

Chinese Journal of Organic Chemistry >
Design and Synthesis of 5-Aminopyrazole and 5-Aminotriazole Derivatives as Fibroblast Growth Factor Receptor Inhibitors
Received date: 2015-03-27
Revised date: 2015-04-30
Online published: 2015-05-15
Supported by
Project supported by the Specialized Research Fund for the Doctoral Program of Higher Education of China (No. 20130071110071) and the Shanghai Municipal Science & Technology Pillar Program for Bio-pharmaceuticals (No. 13431900102).
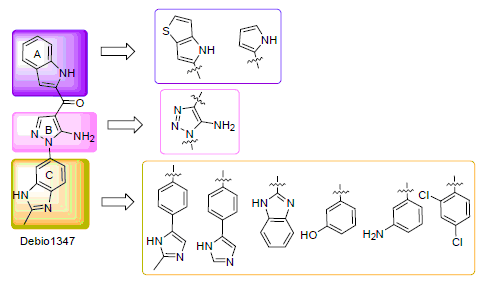
Fibroblast growth factor receptor (FGFR) is a promising therapeutic target for cancer treatment. A series of 5-aminopyrazole and 5-amino-1,2,3-triazole derivatives were designed as FGFR inhibitors based on the docking mode of Debio1347 within the FGFR kinase domain. The inhibitory effects on FGFR2 kinase and the antitumor activities against human gastric cancer SNU16 cell lines were evaluated. Furthermore, the preliminary structure-activity relationships (SAR) was discussed. The results demonstrated that several compounds present good potency against FGFR2 kinase. 1-(5-Amino-1-(2-methyl-1H-benzo[d]imidazol-6-yl)-1H-pyrazol-4-yl)-1-(1H-thieno[3,2-b]pyrrol-2-yl)methanone (8) and 1-(5-amino-1-(2-methyl-1H-benzo[d]imidazol-6-yl)-1H-1,2,3-triazol-4-yl)-1-(1H-indol-2-yl)methanone (18) were the most potent with IC50 values of 3.3 and 2.3 nmol·L-1, respectively, which are similar to that of Debio1347. Compounds 8 and 18 exhibited slightly weaker antitumor activity against SNU16 cell lines than Debio1347 with IC50 value of 77.3, 155.2 nmol·L-1, respectively.
Key words: 5-aminopyrazole; 5-amino-1,2,3-triazole; FGFR inhibitors; anti-tumor agent
Dong Qian , Peng Xia , Wang Wen , Dai Yang , Zhao Weili , Ai Jing , Dong Xiaochun . Design and Synthesis of 5-Aminopyrazole and 5-Aminotriazole Derivatives as Fibroblast Growth Factor Receptor Inhibitors[J]. Chinese Journal of Organic Chemistry, 2015 , 35(9) : 1939 -1947 . DOI: 10.6023/cjoc201503045
[1] Eswarakumar, V. P.; Lax, I.; Schlessinger, J. Cytokine Growth F. R. 2005, 16, 139.
[2] Turner, N.; Grose, R. Nat. Rev. Cancer 2010, 10, 116.
[3] Brooks, A. N.; Kilgour, E.; Smith, P. D. Clin. Cancer Res. 2012, 18, 1855.
[4] Greulich, H.; Pollock, P. M. Trends Mol. Med. 2011, 17, 283.
[5] Wesche, J.; Haglund, K.; Haugsten, E. M. Biochem. J. 2011, 437, 199.
[6] Courjal, F.; Cuny, M.; SimonyLafontaine, J.; Louason, G.; Speiser, P.; Zeillinger, R.; Rodriguez, C.; Theillet, C. Cancer Res. 1997, 57, 4360.
[7] Kunii, K.; Davis, L.; Gorenstein, J.; Hatch, H.; Yashiro, M.; Di Bacco, A.; Elbi, C.; Lutterbach, B. Cancer Res. 2008, 68, 2340.
[8] Jung, E.-J.; Jung, E.-J.; Min, S. Y.; Kim, M. A.; Kim, W. H. Hum. Pathol. 2012, 43, 1559.
[9] Cappellen, D.; De Oliveira, C.; Ricol, D.; de Medina, S. G. D.; Bourdin, J.; Sastre-Garau, X.; Chopin, D.; Thiery, J. P.; Radvanyi, F. Nat. Genet. 1999, 23, 18.
[10] Dutt, A.; Salvesen, H. B.; Chent, T.-H.; Ramos, A. H.; Onofrio, R. C.; Hatton, C.; Nicoletti, R.; Winckler, W.; Grewal, R.; Hanna, M.; Wyhs, N.; Ziaugra, L.; Richter, D. J.; Trovik, J.; Engelsen, I. B.; Stefansson, I. M.; Fennell, T.; Cibulskis, K.; Zody, M. C.; Akslen, L. A.; Gabriel, S.; Wong, K.-K.; Sellers, W. R.; Meyerson, M.; Greulich, H. Proc. Natl. Acad. Sci. U. S. A. 2008, 105, 8713.
