

Chinese Journal of Organic Chemistry >
Brønsted Acid Catalyzed Reaction of ortho-Hydroxylstyrenes with Indoles: Synthesis of 1,1-Diarylethanes
Received date: 2016-01-02
Revised date: 2016-01-26
Online published: 2016-02-02
Supported by
Project supported by the National Natural Science Foundation of China (No. 21372002) and the Brand Major of Universities in Jiangsu Province.
The skeleton of 1,1-diarylethane exists in many biologically important molecules. So, the synthesis of 1,1-diarylethanes has absorbed great attention from the chemists. This work has established a reaction of ortho-hydroxylstyrenes with indoles in the presence of racemic phosphoric acid, leading to the efficient synthesis of a series of 1,1-diarylethanes with structural diversity in high yields (up to 99%). This reaction is based on the fact that ortho-hydroxylstyrenes could transform into ortho-quinone methide under the catalysis of racemic phosphoric acid. The dual activation mode of racemic phosphoric acid to ortho-quinone methide and indole via forming two hydrogen bonds contributed greatly to the efficiency of the reaction. The mild reaction condition and wide substrate scope of the reaction will not only provide a useful strategy for solving the problems of residual metal and harsh reaction condition associated with metal catalysis, but also expand the applicability of organocatalytic reactions for the synthesis of 1,1-diarylethanes.
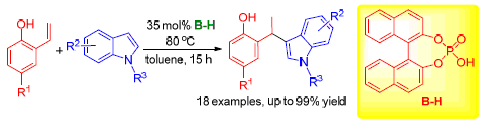
Jiang Xiaoli , Dai Wei , Zhao Jiajia , Shi Feng . Brønsted Acid Catalyzed Reaction of ortho-Hydroxylstyrenes with Indoles: Synthesis of 1,1-Diarylethanes[J]. Chinese Journal of Organic Chemistry, 2016 , 36(5) : 1014 -1020 . DOI: 10.6023/cjoc201601002
[1] (a) Cheltsov, A. V.; Aoyagi, M.; Aleshin, A.; Yu, E. C.-W.; Gilliland, T.; Zhai, D.; Bobkov, A. A.; Reed, J. C.; Liddington, R. C.; Abagyan, R. J. Med. Chem.2010, 53, 3899.
(b) Pathak, T. P.; Osiak, J. G.; Vaden, R. M.; Welm, B. E.; Sigman, M. S. Tetrahedron 2012, 68, 5203.
(c) Messaoudi, S.; Hamze, A.; Provot, O.; Tréguier, B.; Rodrigo De Losada, J.; Bignon, J.; Liu, J.-M.; Wdzieczak-Bakala, J.; Thoret, S.; Dubois, J.; Brion, J.-D.; Alami, M. ChemMedChem 2011, 6, 488.
[2] (a) Nakao, Y.; Kanyiva, K. S.; Oda, S.; Hiyama, T. J. Am. Chem. Soc. 2006, 128, 8146.
(b) Kanyiva, K. S.; Nakao, Y.; Hiyama, T. Heterocycles 2007, 72, 677.
(c) Wang, M.-Z.; Wong, M.-K.; Che, C.-M. Chem. Eur. J. 2008, 14, 8353.
(d) Xiao, Y.-P.; Liu, X.-Y.; Che, C.-M. J. Organomet.Chem. 2009, 694, 494.
(e) Nakao, Y.; Kashihara, N.; Kanyiva, K. S.; Hiyama, T. Angew. Chem., Int. Ed. 2010, 49, 4451.
(f) Zhou, F.; Cao, Z.-Y.; Zhang, J.; Yang, H.-B.; Zhou, J. Chem. Asian J.2012, 7, 233.
(g) Chen, L.; Zhou, J. Chem. Asian J. 2012, 7, 2510.
[3] Pathak, T. P.; Sigman, M. S. Org. Lett. 2011, 13, 2774.
[4] (a) Zheng, C.; You, S.-L. Chem. Soc.Rev.2012, 41, 2498; (b) Pellissier, H. Adv. Synth. Catal.2012, 354, 237.
[5] (a) Akiyama, T. Chem.Rev.2007, 107, 5744.
(b) Terada, M. Chem. Commun.2008, 4097.
(c) Terada, M. Synthesis2010, 1929.
(d) Yu, J.; Shi, F.; Gong, L.-Z. Acc. Chem. Res.2011, 44, 1156.
(e) Parmar, D.; Sugiono, E.; Raja, S.; Rueping, M. Chem.Rev.2014, 114, 9047.
(f) Huang, J.; Luo, S.; Gong, L. Acta Chim. Sinica2013, 71, 879.
[6] (a) Tan, W.; Li, X.; Gong, Y.-X.; Ge, M.-D.; Shi, F. Chem.Commun. 2014, 50, 15901.
(b) Zhang, Y.-C.; Zhao, J.-J.; Jiang, F.; Sun, S.-B.; Shi, F. Angew. Chem., Int. Ed. 2014, 53, 13912.
(c) Zhao, J.-J.; Sun, S.-B.; He, S.-H.; Wu, Q.; Shi, F. Angew. Chem., Int. Ed.2015, 54, 5460.
[7] Dai, W.; Lu, H.; Jiang, X.-L.; Gao, T.-T.; Shi, F. Tetrahedron: Asymmetry 2015, 26, 109.
[8] Wang, Z.-B.; Ai, F.-J.; Wang, Z.; Zhao,W.-X.; Zhu, G.-Y.; Lin, Z.-Y.; Sun, J.-W. J. Am.Chem. Soc. 2015, 137, 383.
[9] Li, M.-L.; Chen, D.-F.; Luo, S.-W.; Wu, X. Tetrahedron: Asymmetry 2015, 26, 219.
/
| 〈 |
|
〉 |