

Chinese Journal of Organic Chemistry >
Structure Transformations on the Groups Linked with C-Pyrrol Subring of Pheophorbide-a and Synthesis of Chlorin Derivatives
Received date: 2015-03-31
Revised date: 2015-05-15
Online published: 2015-05-26
Supported by
Project supported by the National Natural Science Foundation of China (No. 21272048).
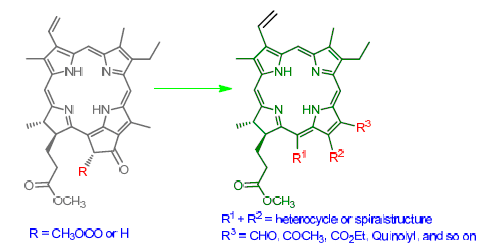
Methyl pheophorbide-a ester was used as starting material, and the chemical reactions, including allomerization, aldol condensation with formaldehyde, nucleophilic addition on the cycloketone, Tiffeneau-demjanov rearrangement with diazomethane, were carried out making use of active reaction regions linked with C-pyrrol subring to establish different carbonyl structures on the exocyclic ring and 12-position. From these new-formed active groups, the novel structures such as nitrogenous heterocycle and spirocycle were formed by condensations with o-phenylenediamine or o-aminobenzaldehyde, 1,3-dipolar cycloaddition with formaldehyde and sarcosine, and other structure transformations. A series of unreported chlorins related to chlorophyll were synthesized and their chemical structures were characterized by elemental analysis, UV, IR and 1H NMR spectra.
Key words: chlorophyll-a; chlorin; C-pyrrol subring; chemical modification; synthesis
Zhang Zhu , Jiang Qiyong , Zhang Qian , Wu Jin , Wang Jinjun . Structure Transformations on the Groups Linked with C-Pyrrol Subring of Pheophorbide-a and Synthesis of Chlorin Derivatives[J]. Chinese Journal of Organic Chemistry, 2015 , 35(9) : 1929 -1938 . DOI: 10.6023/cjoc201503050
[1] (a) Chen, Y. H.; Li, G. L.; Pandey, R. K. Curr. Org. Chem. 2004, 8, 1105.(b) Wang, J.-J. Chin. J. Org. Chem. 2005, 25, 1353 (in Chinese).(王进军, 有机化学, 2005, 25, 1353.)
[2] Pavlov, V. Y.; Ponomarev, G. V. Chem. Heterocycl. Compd. 2004, 40, 393.
[3] (a) Liu, C.; Dobhal, M. P.; Ethirajan, M.; Missert, J. R.; Pandey, R. K.; Balasubramanian, S.; Sukumaran, D. K.; Zhang, M.; Kadish, K. M.; Ohkubo, K.; Fukuzumi, S. J. Am. Chem. Soc. 2008, 130, 14311.(b) Machida, S.; Isoda, Y.; Kunieda, M.; Tamiaki, H. Tetrahedron Lett. 2012, 53, 6277.(c) Fajer, J. Photosynth. Res. 2004, 80, 165.(d) Bruckner, C.; McCarthy, J. R.; Daniell, H. W.; Pendon, Z. D.; Iiagan, R. P.; Francis, T. M.; Ren, L.; Birge, R. R.; Frank, H. A. Chem. Phys. 2003, 294, 285.
[4] (a) Gil, M.; Bieniaszl, M.; Seshadri, M.; Fisher, D.; Ciesielski, M. J.; Chen, Y.; Pandey, R. K.; Kozbor, D. Brit. J. Cancer 2011, 103(10), 1.(b) Ethirajan, M.; Joshi, P.; William, W. H.; Ohkubo, K.; Fukuzumi, S.; Pandey, R. K. Org. Lett. 2011, 8, 1956 (c) Bellnier, D. A.; Greco, W. R.; Loewen, G. M.; Nava, H.; Oseroff, A. R.; Pandey, R. K.; Tsuchida, T.; Dougherty, T. J. Cancer Res. 2003, 63, 1806.(d) Wang, J.-J.; Li, J.-Z.; Jakus. J.; Shim, Y. K. J. Porphyrins Phthalocyanines 2012, 16, 122
[5] Ethirajan, M.; Joshi, P.; William, W. H.; Ohkubo, K.; Fukuzumi, S.; Pandey, R. K. Org. Lett. 2011, 13, 1956.
