

Chinese Journal of Organic Chemistry >
Agarose Hydrogel Entrapped Trisodium Citrate Catalyzed Multicomponent Reactions for the Synthesis of Benzopyran and Pyranopyrazole Derivatives
Received date: 2015-10-11
Revised date: 2015-11-25
Online published: 2015-12-07
Supported by
Project supported by the National Natural Science Foundation of China (Nos. 21372099, 21072077), the Guangdong Natural Science Foundation (Nos. 10151063201000051, 8151063201000016).
An efficient method for the synthesis of benzopyran derivatives and pyrano[2,3-c]pyrazole derivatives via multicomponent reaction in the presence of agarose hydrogel entrapped trisodium citrate (gel-citrate) as catalyst is developed. The protocol presented here has the merits of easy preparation and handling of catalyst, environmentally benign, simple operation and convenient workup, and excellent yields. Moreover, the catalyst could be recovered and reused at least 6 cycles without apparently losing its activities.
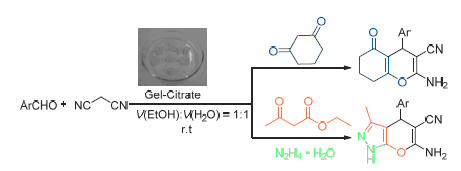
Li Zhipeng , Chen Feiran , Wang Dongyang , Huang Xingtian , Li Yiqun . Agarose Hydrogel Entrapped Trisodium Citrate Catalyzed Multicomponent Reactions for the Synthesis of Benzopyran and Pyranopyrazole Derivatives[J]. Chinese Journal of Organic Chemistry, 2016 , 36(4) : 838 -843 . DOI: 10.6023/cjoc201510010
[1] Strecker, A. Ann. 1850, 75, 27.
[2] Hantzsch, A. Ber. Dtsch. Chem. Ges. 1881, 14, 1637.
[3] Mannich, C.; Krösche, W. Arch. Pharm. 1912, 250, 647.
[4] Ugi, I. Angew. Chem., Int. Ed. Engl. 1962, 1, 8.
[5] (a) Ruijter, E.; Scheffelaar, R.; Orru, R. V. Angew. Chem., Int. Ed. Engl. 2011, 50, 6234.
(b) Wang, J.; Shen, Q.; Li, P.; Peng, Y.; Song, G. Org. Biomol. Chem. 2014, 12, 5597.
(c) Banfi, L.; Basso, A.; Moni, L.; Riva, R. Eur. J. Org. Chem. 2014, 2014, 2005.
(d) Bharti, R.; Parvin, T. RSC Adv. 2015, 5, 66833.
(e) Nikbakht, A.; Ramezanpour, S.; Balalaie, S.; Rominger, F. Tetrahedron 2015, 71, 6790.
(f) Liu, B.; Wei, E.; Lin, S.; Zhao, B.; Liang, F. Chem. Commun. 2014, 50, 6995.
[6] (a) Wang, Q. F.; Song, X. K.; Yan, C. G. Prog. Chem. 2009, 21, 997 (in Chinese). (王琦芳, 宋肖锴, 颜朝国, 化学进展, 2009, 21, 997.)
(b) Guo, H. Y.; Tian, J. J. Chin. J. Org. Chem. 2011, 31, 1752 (in Chinese). (郭红云, 田金金, 有机化学, 2011, 31, 1752.)
(c) Han, Y.; Sun, J.; Sun, Y.; Gao, H.; Yan, C. G. Chin. J. Org. Chem. 2012, 32, 1577 (in Chinese). (韩莹, 孙晶, 孙岩, 高红, 颜朝国, 有机化学, 2012, 32, 1577.)
(d) Xiao, L. W.; Peng, X. X.; Zhou, Q. X.; Kou, W.; Shi, Y. R. Chin. J. Org. Chem. 2015, 35, 1204 (in Chinese). (肖立伟, 彭晓霞, 周秋香, 寇伟, 时亚茹, 有机化学, 2015, 35, 1204.)
(e) Tian, J. J.; Guo, H. Y. Chin. J. Org. Chem. 2011, 31, 2009 (in Chinese). (田金金, 郭红云, 有机化学, 2011, 31, 2009.)
[7] (a) Bonsignore, L.; Loy, G.; Secci, D.; Calignano, A. Eur. J. Med. Chem. 1993, 28, 517.
(b) Zhang, G.; Zhang, Y. H.; Yan, J. X.; Chen, R.; Wang, S. L.; Ma, Y. X.; Wang, R. J. Org. Chem. 2012, 77, 878.
(c) Kuo, S. C.; Huang, L. J.; Nakamura, H. J. Med. Chem. 1984, 27, 539.
(d) Wang, J.-L.; Liu, D.; Zhang, Z.-J.; Shan, S.; Han, X.; Srinivasula, S. M.; Croce, C. M.; Alnemri, E. S.; Huang, Z. P. Natl. Acad. Sci. 2000, 97, 7124.
(e) Abdelrazek, F. M.; Metz, P.; Metwally, N. H.; El-Mahrouky, S. F. Arch. Pharm. 2006, 339, 456.
[8] Litvinov, Y. M.; Shestopalov, A. A.; Rodinovskaya, L. A.; Shestopalov, A. M. J. Comb. Chem. 2009, 11, 914.
