

Chinese Journal of Organic Chemistry >
Recent Progress in Fluorescent Probes for Pyrophosphate Based on Small Organic Molecules
Received date: 2013-11-26
Revised date: 2013-12-12
Online published: 2013-12-23
Supported by
Project supported by the National Natural Science Foundation of China (No. 21272027), the Beijing Natural Science Foundation (No. 2122031), and the Beijing Municipal Commission of Education.
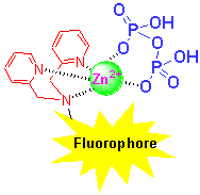
Pyrophosphate (PPi), a kind of important biological functional anion, plays crucial roles in life science, environmental science, medicine and chemical process. It has attracted extensive interests in supramolecular chemistry to design efficient fluorescent PPi sensors due to the simplicity and high sensitivity of fluorometric analysis. In this review, several kinds of strategies and theories of PPi fluorescent recognition and sensing are summarized, including off-on/on-off fluorescent response, excimer formation, hydrogen-bonding or electrostatic interactions and fluorescent indicator displacement assay. DPA-metal ion complexes, especially DPA-Zn2+ complex, have remarkable binding affinity and high selectivity for PPi. The chemosensing ensembles, consisting of DPA-Zn2+ complex and various fluorophores or fluorescent indicators, have been widely applied to the PPi fluorescent recognition and sensing.
Xu Qinchao , Jin Can , Zhu Xuehui , Xing Guowen . Recent Progress in Fluorescent Probes for Pyrophosphate Based on Small Organic Molecules[J]. Chinese Journal of Organic Chemistry, 2014 , 34(4) : 647 -661 . DOI: 10.6023/cjoc201311043
[1] Heinonen, J. K. Biological Role of Inorganic Pyrophosphate, Kluwer Academic Publishers, Norwell, 2001.
[2] Ronaghi, M.; Karamohamed, S.; Pettersson, B.; Uhlén, M.; Nyrén, P. Anal. Biochem. 1996, 242, 84.
[3] Xu, S.; He, M.; Yu, H.; Cai, X.; Tan, X.; Lu, B.; Shu, B. Anal. Biochem. 2001, 299, 188.
[4] (a) Kim, S. K.; Lee, D. H.; Hong, J.-I.; Yoon, J. Acc. Chem. Res. 2009, 42, 23.(b) Hargrove, A. E.; Nieto, S.; Zhang, T.; Sessler, J. L.; Anslyn, E. V. Chem. Rev., 2011, 111, 6603. (c) Ngo, H. T.; Liu, X.; Jolliffe, K. A. Chem. Soc. Rev. 2012, 41, 4928.
[5] Vance, D. H.; Czarnik, A. W. J. Am. Chem. Soc. 1994, 116, 9397.
[6] Lee, D. H.; Im, J. H.; Son, S. U.; Chung, Y. K.; Hong, J.-I. J. Am. Chem. Soc. 2003, 125, 7752.
[7] Jang, Y, J.; Jun, E. J.; Lee, Y. J.; Kim, Y. S.; Kim, J. S.; Yoon, J. J. Org. Chem. 2005, 70, 9603.
[8] Lee, H. N.; Swamy, K. M. K.; Kim, S. K.; Kwon, J.-Y.; Kim, Y.; Kim, S.-J.; Yoon, Y. J.; Yoon, J. Org. Lett. 2007, 9, 243.
[9] Chen, W.-H.; Xing, Y.; Pang, Y. Org. Lett. 2011, 13, 1362.
[10] Kim, M. J.; Swamy, M. K. K.; Lee, K. M.; Jagdale, A. R.; Kim, Y.; Kim, S.-J.; Yoo, K. H.; Yoon, J. Chem. Commun. 2009, 7215.
[11] Das, P.; Bhattacharya, S.; Mishra, S.; Das, A. Chem. Commun. 2011, 47, 8118.
[12] Kim, H. J.; Lee, J. H.; Hong, J.-I. Tetrahedron Lett. 2011, 52, 4944.
[13] Zhang, J. F.; Kim, S.; Han, J. H.; Lee, S.-J.; Pradhan, T.; Cao, Q. Y.; Lee, S. J.; Kang, C.; Kim, J. S. Org. Lett. 2011, 13, 5294.
[14] Jia, J.; Tang, X.; He, Y. F.; Zhang, M. Y.; Xing, G. W. Chin. J. Org. Chem. 2012, 32, 1803 (in Chinese). (郏佳, 唐茜, 何颖芳, 张梦雨, 邢国文, 有机化学, 2012, 32, 1803.)
[15] Su, G.; Liu, Z.; Xie, Z.; Qian, F.; He, W.; Guo, Z. Dalton Trans. 2009, 7888.
[16] Yang, S.; Feng, G.; Williams, N. H. Org. Biomol. Chem. 2012, 10, 5606.
[17] Gu, Z. Y, Xu, Q. C, Xing, G. W. Prog. Chem. 2013, 25, 1302 (in Chinese). (古振远, 徐勤超, 邢国文, 化学进展, 2013, 25, 1302.)
[18] Lee, D. H.; Kim, S. Y.; Hong, J.-I. Angew. Chem., Int. Ed. 2004, 43, 4777.
