

Chinese Journal of Organic Chemistry >
Design, Synthesis and Cytotoxic Activity of Novel Hybrid Compounds between Aza-brazilin and Imidazolium
Received date: 2014-12-31
Revised date: 2015-01-26
Online published: 2015-02-05
Supported by
Project supported by the Program for Changjiang Scholars and Innovative Research Team in University (No. IRT13095), the National Natural Science Foundation of China (Nos. 21462049, 21332007, U1402227), and the Natural Science Foundation of Yunnan Province (Nos. 2013FA028, 2012FB113, 2010GA014).
A series of novel hybrid compounds between aza-brazilin and imidazole have been prepared from 3-(3,4-dimeth- oxyphenyl)propanoic acid. Their structures were confirmed by 1H NMR, 13C NMR, HR-ESI-MS and X-ray crystallographic analysis. These compounds have been evaluated in vitro against a panel of human tumor cell lines. 2-Methyl-3-(naphthalen- 2-ylmethyl)-1-(2-oxo-2-(4,9,10-trimethoxy-6,6a,7,11b-tetrahydro-5H-indeno[2,1-c]quinolin-5-yl)ethyl)-1H-benzo[d]imidazol- 3-ium bromide (26) was found to be the most potent derivative against four strains human tumor lines and more active than cisplatin, and exhibited the most potent cytotoxic activities selectively against HL-60, MCF-7 and SW-480.
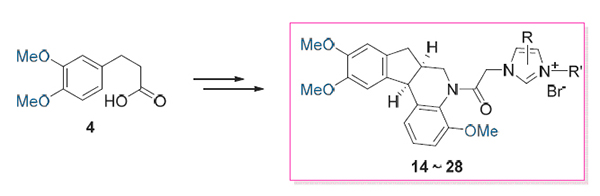
Key words: aza-brazilin; imidazole; hybrid compound; cytotoxic activity
Wang Xuequan , Li Yan , Yang Xiaodong , Zhang Hongbin . Design, Synthesis and Cytotoxic Activity of Novel Hybrid Compounds between Aza-brazilin and Imidazolium[J]. Chinese Journal of Organic Chemistry, 2015 , 35(6) : 1276 -1285 . DOI: 10.6023/cjoc201412055
[1] Pan, C. X.; Guan, Y. F.; Zhang, H. B. Chin. J. Org. Chem. 2012, 32, 1116 (in Chinese). (潘成学, 关一富, 张洪彬, 有机化学, 2012, 32, 1116.)
[2] Robinson, R. Bull. Soc. Chim. Fr. 1958, 125.
[3] Kim, B. H.; Kim, S. J.; Jeong, E. J.; Sohn, J. H.; Jung, M.; Lee, H.; Kim, S. H. J. Agric. Food Chem. 2012, 60, 9882.
[4] (a) Kim, S. G.; Kim, Y. M.; Khil, L. Y.; Jeon, S. D.; So, D. S.; Moon, C. H.; Moon, C. K. Arch. Pharm. Res. 1998, 21, 140. (b) Hwang, G. S.; Kim, J. Y.; Chang, T. S.; Jeon, S. D.; So, D. S.; Moon, C. K. Arch. Pharm. Res. 1998, 21, 774.
[5] Hikino, H.; Taguchi, T.; Fujimura, H.; Hiramatsu, Y. Planta Med. 1977, 31, 214.
[6] Xu, H. X.; Lee, S. F. Phytother. Res. 2004, 18, 647.
[7] Pan, C. X.; Zeng, X. H.; Guan, Y. F.; Jiang, X. L.; Li, L.; Zhang, H. B. Synlett 2011, 425.
[8] (a) Vlahakis, J. Z.; Lazar, C.; Crandall, I. E.; Szarek, W. A. Bioorg. Med. Chem. 2010, 18, 6184. (b) Dominianni, S. J.; Yen, T. T. J. Med. Chem. 1989, 32, 2301. (c) Pardin, C.; Schmitzer, A. R.; Leclercq, L. Chem. Eur. J. 2010, 16, 4686.
[9] (a) Fortuna, C. G.; Barresi, V.; Berellini, G..; Musumarra, G. Bioorg. Med. Chem. 2008, 16, 4150. (b) Saberi, M. R.; Vinh, T. K.; Yee, S. W.; Griffiths, B. J. N.; Evan, P. J.; Simsons, C. J. Med. Chem. 2006, 49, 1016.
[10] (a) Jnr, C. V.; Danuello, A.; Bolzani, V. S.; Barreiro, E. J.; Fraga, C. A. M. Curr. Med. Chem. 2007, 14, 1829. (b) Walsh, J. J.; Bell, A. Curr. Pharm. Des. 2009, 15, 2970. (c) Zhu, P. F.; Zhao, J. F.; Yang, X. D.; Zhang, H. B. Chin. J. Org. Chem. 2014, 34, 1167 (in Chinese). (朱培芳, 赵静峰, 羊晓东, 张洪彬, 有机化学, 2014, 34, 1167.)
[11] CCDC 1041451 contains the supplementary crystallographic data for compound 22.These data can be obtained free of charge from The Cambridge Crystallographic Data Center via www.ccdc.cam. ac.uk/data_request/cif.
/
| 〈 |
|
〉 |