mulla, K.; Shaik, H.; Thompson, D. W.; Zhao, Y. M. Org. Lett. 2013, 15, 4532.
[42] Li, C.; Henry, E.; Mani, N. K.; Tang, J.; Brochon, J. C.; Deprez, E.; Xie, J. Eur. J. Org. Chem. 2010, 2395.
[43] Ruan, Y.-B.; Yu, Y.-H.; Li, C.; Bogliotti, N.; Tang, J.; Xie, J. Tetrahedron 2013, 69, 4603.
[44] Brombosz, S. M.; Appleton, A. L.; Zappas, A. J.; Bunz, U. H. F. Chem. Commun. 2010, 46, 1419.
[45] Shi, W.-J.; Liu, J.-Y.; Ng, D. K. P. Chem. Asian J. 2012, 7, 196.
[46] Zhou, M.-Z.; Zhang, X.; Bai, M.-F.; Shen, D.-W.; Xu, B.-G.; Kao, J.; Ge, X.; Achilefu, X. RSC Adv. 2013, 3, 6756.
[47] Wei, J.-J.; Jin, L.; Wan, K.; Zhou, C.-H. Bull. Korean Chem. Soc. 2011, 32, 229.
[48] Lau, Y. H.; Rutledge, P. J.; Watkinson, M.; Todd, M. H. Chem. Soc. Rev. 2011, 40, 2848.
[49] Hemamalinia, A.; Das, T. M. New J. Chem. 2013, 37, 2419.
[50] Xu, H.-R.; Li, K.; Liu, Q.; Wu, T.-M.; Wang, M.-Q.; Hou, J.-T.; Huang, Z.; Xie, Y.-M.; Yu, X.-Q. Analyst 2013, 2329.
[51] White, N. G.; Beer, P. D. Org. Biomol. Chem. 2013, 11, 1326.
[52] Mohammed, A. I.; Abboud, Z. H.; Alghanimi, A. H. O. Tetrahedron Lett. 2012, 53, 5081.
[53] Mekni, N.; Baklouti, A. J. Soc. Chim. Tunisie 2009, 11, 15.
[54] Jervis, P. J.; Moulis, M.; Jukes, J. P.; Ghadbane, H.; Cox, L. R.; Cerundolo, V.; Besra, G. S. Carbohydr. Res. 2012, 356, 152.
[55] Bedard, A.-C.; Collins, S. K. J. Am. Chem. Soc. 2011, 133, 19976.
[56] Damodiran, M.; Muralidharan, D.; Perumal, P. T. Bioorg. Med. Chem. Lett. 2009, 19, 3611.
[57] Lal, K.; Kumar, A.; Pavan, M. S.; Kaushik, C. P. Bioorg. Med. Chem. Lett. 2012, 22, 4353.
[58] Singh, M. K.; Tilak, R.; Nath, G.; Awasthi, S. K.; Agarwal, A. Eur. J. Med. Chem. 2013, 63, 635.
[59] Skarpos, H.; Osipov, S. N.; Vorob'eva, D. V.; Odinets, I. L.; Lork, E.; Roeschenthaler, G.-V. Org. Biomol. Chem. 2007, 5, 2361.
[60] Pereira, G. R.; Santos, L. J.; Luduvico, I.; Alves, R. B.; de Freitas, R. P. Tetrahedron Lett. 2010, 51, 1022.
[61] Rajesh, R.; Periyasami, G.; Raghunathan, R. Tetrahedron Lett. 2010, 51, 1896.
[62] Megiatto, J. D.; Schuster, D. I. J. Am. Chem. Soc. 2008, 130, 12872.
[63] Mames, I.; Wawrzyniak, U. E.; Wo?ny, M.; Bilewicz, R.; Korybut-Daszkiewicz, B. Dalton Trans. 2013, 42, 2382.
[64] Mekni, N. H.; Baklouti, A. Heterocycles 2012, 85, 1727.
[65] Yoshida, Y.; Takizawa, S.; Sasai, H. Tetrahedron Lett. 2011, 52, 6877.
[66] Lin, H. N. Walsh, C. T. J. Am. Chem. Soc. 2004, 126, 13998.
[67] Krim, J.; Taourirte, M.; Engels, J. W. Molecules 2012, 17, 179.
[68] Huo, J.-P.; Lv, M.-X.; Wang, Z.-Y.; Li, Y.-Z. Chin. J. Chem. 2012, 30, 2411.
[69] Mao, C.-X.; Wang, Z.-Y.; Tan, Y.-H.; Xue, F.-L. Chin. J. Org. Chem. 2011, 31, 1377 (in Chinese).
(毛超旭, 汪朝阳, 谭越河, 薛福玲, 有机化学, 2011, 31, 1377.)
[70] Mo, Y.-Q.; Wang, Z.-Y.; Mei, W.-J.; Fu, J.-H.; Tan, Y.-H.; Luo, S.-H. Monatsh. Chem. 2012, 143, 443.
[71] Luo, S.-H.; Wang, Q.-F.; Wang, Z.-Y.; Peng P. Res. Chem. Intermed. 2013, 39, 2513.
