

Chinese Journal of Organic Chemistry >
A General and Efficient Method for the Formylation of Amines Catalyzed by Bromodimethylsulfonium Bromide
Received date: 2015-04-12
Revised date: 2015-06-18
Online published: 2015-07-02
Supported by
Project supported by the Natural Science Foundation of Tianjin City (No. 12JCZDJC34300), the College Students' Entrepreneurship Training Program (No. 201447), the Doctoral Fund of Tianjin Normal University (No. 52XB1111).
A fast, efficient and simple route for the N-formylation of amines has been developed by treating amines with 85% formic acid at room temperature in the presence of 5 mol% of bromodimethylsulfonium bromide as catalyst under solvent-free condition. This method provides a green and much improved protocol over the existing methods.
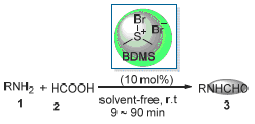
Key words: Amine; N-formylation; bromodimethylsulfonium bromide (BDMS); solvent-free
Huang Haijing , Liu Juyan , Ma Enzhong , Cao Yingyu . A General and Efficient Method for the Formylation of Amines Catalyzed by Bromodimethylsulfonium Bromide[J]. Chinese Journal of Organic Chemistry, 2015 , 35(11) : 2372 -2376 . DOI: 10.6023/cjoc201504015
[1] Sheehan, J. C.; Yang, D. D. H. J. Am. Chem. Soc. 1958, 80, 1154.
[2] (a) Chen, B. C.; Bednarz, M. S.; Zhao, R.; Sundeen, J. E.; Chen, P.; Shen, Z.; Skoumbourdis, A. P.; Barrish, A. P. Tetrahedron Lett. 2000, 41, 5453.(b) Jackson, A.; Meth-Cohn, O. J. Chem. Soc., Chem. Commun. 1995, 1, 1319.
[3] (a) Waki, J.; Meienhofer, J. J. Org. Chem.1977, 42, 2019.(b) Ugi, I. Angew. Chem., Int. Ed. Engl.1982, 21, 810.
[4] Schollkopf, U. Angew. Chem., Int. Ed. Engl. 1977, 16, 339.
[5] Han, Y.; Cai, L. Tetrahedron Lett. 1997, 38, 5423.
[6] (a) Iseki, K.; Mizuno, S.; Kuroki, Y.; Kobayashi, Y. Tetrahedron 1999, 55, 977;(b) Kobayashi, S.; Nishio, K. J. Org. Chem. 1994, 59, 6620.
[7] (a) Martinez, J.; Laur, J. Synthesis1982, 979.(b) Floresheimer, A.; Kula, M. R. Monatsh. Chem.1988, 119, 1323.
[8] Fieser, L. F.; Jones, J. E. Org. Synth. Coll. 1955, III, 590.
[9] Blicke, F. F.; Lu, C.-J. J. Am. Chem. Soc. 1952, 74, 3933.
[10] Chen, F. M. F.; Benoiton, N. L. Synthesis 1979, 709.
[11] (a) Yale, H. L. J. Org. Chem. 1971, 36, 3238.(b) Kisfaludy, L.; Laszlo, O. Synthesis 1987, 510.(c) Neveux, M.; Bruneau, C.; Dixneuf, P. H. J. Chem. Soc., Perkin Trans. 1 1991, 1197.(d) Duczek, W.; Deutsch, J.; Vieth, S.; Niclas, H.-J. Synthesis 1996, 37.
[12] Schmidhammer, H.; Brossi, A. Can. J. Chem.1982, 60, 3055.
[13] Reddy, P. G.; Kumar, G. D. K.; Baskaran, S. Tetrahedron Lett.2000, 41, 9149.
[14] Strazzolini, P.; Giumanini, A. G.; Cauci, S. Tetrahedron1990, 46, 1081.
[15] Kim, J.-G.; Jang, D. O. Bull. Korean Chem. Soc. 2010, 31, 2989.
[16] Hosseni-Sarvari, M.; Sharghi, H. J. Org. Chem.2006, 71, 6652.
[17] Akbari, J.; Hekmati, M.; Sheykhan, M.; Heydari, A. ARKIVOC2009, xi, 123.
[18] Desai, B.; Danks, T. N.; Wagner, G. Tetrahedron Lett. 2005, 46, 955.
[19] Jung, S. H.; Ahn, J. H.; Park, S. K.; Choi, J. K. Bull. Korean Chem. Soc.2002, 23, 149.
[20] Das, B.; Krishnaiah, M.; Balasubramanyam, P.; Veeranjaneyulu, B.; Nandakumar, D. Tetrahedron Lett. 2008, 49, 2225.
[21] Chandra Shekhar, A.; Ravi Kumar, A.; Sathaiah, G.; Luke Paul, V.; Sridhar, M.; Shanthan Rao, P. Tetrahedron Lett.2009, 50, 7099.
[22] Rahman, M.; Kundu, D.; Hajra, A.; Majee, A. Tetrahedron Lett. 2010, 51, 2896.
[23] (a) Zhao, C. Y.; Liu, J. Y.; Wang, Y.; Zhao, X. J.; Yuan, B.; Yue, M. M. Synth. Commun. 2014, 44, 827.(b) Yue, M. M.; Liu, J. Y.; Wang, Y.; Yuan, B. Chin. J. Org. Chem. 2014, 34, 190.(c) Zhang, L. J.; Liu, J. Y.; Wang, Y.; Chin. J. Org. Chem. 2013, 33, 339.
[24] Ansari, M. I.; Hussain, M. K.; Yadav, N.; Gupta, P. K.; Hajela, K. Tetrahedron Lett. 2012, 53, 2063.
[25] Thakuria, H.; Borah, B. M.; Das, G. J. Mol. Catal. A: Chem. 2007, 274, 1.
[26] Hosseini-Sarvaia, M.; Sharghi, H. J. Org. Chem. 2006, 71, 6652.
[27] (a) Choudhury, L. H.; Parvin, T.; Khan, A. T. Tetrahedron 2009, 65, 9513.(b) Sajadia, S. M.; Mahamb, M.; Rezaeic, A. Lett. Org. Chem. 2014, 11, 49.
/
| 〈 |
|
〉 |