

Chinese Journal of Organic Chemistry >
Recent Development of Indole Synthesis by Transition Metal Catalyzed C—H Functionalization
Received date: 2015-05-19
Revised date: 2015-06-29
Online published: 2015-07-10
Supported by
Project supported by the National Natural Science Foundation of China (No. 21402027), the Natural Science Foundation of Fujian Province (No. 2015J05046) and the Education Department of Fujian Province (No. JA14108).
Indoles represent important scaffolds in a large family of bioactive molecules and natural products. These compounds display a wide range of unique properties, which have been widely applied to clinical pharmaceuticals, materials and industries. In recent years, transition metal catalyzed C—H bond functionalization for the efficient construction of indole motifs has attracted much attention from chemical society. This review presents the recent development in this area and highlights the different advantages associated with transition metal catalyzed C—H bond functionalizations. The applications of the methodology on the synthesis of natural products and bioactive molecules containing indole skeletons have also been introduced.
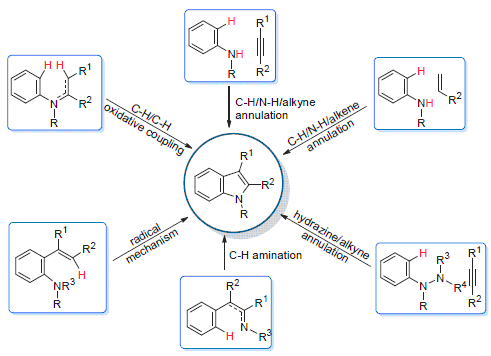
Key words: indole; transition metal catalysis; C—H functionalization
Lu Beili , Li Xianyan , Lin Yongmei . Recent Development of Indole Synthesis by Transition Metal Catalyzed C—H Functionalization[J]. Chinese Journal of Organic Chemistry, 2015 , 35(11) : 2275 -2290 . DOI: 10.6023/cjoc201505031
[1] Kochanowska-Karamyan, A. J.; Hamann, M. T. Chem. Rev. 2010, 110, 4489.
[2] Dan, D.; Jia, Y.-X. Chin. J. Org. Chem. 2013, 33, 1144 (in Chinese).(单冬, 贾彦兴, 有机化学, 2013, 33, 1144.)
[3] (a) Fischer, E.; Jourdan, F. Ber. Dtsch. Chem. Ges. 1883, 16, 2241. (b) Robinson, B. The Fischer Indole Synthesis, John Wiley & Sons Inc., New York, 1982. (c) Jiang, J.-Z.; Wang, Y. Chin. J. Org. Chem. 2006, 26, 1025 (in Chinese). (蒋金枝, 王艳, 有机化学, 2006, 26, 1025.)
[4] Larock, R. C.; Yum, E. K. J. Am. Chem. Soc. 1991, 113, 6689.
[5] (a) Song, J. J.; Reeves, J. T.; Fandrich, D. R.; Tan, Z.; Yee, N. K.; Senanayake, C. H. ARKIVOC 2010, 390. (b) Jin, D.; Zhang, F.; Zhang, D.-C. Chin. J. Org. Chem. 2010, 30, 1005 (in Chinese). (金丹, 张峰, 张德纯, 有机化学, 2010, 30, 1005.) (c) Liu, J.-Q.; Qian, C.; Chen, X.-Z. Chin. J. Org. Chem. 2011, 31, 634 (in Chinese). (刘金强, 钱超, 陈新志, 有机化学, 2011, 31, 634.) (d) Inman, M.; Moody, C. J. Chem. Sci. 2013, 4, 29. (e) Zhang, D. Qin Y. Acta Chim. Sinica 2013, 71, 147 (in Chinese). (张丹, 秦勇, 化学学报, 2013, 71, 147.) (f) Li, S.; Han, J.; Li, A. Acta Chim. Sinica 2013, 71, 295 (in Chinese).(李森, 韩静, 李昂, 化学学报,2013, 71, 295.) (g) Xie, W.; Zuo, Z.; Z, W.; Ma, D. Chin. J. Org. Chem. 2013, 33, 869 (in Chinese). (谢卫青, 左智伟, 资伟伟, 马大为, 有机化学, 2013, 33, 869.) (h) Guo, T.; Huang, F.; Yu, L.; Yu, Z. Tetrahedron Lett. 2015, 56, 296.
[6] (a) Würtz, S.; Rakshit, S.; Neumann, J. J.; Dröge, T.; Glorius, F. Angew. Chem., Int. Ed. 2008, 47, 7230. (b) Neumann, J. J.; Rakshit, S.; Dröge, T.; Würtz, S.; Glorius, F. Chem. Eur. J. 2011, 17, 7298.
[7] Bernini, R.; Fabrizi, G.; Sferrazza, A.; Cacchi, S. Angew. Chem., Int. Ed. 2009, 48, 8078.
[8] Guan, Z.-H.; Yan, Z.-Y.; Ren, Z.-H.; Liu, X.-Y.; Liang, Y.-M. Chem. Commun. 2010, 46, 2823.
[9] Zoller, J.; Fabry, D. C.; Ronge, M. A.; Rueping, M. Angew. Chem., Int. Ed. 2014, 53, 13264.
[10] Wei, Y.; Deb, I.; Yoshikai, N. J. Am. Chem. Soc. 2012, 134, 9098.
