

Chinese Journal of Organic Chemistry >
Synthetic and Structural Studies on Indenyl Phosphine Gold Complexes and Application as Catalyst for Hydration of Alkynes
Received date: 2014-01-23
Revised date: 2014-02-22
Online published: 2014-03-12
Supported by
Project supported by the National Natural Science Foundation of China (Nos. 21072071, 21272088).
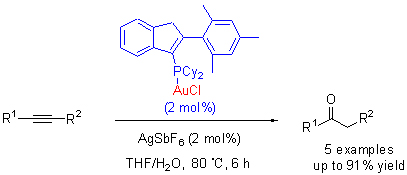
Four novel indenyl phosphine gold complexes were prepared by the reaction of indenyl phosphine with AuCl-(SMe)2. All products 1~4 have been fully characterized by elemental analysis, spectroscopy, and X-ray crystal diffraction techniques for 2 and 4. Complex 2 has been found efficiently to catalyze hydration of alkyne with the advantages of mild reaction conditions, less catalyst, excellent atomic economy and environmental friendly.
Key words: indenyl; phosphine; gold; alkyne; hydration reaction
Han Zhijun , Mao Shulan , Peng Hui , Pi Yunxiao , Chen You , Liu Shenghua , Yu Guangao . Synthetic and Structural Studies on Indenyl Phosphine Gold Complexes and Application as Catalyst for Hydration of Alkynes[J]. Chinese Journal of Organic Chemistry, 2014 , 34(5) : 893 -897 . DOI: 10.6023/cjoc201401037
[1] Otera, J. Modern Carbonyl Chemistry, Wiley-VCH, Weinheim, Germany, 2000.
[2] Kucherov, M. Chem. Ber. 1881, 14, 1540.
[3] Trost, B. M. Science 1991, 254, 1471.
[4] (a) Hiscox, W.; Jennings, P. W. Organometallics 1990, 9, 1997.
(b) Francisco, L. W.; Moreno, D. A.; Atwood, J. D. Organometallics 2001, 20, 4237.
[5] (a) Tokunaga, M.; Wakatsuki, Y. Angew. Chem., Int. Ed. 1998, 37, 2867.
(b) Suzuki, T.; Tokunaga, M.; Wakatsuki, Y. Org. Lett. 2001, 3, 735.
[6] (a) Janout, V.; Regen, S. L. J. Org. Chem. 1982, 47, 3331.
(b) Tachinami, T.; Nishimura, T.; Ushimaru, R.; Noyori, R.; Naka, H. J. Am. Chem. Soc. 2013, 135, 50.
[7] (a) Fukuda, Y.; Utimoto, K. J. Org. Chem. 1991, 56, 3729.
(b) Marion, N.; Ram?n, R. S.; Nolan, S. P. J. Am. Chem. Soc. 2009, 131, 448.
(c) Wang, D.; Cai, R.; Sharma, S.; Jirak, J.; Thummanapelli, S. K.; Akhmedov, N. G.; Zhang, H.; Liu, X.; Petersen, J. L.; Shi, X. J. Am. Chem. Soc. 2012, 134, 9012.
[8] (a) Nun, P.; Ramón, R. S.; Gaillard, S.; Nolan, S. P. J. Organomet. Chem. 2011, 696, 7.
(b) Leyva, A.; Corma, A. J. Org. Chem. 2009, 74, 2067.
[9] Li, Z.; Brouwer, C.; He, C. Chem. Rev. 2008, 108, 3239.
[10] Luzung, M. R.; Markham, J. P.; Toste, F. D. J. Am. Chem. Soc. 2004, 126, 10858.
[11] Mizushima, E.; Hayashi, T.; Tanaka, M. Org. Lett. 2003, 5, 3349.
[12] Alm?ssy, A.; Nagy, C. E.; Bényei, A. C.; Joó, F. Organometallics 2010, 29, 2484.
[13] (a) Wang, X.; Lai, Y.; Wu, H.; Zhang, J.; Li, Y. Chin. J. Org. Chem. 2009, 29, 432 (in Chinese).
(王翔, 赖媛嫒, 吴宏, 张建明, 李永建, 有机化学, 2009, 29, 432.)
(b) Zhang, Y; Zhu, C. Chin. J. Org. Chem. 2012, 32, 2283. (in Chinese).
(张艳, 朱成建, 有机化学, 2012, 32, 2283.)
[14] Zhang, R.; Xu, Q.; Shi, M. Acta Chim. Sinica 2012, 70, 1593 (in Chinese).
(张睿, 徐琴, 施敏, 化学学报, 2012, 70, 1593.)
[15] Chen, L.; Yu, G. A.; Li, F.; Zhu, X.; Zhang, B.; Guo, R.; Li, X.; Yang, Q.; Jin, S.; Li, C.; Liu, S. H. J. Organomet. Chem. 2010, 695, 1768.
[16] Hao, X.; Yuan, J.; Yu, G.-A.; Qiu, M.-Q.; She, N.-F.; Sun, Y.; Zhao, C.; Mao, S.-L.; Yin, J.; Liu, S.-H. J. Organomet. Chem. 2012, 706, 99.
[17] Mao, S.-L.; Sun, Y.; Yu, G.-A.; Zhao, C.; Han, Z.-J.; Yuan, J.; Zhu, X.; Yang, Q.; Liu, S. -H. Org. Biomol. Chem. 2012, 10, 9410.
[18] Yuan, J.; Sun, Y.; Yu, G.-A.; Zhao, C.; She, N.-F.; Mao, S.-L.; Huang, P.-S.; Han, Z.-J.; Yin, J.; Liu, S. H. Dalton Trans. 2012, 41, 10309.
[19] Hooper, T. N.; Butts, C. P.; Green, M.; Haddow, M. F.; McGrady, J.E.; Russell, C. A. Chem. Eur. J. 2009, 15, 12196.
[20] Goossen, L. J.; Manjolinho, F.; Khan, B. A.; Rodríguez, N. J. Org. Chem. 2009, 74, 2620.
[21] Chen, Z.-W.; Ye, D.-N.; Qian, Y.-P.; Ye, M.; Liu, L.-X. Tetrahedron 2013, 69, 6116.Gatti, N. Tetrahedron Lett. 1990, 31, 3933.
/
| 〈 |
|
〉 |