[9] Vasuki, G.; Kumaravel, K. Tetrahedron Lett. 2008, 49, 5636.
[10] Ilovaisky, A. I.; Medvedev, M. G.; Merkulova, V. M.; Elinson, M. N.; Nikishin, G. I. J. Heterocycl. Chem. 2014, 51, 523.
[11] Zheng, J.; Li, Y. Mendeleev Commun. 2011, 21, 280.
[12] Zhao, L. Q.; Li, Y. Q.; Chen, L.; Zhou, B. Chin. J. Org. Chem. 2010, 30, 124 (in Chinese). (赵丽琴, 李毅群, 陈路, 周波, 有机化学, 2010, 30, 124.)
[13] Guo, R. Y.; An, Z. M.; Mo, L. P.; Yang, S. T.; Liu, H. X.; Wang, S. X.; Zhang, Z. H. Tetrahedron 2013, 69, 9931.
[14] Elnagdi, N. M. H.; Al-Hokbany, N. S. Molecules 2012, 17, 4300.
[15] Bihani, M.; Bora, P. P.; Bez, G.; Askari, H. ACS Sustainable Chem. Eng. 2013, 1, 440.
[16] Mecadon, H.; Rohman, M. R.; Rajbangshi, M.; Myrboh, B. Tetrahedron Lett. 2011, 52, 2523.
[17] Wang, X. S.; Shi, D. Q.; Tu, S. J.; Yao, C. S. Synth. Commun. 2003, 33, 119.
[18] Tu, S. J.; Gao, Y.; Guo, C.; Shi, D. Q.; Lu, Z. S. Synth. Commun. 2002, 32, 2137.
[19] (a) Leadbeater, N. E.; Marco, M. Chem. Rev. 2002, 102, 3217.
(b) Buchmeiser, M. R. Chem. Rev. 2008, 109, 303.
[20] (a) De Vos, D. E.; Dams, M.; Sels, B. F.; Jacobs, P. A. Chem. Rev. 2002, 102, 3615.
(b) Thomas, J. M.; Raja, R. Acc. Chem. Res. 2008, 41, 708.
[21] Oded, K.; Musa, S.; Gelman, D.; Blum, J. Catal. Commun. 2012, 20, 68.
[22] Abu-Reziq, R.; Shenglof, M.; Penn, L.; Cohen, T.; Blum, J. J. Mol. Catal. A: Chem. 2008, 290, 30.
[23] Ciriminna, R.; Fidalgo, A.; Pandarus, V.; Béland, F.; Ilharco, L. M.; Pagliaro, M. ChemCatChem 2015, 7, 254.
[24] Diz, P.; Pernas, P.; El Maatougui, A.; Tubio, C. R.; Azuaje, J.; Sotelo, E.; Guitián, F.; Gil, A.; Coelho, A. Appl. Catal. A: Gen. 2015, 502, 86.
[25] Bandgar, B. P.; Uppalla, L. S. Synth. Commun. 2000, 30, 2071.
[26] Chaphekar, S. S.; Samant, S. D. Appl. Catal. A: Gen. 2003, 242, 11.
[27] Shinde, S.; Rashinkar, G.; Kumbhar, A.; Kamble, S.; Salunkhe, R. Helv. Chim. Acta 2011, 94, 1943.
[28] Shinde, S.; Rashinkar, G.; Salunkhe, R. J. Mol. Liq. 2013, 178, 122.
[29] Jadhav, S.; Kumbhar, A.; Kamble, S.; More, P.; Salunkhe, R. C. R. Chim. 2013, 16, 957.
[30] Natekar, R.; Samant, S. Indian J. Chem., Sect. B: Org. Chem. Incl. Med. Chem. 1996, 35, 1347.
[31] (a) Dippy, J. F. J.; Evans, R. M. J. Org. Chem. 1950, 15, 451.
(b) Li, Y. L.; Zhou, J. F.; Gong, G. X.; Zhu, F. X. Chin. J. Org. Chem. 2009, 29, 441 (in Chinese). (李艳伦, 周建峰, 贡桂霞, 朱凤霞, 有机化学, 2009, 29, 441.)
(c) Bian, Q. L.; Xu, S.; Duan, W. L. Chin. J. Org. Chem. 2015, 35, 234 (in Chinese). (边庆龙, 许胜, 段伟良, 有机化学, 2015, 35, 234.)
[32] Nasseri, M. A.; Sadeghzadeh, S. M. Monatsh. Chem. 2013, 144, 1551.
[33] Elinson, M. N.; Dorofeev, A. S.; Feducovich, S. K.; Gorbunov, S. V.; Nasybullin, R. F.; Miloserdov, F. M.; Nikishin, G. I. Eur. J. Org. Chem. 2006, 4335.
[34] Peng, Y.; Song, G.; Dou, R. Green Chem. 2006, 8, 573.
[35] Sharanina, L. G.; Promonenkov, V. K.; Marshtupa, V. P.; Pashchenko, A. V.; Puzanova, V. V.; Sharanin, Y. A.; Klyuev, N. A.; Gusev, L. F.; Gnatusina, A. P. Chem. Heterocycl. Compd. 1982, 18, 607.
/
| 〈 |
|
〉 |