[6] (a) Li, J.-Z.; Zhang, P.; Zhang, S.-G.; Yin, J.-G.; Wang, J.-J. Org. Biochem. 2015, 13, 1992.(b) Li, J.-Z.; Zhang, P.; Yao, N.-N.; Zhao, L.-L.; Wang, J.-J.; Shim, Y.-K. Tetrahedron Lett. 2014, 55, 1086.(c) Yang, X.-Y.; Zhang, S.-G.; Zhang, P.; Zhang, Q.; Wang, Z.; Jin, Y.-X.; Qi, C.-X.; Wang, J.-J. Chin. J. Org. Chem. 2015, 35, 181 (in Chinese).(杨晓英, 张善国, 张朋, 张千, 王振, 金英学, 祁彩霞, 王进军, 有机化学, 2015, 35, 181.)
[7] (a) Xu, X.-S.; Yao, N-N.; Liu, Y.; Yin, J.-G.; Qi, C.-X.; Wang, J.-J. Chin. J. Org. Chem. 2014, 34, 938 (in Chinese).(徐希森, 姚楠楠, 刘洋, 殷军港, 祁彩霞, 王进军, 有机化学, 2014, 34, 938.)(b) Liu, Y.; Xu, X.-S.; Li, J.-Z.; Yin, J.-G.; Qi, C.-X.; Wang, J.-J. Chin. J. Org. Chem. 2014, 34, 552 (in Chinese).(刘洋, 徐希森, 李家柱, 殷军港, 祁彩霞, 王进军, 有机化学, 2014, 34, 552.)(c) Wang, J.-J.; Li, J.-Z.; Li, Y.-W.; Jakus, J.; Shim, Y.-K. J. Porphyrins Phthalocyanines 2010, 14, 859.
[8] (a) Wang, J.-J.; Wang, P.; Li, J.-Z.; Jakus, J.; Shin, Y.-K. Bull. Korean Chem. Soc. 2011, 32, 3473. (b) Wang, J.-J.; Yin, Y.-F.; Yang, Z. J. Iran Chem. Soc. 2013, 10, 583.(c) Wang, J-J.; Ji, J.-Y.; Han, G.F.; Wu, X.-R.; Wang, L.-M.; Shin, Y.-K. Acta Chim. Sinica 2004, 62, 302 (in Chinese).(王进军, 纪建业, 韩光范, 邬旭然, 王鲁敏, 沈荣基, 化学学报, 2004, 62, 302.)
[9] Zhang, Q. M.S. Thesis, Yantai University, Yantai, 2011 (in Chinese). (张千, 硕士论文, 烟台大学, 烟台, 2011.)
[10] (a) Ma, L.; Dolphin, D. J. Org. Chem. 1996, 61, 2501.(b) Ji, J.-Y.; Xia, S.-W.; Liu, Y.; Yin, J.-G.; Qi, C.-X.; Wang, J.-J. Chin. J. Org. Chem. 2014, 34, 1138 (in Chinese).(纪建业, 姚楠楠, 刘洋, 殷军港, 祁彩霞, 王进军, 有机化学, 2014, 34, 1138.)(c) Smith, K. M.; Bissert, G. M. F.; Bushell, M. J. J. Org. Chem. 1980, 45, 2218.(d) Zhang, P. M.S. Thesis, Yantai University, Yantai, 2010 (in Chinese). (张朋, 硕士论文, 烟台大学, 烟台, 2010.)
[11] Liu, Y. M.S. Thesis, Yantai University, Yantai, 2014 (in Chinese). (刘洋, 硕士论文, 烟台大学, 烟台, 2014.)
[12] Smith, K. M.; Gogg, D. A.; Simpson, D. J. J. Am. Chem. Soc. 1985, 107, 4946.
/
| 〈 |
|
〉 |