

Chinese Journal of Organic Chemistry >
Synthesis and Biological Properties of 3-(2-Hydroxyethyl)-2-(phenylamino)quinazolin-4(3H)-ones
Received date: 2013-11-29
Revised date: 2013-12-19
Online published: 2014-01-03
Supported by
Project supported by the Natural Science Mutual Funds of Guizhou Province (No. LKK[2013]03).
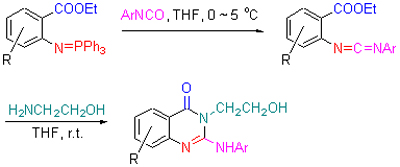
Key intermediate β-ethoxycarbonyl iminophosphorance (4), which was prepared from starting material substituted 2-aminobenzoic acid (1), was reacted with aryl isocyanates, ethanolamine by a three-component tandem aza-Wittig reaction to obtain a series of 2-arylamino-3-hydroxyethyl-4(3H)-quinazolinone derivatives 6. The structures of the target products were confirmed by IR, 1H NMR, MS and elemental analysis. The antibacterial activities of compounds 6 in vitro against tobacco bacterial wilt were further tested. The bioassay of the compounds 6 showed that the inhibitory activities of all the compounds were lower than the reference drug thiediazole copper. However, the antibacterial activity was significantly enhanced when the parent quinazolinone rings of the compounds 6 are substituted. Thus it can be seen that decoration of the parent quinazolinone ring of the compounds 6 can be regard as a kind of effective way of improving their biological property.
Yang Xuhong , Wang Xiang , Wu Minghu . Synthesis and Biological Properties of 3-(2-Hydroxyethyl)-2-(phenylamino)quinazolin-4(3H)-ones[J]. Chinese Journal of Organic Chemistry, 2014 , 34(5) : 1015 -1020 . DOI: 10.6023/cjoc201311046
[1] Kumar, V.; Kumar Tewary, D.; Desikachar Ravindranath, S.; Shanker, A. Food Chem. Toxicol. 2004, 42, 423.
[2] Berg, G. L. Farm Chemicals Handbook, Meister Publishing Company, Willoughby OH, 1986.
[3] Russell, P.; Percival, A.; Coltman, P.; Green, D. In Brighton Crop Protection Conference, Pests and Diseases, Vol. 1, Farnham, UK, 1992, p. 411.
[4] Huapaya, L. V. Am. J. Psychiatry 1979, 136, 985.
[5] Liang, Y.; Ding, M.-W.; Liu, Z.-J.; Liu, X.-P. Synth. Commun. 2003, 33, 2843.
[6] Ding, M. W.; Yang, S. J.; Chen, Y. F. Chin. J. Org. Chem. 2004, 24, 923 (Chinese).
(丁明武, 杨尚君, 陈云峰, 有机化学, 2004, 24, 923.)
[7] Yang, X.; Wu, M.; Ding, M. Chin. J. Org. Chem. 2010, 30, 1032 (in Chinese).
(杨绪红, 吴鸣虎, 丁明武, 有机化学, 2010, 30, 1032.)
[8] Gao, X. W.; Cai, X. J.; Yan, K.; Gao, L. L.; Wang, H. Y.; Chen, Z.; Song, B. A. Chin. J. Org. Chem. 2008, 28, 1785 (Chinese)
(高兴文, 蔡学建, 严凯, 高丽丽, 王和英, 陈卓, 宋宝安, 有机化学, 2008, 28, 1785.)
[9] Wang, X.; Li, P.; Li, Z.; Yin, J.; He, M.; Xue, W.; Chen, Z.; Song, B. J. Agric. Food Chem. 2013, 61, 9575.
[10] Yang, X. H.; Wu, M. H.; Sun, S. F.; Ding, M. W.; Xie, J. L.; Xia, Q. H. J. Heterocycl. Chem. 2008, 45, 1365.
[11] Vlasenko, A. F.; Mandrichenko, B. E.; Rogul'chenko, G. K.; Sinyak, R. S.; Mazur, I. A.; M., K. P. Khim. Geterotsikl. Soedin. 1976, 12, 834.
[12] Ding, M.-W.; Zeng, G.-P.; Wu, T.-J. Synth. Commun. 2000, 30, 1599.
/
| 〈 |
|
〉 |