

Chinese Journal of Organic Chemistry >
Simple Synthesis of Arylamines from Tetraynes via the Hexadehydro-Diels-Alder Reaction
Received date: 2014-09-20
Revised date: 2014-10-15
Online published: 2014-11-07
Supported by
Project supported by the National Natural Science Foundation of China (No. 21272005).
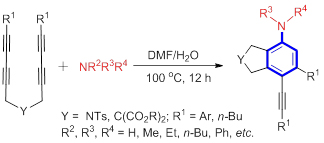
Arylamines are widespread in natural products, pharmaceuticals and molecular materials, and are useful synthetic building blocks. From a synthetic standpoint, the formation of Caryl—N bonds has been achieved by classical methods such as nucleophilic aromatic substitution, electrophilic nitration/reduction, or the transition-metal-mediated arylation of amides. This paper describes an efficient method for the synthesis of arylamines by the hexadehydro-Diels-Alder (HDDA) reaction of tetraynes under catalyst-free condition. This process is achieved in the absence of metal catalyst, base or oxidant, and it does not require the pre-installation of directing groups. This reaction exhibited an excellent regioselectivity, producing highly substituted fused yne-functionalized bicyclic compounds under mild conditions with good to excellent yields. All new products were fully characterized by various spectroscopic techniques and high-resolution mass spectrometry. The molecular structure of 3h was confirmed using single-crystal X-ray analyses.
Key words: arylamines; synthesis; Caryl—N coupling; HDDA reaction
Wu Yuqin , Yu Liangyun , Zhang Qi , Li Lidong . Simple Synthesis of Arylamines from Tetraynes via the Hexadehydro-Diels-Alder Reaction[J]. Chinese Journal of Organic Chemistry, 2015 , 35(3) : 724 -730 . DOI: 10.6023/cjoc201409032
[1] (a) Hartwig, J. F. Nature 2008, 455, 314. (b) Hartwig, J. F.; Stanley, L. M. Acc. Chem. Res. 2010, 43, 1461. (c) Chen, G. F., Ma, S. Angew. Chem., Int. Ed. 2010, 49, 8306.
[2] (a) Paul, F.; Patt, J.; Hartwig, J. F. J. Am. Chem. Soc. 1994, 116, 5969. (b) Louie, J.; Hartwig, J. F. Tetrahedron Lett. 1995, 36, 3609. (c) Stambuli, J. P.; Kuwano, P.; Hartwig, J. F. Angew. Chem. Int. Ed. 2002, 41, 4746.
[3] (a) Guram, A. S.; Buchwald, S. L. J. Am. Chem. Soc. 1994, 116, 7901. (b) Rennels, R. A.; Buchwald, S. L. Angew. Chem., Int. Ed. 1995, 34, 1348. (c) Wolf, J. P.; Buchwald, S. L. Angew. Chem., Int. Ed. 1999, 38, 2413.
[4] (a) Ma, D.; Zhang, Y.; Yao, J.; Wu, S.; Tao, F. J. Am. Chem. Soc. 1998, 120, 12459. (b) Ma, D.; Xia, D. Org. Lett. 2001, 3, 2583. (c) Ma, D.; Cai, Q.; Zhang, H. Org. Lett. 2003, 5, 2453. (d) Zhang, H.; Cai, Q.; Ma, D. J. Org. Chem. 2005, 70, 5164.
[5] Zhang, J.; Yin, H.; Han, S. Chin. J. Org. Chem. 2012, 32, 1429 (in Chinese). (张敬先, 殷慧清, 韩世清, 有机化学, 2012, 32, 1429.)
[6] (a) Wolfe, J. P.; Buchwald, S. L. J. Am. Chem. Soc. 1997, 119, 6054. (b) Ge, S.; Green, R. A.; Hartwig, J. F. J. Am. Chem. Soc. 2014, 136, 1617.
[7] Guo, D. L.; Huang, H.; Xu, J. Y. Org. Lett. 2008, 10, 4513.
[8] Louillat, M. L.; Biafora, A.; Legros, F.; Patureau, F. W. Angew. Chem., Int. Ed. 2014, 53, 3505.
