

Chinese Journal of Organic Chemistry >
Progress of the Research on the Lewis/Brønsted Acid-Catalyzed Nucleophilic Substitution of Propargyl Alcohols
Received date: 2015-12-02
Revised date: 2015-12-30
Online published: 2016-02-01
Supported by
Project supported by the Young National Natural Science Foundation of China (No. 21302096), the Young Natural Science Foundation of Jiangsu Province (Nos. BK20130962, BK20130952) and the Project Fund from the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD).
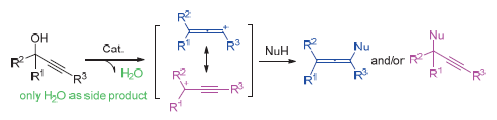
Lewis/Brønsted acid-catalyzed nucleophilic substitution of propargylic alcohols is very important in organic synthetic chemistry, which could be transformed into a variety of acyclic, carbocyclic and heterocyclic synthetic building blocks. A drawback of the traditional propargylation of Nicholas reaction is the generation of waste products resulting from displacement of the leaving group on treating with a catalyst and/or nucleophile. Therefore, the direct acid-catalyzed propargylation of propargylic alcohols is considered as a green method, which generated water as the only side product. In this review, the latest research progress on the Lewis and Brønsted acids catalyzed intermolecular and intramolecular propargylation of a variety of nucleophiles (NuH=C, N, O, S, I) with propargylic alcohols is presented. Finally, the problems and difficulties in research and application of propargylation of propargylic alcohols are discussed and then prospective is provided.
Zhang Xiaoxiang, Lü Chang, Li Ping, Fu Bo, Yao Weiwei . Progress of the Research on the Lewis/Brønsted Acid-Catalyzed Nucleophilic Substitution of Propargyl Alcohols[J]. Chinese Journal of Organic Chemistry, 2016 , 36(6) : 1287 -1298 . DOI: 10.6023/cjoc201512003
[1] (a) Ayers, B. J.; Chan, P. W. H. Synlett 2015, 26, 1305.
(b) Zhu, Y.; Sun, L.; Lu, P.; Wang, Y. ACS Catal. 2014, 4, 1911.
(c) Patil, N. T.; Kavthe, R. D.; Shinde, V. S. Tetrahedron 2012, 68, 8079.
(d) Zhang, L.; Fang, G.; Kumar, R. K.; Bi, X. Synthesis 2015, 47, 2317.
(e) Yang, Z.; Kumar, R. K.; Liao, P.; Liu, Z.; Li, X.; Bi, X. Chem. Commun. 2016, 52, 5936.
(f) Patil, N. T.; Kavthe, R. D.; Shinde, V. S. Tetrahedron 2012, 68, 8079.
(g) Shu, X.-Z.; Shu, D.; Schienebeck, C. M.; Tang, W. Chem. Soc. Rev. 2012, 41, 7698.
(h) Leyva-Pérez, A.; Corma, A. Angew. Chem., Int. Ed. 2012, 51, 614.
(i) Hao, L.; Zhan, Z.-P. Curr. Org. Chem. 2011, 15, 1625.
(j) Michelet, V.; Toullec, P. Y.; Genêt, J.-P. Angew. Chem., Int. Ed. 2008, 47, 4268.
(k) Patil, N. T.; Yamamoto, Y. Chem. Rev. 2008, 108, 3395.
(l) Yamamoto, Y. J. Org. Chem. 2007, 72, 7817.
[2] (a) Zhang, X.; Sun, X.; Tan, J.; Fan, H.; Rao, W. Chin. J. Org. Chem. 2015, 35, 2049 (in Chinese). (张小祥, 孙小萍, 谈继淮, 樊辉, 饶卫东, 有机化学, 2015, 35, 2049.)
(b) Zhang, X.; Sun, X.; Rao, W. Chin. J. Org. Chem. 2015, 35, 1500 (in Chinese). (张小祥, 孙小萍, 饶卫东, 有机化学, 2015, 35, 1500.)