[11] Stuart, D. R.; Bertrand-Laperle, M.; Burgess, K. M. N.; Fagnou, K. J. Am. Chem. Soc. 2008, 130, 16474.
[12] Stuart, D. R.; Alsabeh, P.; Kuhn, M.; Fagnou, K. J. Am. Chem. Soc. 2010, 132, 18326.
[13] Shi, Z.; Zhang, C.; Li, S.; Pan, D.; Ding, S.; Cui, Y.; Jiao, N. Angew. Chem., Int. Ed. 2009, 48, 4572.
[14] Chen, X.; Li, X.; Wang, N.; Jin, J.; Lu, P.; Wang, Y. Eur. J. Org. Chem. 2012, 4380.
[15] Ren, L.; Shi, Z.; Jiao, N. Tetrahedron 2013, 69, 4408.
[16] Chen, J.; Song, G.; Pan, C.-L.; Li, X. Org. Lett. 2010, 12, 5426.
[17] Chen, J.; Pang, Q.; Sun, Y.; Li, X. J. Org. Chem. 2011, 76, 3523.
[18] Zhou, F.; Han, X.; Lu, X. Tetrahedron Lett. 2011, 52, 4681.
[19] Huestis, M. P.; Chan, L.; Stuart, D. R.; Fagnou, K. Angew. Chem., Int. Ed. 2011, 50, 1338.
[20] Ackermann, L.; Lygin, A. V. Org. Lett. 2012, 14, 764.
[21] Song, W.; Ackermann, L. Chem. Commun. 2013, 49, 6638.
[22] Zhang, G.; Yu, H.; Qin, G.; Huang, H. Chem. Commun. 2014, 50, 4331.
[23] Zhang, X.; Li, Y.; Shi, H.; Zhang, L.; Zhang, S.; Xu, X.; Liu, Q. Chem. Commun. 2014, 50, 7306.
[24] Tao, P.; Jia, Y. Chem. Commun. 2014, 50, 7367.
[25] Zhou, B.; Yang, Y.; Tang, H.; Du, J.; Feng, H.; Li, Y. Org. Lett. 2014, 16, 3900.
[26] Cajaraville, A.; López, S.; Varela, J. A.; Saá, C. Org. Lett. 2013, 15, 4576.
[27] Sharma, U.; Kancherla, R.; Naveen, T.; Agasti, S.; Maiti, D. Angew. Chem., Int. Ed. 2014, 53, 11895.
[28] Zhao, D.; Shi, Z.; Glorius, F. Angew. Chem., Int. Ed. 2013, 52, 12426.
[29] Zhang, Z.; Jiang, H.; Huang, Y. Org. Lett. 2014, 16, 5976.
[30] Fan, Z.; Song, S.; Li, W.; Geng K.; Xu, Y.; Miao, Z.-H.; Zhang, A. Org. Lett. 2015, 17, 310.
[31] Liu, B.; Song, C.; Sun, C.; Zhou, S.; Zhu, J. J. Am. Chem. Soc. 2013, 135, 16625.
[32] Wang, C.; Huang, Y. Org. Lett. 2013, 15, 5294.
[33] Matsuda, T.; Tomaru, Y. Tetrahedron Lett. 2014, 55, 3302.
[34] Zheng, L.; Hua, R. Chem. Eur. J. 2014, 20, 2352.
[35] Muralirajan, K.; Cheng, C.-H. Adv. Synth. Catal. 2014, 356, 1571.
[36] Wang, C.; Sun, H.; Fang, Y.; Huang, Y. Angew. Chem., Int. Ed. 2013, 52, 5795.
[37] Sun, H.; Wang, C.; Yang, Y.-F.; Chen, P.; Wu, Y.-D.; Zhang, X.; Huang, Y. J. Org. Chem. 2014, 79, 11863.
[38] Tan, Y.; Hartwig, J. F. J. Am. Chem. Soc. 2010, 132, 3676.
[39] Stokes, B. J.; Dong, H.; Leslie, B. E.; Pumphrey, A. L.; Driver, T. G. J. Am. Chem. Soc. 2007, 129, 7500.
[40] Shen, M.; Leslie, B. E.; Driver, T. G. Angew. Chem., Int. Ed. 2008, 47, 5056.
[41] Maity, S.; Zheng, N. Angew. Chem., Int. Ed. 2012, 51, 9562.
[42] Zhu, S.; Das, A.; Bui, L.; Zhou, H.; Curran, D. P.; Rueping, M. J. Am. Chem. Soc. 2013, 135, 1823.
[43] Gogoi, A.; Guin, S.; Rout, S. K.; Patel, B. K. Org. Lett. 2013, 15, 1802.
[44] Shibata, T.; Hirashima, H.; Kasagawa, M.; Tsuchikama, K.; Endo, K. Synlett 2011, 2171.
[45] Nanjo, T.; Tsukano, C.; Takemoto, Y. Org. Lett. 2012, 14, 4270.
[46] Yamagishi, M.; Nishigai, K.; Ishii, A.; Hata, T.; Urabe, H. Angew. Chem. Int. Ed. 2012, 51, 6471.
[47] Dangel, B. D.; Godula, K.; Youn, S. W.; Sezen, B.; Sames, D. J. Am. Chem. Soc. 2002, 124, 11856.
/
| 〈 |
|
〉 |