[9] (a) Coeffard, V.; Moreau, X.; Thomassigny, C.; Greck, C. Angew. Chem., Int. Ed. 2013, 52, 5684. (b) Xiao, Q.; Tian, L.; Tan, R.; Xia, Y.; Qiu, D.; Zhang, Y.; Wang, J. Org. Lett. 2012, 14, 4230. (c) Mlynarski, S. N.; Karns, A. S.; Morken, J. P. J. Am. Chem. Soc. 2012, 134, 16449. (d) Zhu, C.; Li, G.; Ess, D. H.; Falck, J. R.; Kürti, L. J. Am. Chem. Soc. 2012, 134, 18253.
[10] (a) Hoye, T. R.; Baire, B.; Niu, D.; Willoughby, P. H.; Woods, B. P. Nature 2012, 490, 208. (b) Niu, D.; Hoye, T. R. Nat. Chem. 2013, 6, 34. (c) Baire, B.; Niu, D.; Willoughby, P. H.; Woods, B. P.; Hoye, T. R. Nat. Protoc. 2013, 8, 501. (d) Niu, D.; Willoughby, P. H.; Woods, B. P.; Baire, B.; Hoye, T. R. Nature 2013, 501, 531. (e) Niu, D.; Wang, T.; Woods, B. P.; Hoye, T. R. Org. Lett. 2014, 16, 254.
[11] (a) Tadross, P. M.; Stoltz, B. M. Chem. Rev. 2012, 112, 3550. (b) Gampe, C. M.; Carreira, E. M. Angew. Chem., Int. Ed. 2012, 51, 3766. (c) Hoffmann, R. W.; Suzuki, K. Angew. Chem., Int. Ed. 2013, 52, 2655. (d) Holden, C.; Greaney, M. F. Angew. Chem., Int. Ed. 2014, 53, 5746.
[12] (a) Yun, S. Y.; Wang, K.-P.; Lee, N.-K.; Mamidipalli, P.; Lee, D. J. Am. Chem. Soc. 2013, 135, 4668. (b) Wang, K.-P.; Yun, S. Y.; Mamidipalli, P.; Lee, D. Chem. Sci. 2013, 4, 3205. (c) Karmakar, R.; Mamidipalli, P.; Yun, S. Y.; Lee, D. Org. Lett. 2013, 15, 1938. (d) Karmakar, R.; Yun, S. Y.; Wang, K.-P.; Lee, D. Org. Lett. 2014, 16, 6. (e) Mamidipalli, P.; Yun, S. Y.; Wang, K.-P.; Zhou, T.; Xia, Y.; Lee, D. Chem. Sci. 2014, 5, 2362.
[13] (a) Hu, Y. M.; Yu, C. L.; Ren, D.; Hu, Q.; Zhang, L. D.; Cheng, D. Angew. Chem., Int. Ed. 2009, 48, 5448. (b) Hu, Y. M.; Ouyang, Y.; Qu, Y.; Hu, Q.; Yao, H. Chem. Commun. 2009, 4575. (c) Hu, Y.; Yao, H.; Sun, Y.; Wan, J.; Lin, X.; Zhu, T. Chem.-Eur. J. 2010, 16, 7635. (d) Zhao, Q. S.; Hu, Q.; Wen, L.; Wu, M.; Hu, Y. M. Adv. Synth. Catal. 2012, 354, 2113. (e) Li, Li.; Hu, Q.; Zhou, P.; Xie, H.; Zhang, X.; Hu, Y. Org. Biomol. Chem. 2014, 12, 5356.
[14] Zhang, H.; Hu, Q.; Li, L.; Hu, Y.; Zhou, P.; Zhang, X.; Xie, H.; Yin, F.; Hu, Y.; Wang, S. Chem. Commun. 2014, 50, 3335.
[15] Rideout, D. C.; Breslow, R. J. Am. Chem. Soc. 1980, 102, 7816.
[16] (a) Breslow, R.; Rizzo, C. J. J. Am. Chem. Soc. 1991, 113, 4340. (b) Hake, R.; McLendon, G.; Corin, A.; Holzschu, D. J. Am. Chem. Soc. 1992, 114, 5442.
/
| 〈 |
|
〉 |