

Chinese Journal of Organic Chemistry >
Synthesis of Hexapeptide by Liquid-Phase Fragments Coupling Strategy
Received date: 2014-03-20
Revised date: 2014-05-12
Online published: 2014-06-09
Supported by
Project supported by the National Natural Science Foundation of China (Nos. 20872118, 30070905), the Key Laboratory Fund of Shaanxi Province of China (Nos. 2010JS097, 11JS090, 12JS110), the Foundation of the Education Department of Shaanxi Province (No. 08jk477) and the Technology Innovation Fund of Xi'an City (No. CX13120)
The hexapeptide (Ac-EEMQRR-NH2) is a non-toxic cosmeceutical which can mimic the function of botulinum neurotoxins. This paper reports a solid-phase synthesis of hexapeptide by employing “A+B” fragments coupling strategy while fragment A [Ac-Glu(OtBu)-Glu(OtBu)-Met-OH] and B [H-Gln(Trt)-Arg-Arg-OH] were prepared by classical solution methods. The final product (coupling yield>65%, purity>98%) was obtained by cleavage cocktail treatment and HPLC purification. The influencing factors of coupling condition were discussed. In conclusion, this strategy is a relatively quickly, efficient and high yield method for the synthesis of hexapeptide by combining the advantages of solid phase and liquid phase synthesis.
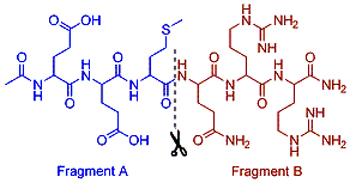
Key words: hexapeptide; peptide fragments; synthesis
Zhang Teng , Song Wei , Han Bin , Liu Lin , Feng Lingyun , Zhao Jinli , Liu Jianli . Synthesis of Hexapeptide by Liquid-Phase Fragments Coupling Strategy[J]. Chinese Journal of Organic Chemistry, 2014 , 34(10) : 2156 -2162 . DOI: 10.6023/cjoc201403043
[1] Merrifield, R. B. J. Am. Chem. Soc. 1936, 85, 2149.
[2] Bayer, E.; Mutter, M. Nature 1972, 237, 512.
[3] Ovchinnikov, M. V.; Bespalova, Z. D.; Molokoedov, A. S.; Revenko, I. V.; Sepetov, N. F.; Isakova, O. L.; Titov, M. I. Collect. Czech. Chem. Commun. 1989, 54, 772; 1989, 54, 784.
[4] (a) Gutiérrez, L. M.; Cànaves, J. M.; Ferrer-Montiel, A. V.; Reig, J. A.; Montal, M.; Viniegra, S. FEBS Lett. 1995, 372, 39.
(b) Ferrer-Montiel, A.V.; Gutiérrez, L. M.; Apland, J. P.; Canaves, J. M.; Gil, A.; Viniegra, S.; Biser, J.A.; Adler, M.; Montal, M. FEBS Lett. 1998, 435, 84.
(c) Blanes-Mira, C.; Merino, J. M.; Valera, E.; Fernández-Ballester, G.; Gutiérrez, L. M.; Viniegra, S.; Pérez-Payá, E.; Ferrer-Montiel, A. J. Neurochem. 2004, 88, 124.
[5] (a) Lungu, C.; Considine, E.; Zahir, S.; Ponsati, B.; Arrastia. S.; Hallett, M. Eur. J. Neurol. 2013, 20, 515.
(b) Fields, K.; Falla, T. J.; Rodan, K.; Bush, L. J. Cosmet., Dermatol. 2009, 8, 8.
(c) Lijuan, Z.; Falla, T. J. Clin. Dermatol. 2009, 27, 485.
[6] (a) Wang, Y.; Wang, M.; Xiao, X. S.; Huo, J.; Zhang, W. D. J. Cosmet. Laser Ther. 2013, 15, 237.
(b) Wang, Y.; Wang, M.; Xiao, S.; Pan, P.; Li, P.; Huo, J. Am. J. Clin. Dermatol. 2013, 14, 147.
[7] Blanes-Mira, C.; Clemente, J.; Jodas, G.; Gil, A.; Fernández-Ball- ester, G.; Ponsati, B.; Gutierrez, L.; Pérez-Payá, E.; Ferrer-Montiel, A. Int. J. Cosmet. Sci. 2002, 24, 303.
[8] (a) Jencks, W. P.; Carriuolo, J. J. Am. Chem. Soc. 1960, 82, 675.
(b) Jencks, W. P.; Gilchrist, M. J. Am. Chem. Soc. 1966, 88, 104.
[9] Zhu, L.-L.; Sheng, Z.-C.; Chen, Y.-W.; Shen, S.-B. J. Chem. Eng. Chin. Univ. 2013, 27, 791 (in Chinese).
(朱亮亮, 绳则翠, 陈英文, 沈树宝, 高校化学工程学报, 2013, 27, 791.)
[10] Mutter, M.; Maser, F. In Peptides 1982, Ed.: Blaha, K., Berlin, 1983, p. 729.
[11] Han, X.; Wang, D. X. Acta Pharm. Sin. 2007, 42, 111 (in Chinese).
(韩香, 王德心, 药学学报, 2007, 42, 111.)
/
| 〈 |
|
〉 |