[19] Roy, B.; Rao, A. S.; Ahn, K. H. Org. Biomol. Chem. 2011, 9, 7774.
[20] Pathberiya, L. G.; Barlow, N.; Nguyen, T.; Graham, B.; Tuck, K. L. Tetrahedron 2012, 68, 9435.
[21] Lee, D.-N.; Jo, A.; Park, S. B.; Hong, J.-I. Tetrahedron Lett. 2012, 53, 5528.
[22] Huang, X.; Guo, Z.; Zhu, W.; Xie, Y.; Tian, H. Chem. Commun. 2008, 5143.
[23] Shao, N.; Wang, H.; Gao, X.; Yang, R.; Chan, W. Anal. Chem. 2010, 82, 4628.
[24] Xu, Q. C; Wang, X. F.; Xing, G. W.; Zhang, Y. RSC Adv. 2013, 3, 15834.
[25] Park, C.; Hong, J.-I. Tetrahedron Lett. 2010, 51, 1960.
[26] Ravikumar, I.; Ghosh, P. Inorg. Chem. 2011, 50, 4229.
[27] Cheng, T.; Wang, T.; Zhu, W.; Chen, X.; Yang, Y.; Xu, Y.; Qian, X. Org. Lett. 2011, 13, 3656.
[28] Cho, H. K.; Lee, D. H.; Hong, J.-I. Chem. Commun. 2005, 1690.
[29] Kim, S. Y.; Hong, J.-I. Bull. Korean Chem. Soc. 2010, 31, 716.
[30] Lee, H. N.; Xu, Z.; Kim, S. K.; Swamy, K. M. K.; Kim, Y.; Kim, S.-J.; Yoon, J. J. Am. Chem. Soc. 2007, 129, 3828.
[31] Wen, J.; Geng, Z.; Yin, Y.; Zhang, Z.; Wang, Z. Dalton Trans. 2011, 40, 1984.
[32] Han, M. S.; Kim, D. H. Angew. Chem., Int. Ed. 2002, 41, 3809.
[33] Han, M. S.; Kim, D. H. Bioorg. Med. Chem. Lett. 2003, 13, 1079.
[34] Hanshaw, R. G.; Hilkert, S. M.; Jiang, H.; Smith, B. D. Tetrahedron Lett. 2004, 45, 8721.
[35] Morgan, B. P.; He, S.; Smith, R. C. Inorg. Chem. 2007, 46, 9262.
[36] Lee, J. H.; Park, J.; Lah, M. S.; Chin, J.; Hong, J. Org. Lett. 2007, 9, 3729.
[37] Huang, F.; Cheng, C.; Feng, G. J. Org. Chem. 2012, 77, 11405.
[38] Surman, A. J.; Bonnet, C. S.; Lowe, M. P.; Kenny, G. D.; Bell, J. D.; Toth, E.; Vilar, R. Chem. Eur. J. 2011, 17, 223.
[39] Nonaka, A.; Horie, S.; Jamesb, T. D.; Kubo, Y. Org. Biomol. Chem. 2008, 6, 3621.
[40] McDonough, M. J.; Reynolds, A. J.; Lee, W. Y. G.; Jolliffe, K. A. Chem. Commun. 2006, 2971.
[41] Butler, S. J.; Jolliffe, K. A. Org. Biomol. Chem. 2011, 9, 3471.
[42] Carolan, J. V.; Butler, S. J.; Jolliffe, K. A. J. Org. Chem. 2009, 74, 2992.
[43] Lee, D. H.; Kim, S. Y.; Hong, J.-I. Tetrahedron Lett. 2007, 48 4477.
[44] Chen, Z.; Lu, Y.; He, Y.; Huang, X. Sens. Actuators, B: Chem. 2010, 149, 407.
[45] Sokkalingam, P.; Kim, D. S.; Hwang, H.; Sessler, J. L.; Lee, C.-H. Chem. Sci. 2012, 3, 1819.
[46] Kim, S. Y.; Hong, J.-I. Tetrahedron Lett. 2009, 50, 1951.
[47] Nishizawa, S.; Kato, Y.; Teramae, N. J. Am. Chem. Soc. 1999, 121, 9463.
[48] Kim, S. K.; Singh, N. J.; Kwon, J.; Hwang, I.-C.; Park, S. J.; Kim, K. S.; Yoon, J. Tetrahedron 2006, 62, 6065.
[49] Anzenbacher, P. Jr; Jursíková, K.; Sessler, J. L. J. Am. Chem. Soc. 2000, 122, 9350.
[50] Lee, J. H.; Jeong, A. R.; Jung, J.-H.; Park, C.-M.; Hong, J.-I. J. Org. Chem. 2011, 76, 417.
[51] (a) Zhu, C.; Liu, L.; Yang, Q.; Lv, F.; Wang, S. Chem. Rev. 2012, 112 4687.(b) Wang, Y.-J.; Gu, Z.-Y., Xing, G. W. Chem. Asian J. 2012, 7, 489.(c) Wang, Y.-J.; Xin, B.-J.; Duan, X.-R.; Xing, G.-W.; Wang, S. Macromol. Rapid Commun. 2010, 31, 1473. Zhao, X.; Schanze, K. S. Chem. Commun. 2010, 46, 6075.
/
| 〈 |
|
〉 |