[72] Tan, Y.-H.; Li, J.-X.; Xue, F.-L.; Qi. J.; Wang, Z.-Y. Tetrahedron 2012, 68, 2827.
[73] Huo, J. P.; Luo, J. C.; Wu, W.; Xiong, J. F.; Mo, G. Z.; Wang, Z. Y. Ind. Eng. Chem. Res. 2013, 52, 11850.
[74] Kumar, K.; Sagar, S.; Esau, L.; Kaur, M.; Kumar, V. Eur. J. Med. Chem. 2012, 58, 153.
[75] Kumar, K.; Carrere-Kremer, S.; Kremer, L.; Guerardel, Y.; Biot, C.; Kumar, V. Dalton Trans. 2013, 42, 1492.
[76] Midya, G. C.; Paladhi, S.; Bhowmik, S.; Saha, S.; Dash, J. Org. Biomol. Chem. 2013, 11, 3057.
[77] Elamari, H.; Jlalia, I.; Louet, C.; Herscovici, J.; Meganem, F.; Girard, C. Tetrahedron: Asymmetry 2010, 21, 1179.
[78] Elamari, H.; Meganem, F.; Herscovici, J.; Girard, C. Tetrahedron Lett. 2011, 52, 658.
[79] Elamari, H.; Slimi, R.; Chabot, G. G.; Quentin, L.; Scherman, D.; Girard, C. Eur. J. Med. Chem. 2013, 60, 360.
[80] Isaacman, M. J.; Corigliano, E. M.; Theogarajan, L. S. Biomacro- molecules 2013, 14, 2996.
[81] Zhou, Q. H.; Gui, J. H.; Pan, C.-M.; Albone, E.; Cheng, X.; Suh, E. M.; Grasso, L.; Ishihara, Y.; Baran, P. S. J. Am. Chem. Soc. 2013, 135, 12994.
[82] Sapsford, K. E.; Algar, W. R.; Berti, L.; Gemmill, K. B.; Casey, B. J.; Oh, E.; Stewart, M. H.; Medintz, I. L. Chem. Rev. 2013, 113, 1904.
[83] Beal, D. M.; Albrow, V. E.; Burslem, G.; Hitchen, L.; Fernandes, C.; Lapthorn, C.; Roberts, L. R.; Selby, M. D.; Jones, L. H. Org. Biomol. Chem. 2012, 10, 548.
[84] Aucagne, V.; Leigh, D. A. Org. Lett. 2006, 8, 4505.
[85] Fiandanese, V.; Bottalico, D.; Marchese, G.; Punzi, A.; Capuzzolo, F. Tetrahedron 2009, 65, 10573.
[86] Aizpurua, J. M.; Azcune, I.; Fratila, R. M.; Balentova, E.; Sagartzazu-Aizpurua, M.; Miranda, J. I. Org. Lett. 2010, 12, 1584.
[87] Doak, B. C.; Scanlon, M. J.; Simpson, J. S. Org. Lett. 2011, 13, 537.
[88] Kamal, A.; Shankaraiah, N.; Reddy. C. R.; Prabhakar, S.; Markandeya, N.; Srivastava, H. K.; Sastry, G. N. Tetrahedron 2010, 66, 5498.
[89] Kovalovs, A.; Novosjolova, I.; Bizdena, E.; Bizane, I.; Skardziute, L.; Kazlauskas, K.; Jursenas, S.; Turks, M. Tetrahedron Lett. 2013, 54, 850.
[90] Potewar, T. M.; Petrova, K. T.; Barros, M. T. Carbohydr. Res. 2013, 79, 60.
[91] Westermann, B.; Dörner, S.; Brauch, S.; Schaks, A.; Heinke, R.; Stark, S.; van Delft, F. L.; van Berkel, S. S. Carbohydr. Res. 2013, 371, 61.
[92] Yuan, Z; Kuang, G.-C.; Clark, R. J.; Zhu, L. Org. Lett. 2012, 14, 2590.
[93] Guiard, J.; Fiege, B.; Kitov, P. I.; Peters, T.; Bundle, D. R. Chem. Eur. J. 2011, 17, 7438.
[94] Wang, Z.-Y.; Tan Y.-H.; Huo, J.-P.; Qi, J.; Li, D. In 12th International Symposium for Chinese Organic Chemists, Eds.: Zhai, H.-B.; Wang, W., Lanzhou University, Lanzhou, 2012, pp. 27.
[95] Banday, A. H.; Arora, B. S.; Alam, M. S.; Kumar, H. M. S. Helv. Chim. Acta 2007, 90, 2368.
[96] Arora, B. S.; Shafi, S.; Singh, S.; Ismail, T.; Kumar, H. M. S. Carbohydr. Res. 2008, 343, 139.
[97] Yan, W.-M.; Wang, Q.-Y.; Chen, Y.-F.; Petersen, J. L.; Shi, X.-D. Org. Lett. 2010, 12, 3308.
/
| 〈 |
|
〉 |