[11] Chesi, M.; Nardini, E.; Brents, L. A.; Schrock, E.; Ried, T.; Kuehl, W. M.; Bergsagel, P. L. Nat. Genet. 1997, 16, 260.
[12] Liang, G.; Chen, G.-Z.; Wei, X.-Y.; Zhao, Y.-J.; Li, X.-K. Cytokine Growth Factor Rev. 2013, 24, 467.
[13] Ho, H. K.; Yeo, A. H. L.; Kang, T. S.; Chua, B. T. Drug Discovery Today 2014, 19, 51.
[14] Gavine, P. R.; Mooney, L.; Kilgour, E.; Thomas, A. P.; Al-Kadhimi, K.; Beck, S.; Rooney, C.; Coleman, T.; Baker, D.; Mellor, M. J.; Brooks, A. N.; Klinowska, T. Cancer Res. 2012, 72, 2045.
[15] Guagnano, V.; Furet, P.; Spanka, C.; Bordas, V.; Le Douget, M.; Stamm, C.; Brueggen, J.; Jensen, M. R.; Schnell, C.; Schmid, H.; Wartmann, M.; Berghausen, J.; Drueckes, P.; Zimmerlin, A.; Bussiere, D.; Murray, J.; Porta, D. G. J. Med. Chem. 2011, 54, 7066.
[16] Zhao, G.-S.; Li, W.-Y.; Chen, D.-H.; Henry, J. R.; Li, H.-Y.; Chen, Z.-G.; Zia-Ebrahimi, M.; Bloem, L.; Zhai, Y.; Huss, K.; Peng, S.-B.; McCann, D. J. Mol. Cancer Ther. 2011, 10, 2200.
[17] Nakanishi, Y.; Akiyama, N.; Tsukaguchi, T.; Fujii, T.; Sakata, K.; Sase, H.; Isobe, T.; Morikami, K.; Shindoh, H.; Mio, T.; Ebiike, H.; Taka, N.; Aoki, Y.; Ishii, N. Mol. Cancer Ther. 2014, 13, 2547.
[18] Taka, N.; Ohmori, M.; Takami, K.; Matsushita, M.; Hayase, T.; Hyodo, I.; Kochi, M.; Nishii, H.; Ebiike, H.; Nakanishi, Y.; Mio, T.; Wang, L.; Zhao, W.-L. WO 2011016528, 2011 [Chem. Abstr. 2011, 154, 234696].
[19] Kislyi, V. P.; Danilova, E. B.; Semenov, V. V. Russ. Chem. B 2003, 52, 1770.
[20] Wu, Y.-Y.; Cheng, C.; Jiao, L.-J.; Yu, C.-J.; Wang, S.-F.; Wei, Y.; Mu, X.-L.; Hao, E.-H. Org. Lett. 2014, 16, 748.
[21] Slatt, J.; Romero, I.; Bergman, J. Synthesis-Stuttgart. 2004, 16, 2760.
[22] Hassaneen, H. M. E. Synth. Commun. 2007, 37, 3579.
[23] Novelli, A.; De Santis, A. Tetrahedron Lett. 1967, 3, 265.
[24] Walter, R.; Anderskewitz, R.; Kley, J.; Dollinger, H.; Goeggel, R.; Jung, B.; Mack, J.; Nickolaus, P. WO 2006114371, 2006 [Chem. Abstr. 2006, 145, 471367].
[25] Ishida, T.; In, Y.; Inoue, M.; Kurihara, T.; Moriomoto, K.; Morisaka, K.; Shibata, K. Chem. Pharm. Bull. 1990, 38, 1803.
[26] de Candia, M.; Liantonio, F.; Carotti, A.; De Cristofaro, R.; Altomare, C. J. Med. Chem. 2009, 52, 1018.
[27] Morton, A. A.; Slaunwhite, W. R. J. Biol. Chem. 1949, 179, 259.
[28] Keglevic, D. Croat. Chem. Acta 1961, 33, 83.
[29] Mokrushina, G. A.; Kotovskaya, S. K.; Tyurenkova, G. N.; Ilyenko, V. I.; Platonov, V. G.; Kiseleva, I. V. Khim.-Farm. Zh. 1988, 22, 195.
[30] Neugebauer, W.; Tomanek, M. US 3061435, 1960 [Chem. Abstr. 1960, 54, 73191].
/
| 〈 |
|
〉 |