(c) Zhang, X.; Teo, J. W.; Ma, D.-L.; Leung, C.-H.; Chan, P. W. H. Tetrahedron Lett. 2014, 55, 6703.
(d) Zhang, X.; Teo, W. T.; Rao, W.; Ma, D.-L.; Leung, C.-H.; Chan, P. W. H. Tetrahedron Lett. 2014, 55, 3881.
(e) Zhang, X.; Teo, W. T.; Chan, P. W. H. J. Organomet. Chem. 2011, 696, 331.
(f) Zhang, X.; Teo, W. T.; Sally, Chan, P. W. H. J. Org. Chem. 2010, 75, 6290.
(g) Zhang, X.; Teo, W. T.; Chan, P. W. H. Org. Lett. 2009, 11, 4990.
(h) Kothandaraman, P.; Rao, W.; Zhang, X.; Chan, P. W. H. Tetrahedron 2009, 65, 1833.
(i) Rao, W.; Zhang, X.; Sze, E. M. L.; Chan, P. W. H. J. Org. Chem. 2009, 74, 1740.
(j) Zhang, X.; Rao, W.; Sally, Chan, P. W. H. Org. Biomol. Chem. 2009, 7, 4186.
(k) Zhang, X.; Rao, W.; Chan, P. W. H. Synlett 2008, 2204.
[3] (a) Caffyn, A. J. M.; Nicholas, K. M. In Comprehensive Organometallic Chemistry II, Vol. 12, Eds.: Abel, E. W.; Stone, F. G. A.; Wilkinson, J., Pergamon, Oxford, 1995, Chapter 7.1.
(b) Nicholas, K. M.; Pettit, R. J. Organomet. Chem. 1972, 44, 21.
(c) Nicholas, K. M. Acc. Chem. Res. 1987, 20, 207.
(d) Green, J. R. Curr. Org. Chem. 2001, 5, 809.
(e) Teobald, B. J. Tetrahedron 2002, 58, 4133.
(f) Kuhn, O.; Rau, D.; Maayr, H. J. Am. Chem. Soc. 1998, 120, 900. (g) Müller, T. J. J. Eur. J. Org. Chem. 2001, 2021.
(h) Nishibayashi, Y.; Uemura, S. Curr. Org. Chem. 2006, 10, 135.
[4] Georgy, M.; Boucard, V.; Campagne, J. M. J. Am. Chem. Soc. 2005, 127, 14180.
[5] Zhan, Z.; Wang, W.; Yang, R.; Yu, J.; Li, J.; Liu, H. Chem. Commun. 2006, 3352.
[6] Zhan, Z.; Liu, J.; Liu, H.; Cui, Y.; Yang, R.; Yang, W.; Li, J. J. Org. Chem. 2006, 71, 8298.
[7] Sanz, R.; Miguel, D.; Martinez, A.; Alvarez-Gutierrez, J. M.; Rodriguez, F. Org. Lett. 2007, 9, 727.
[8] Yoshimatsu, M.; Otani, T.; Matsuda, S.; Yamamoto, T.; Sawa, A. Org. Lett. 2008, 10, 4251.
[9] Sanz, R.; Miguel, D.; Martinez, A.; Gohain, M.; Garcia-Garcia, P.; Fernandez-Rodriguez, A. M.; Alvarez, E.; Rodriguez, F. Eur. J. Org. Chem. 2010, 36, 7027.
[10] Silveira, C. C.; Mendes, S. R.; Wolf, L.; Martins, G. M. Tetrahedron Lett. 2010, 51, 4560.
[11] Gohain, M.; Marais, C.; Bezuidenhoudt, B. C. B. Tetrahedron Lett. 2012, 53, 4704.
[12] Yadav, J. S.; Reddy, B. V. S.; Thrimurtulu, N.; Mallikarjuna Reddy, N.; Prasad, A. R. Tetrahedron Lett. 2008, 49, 2031.
[13] Wang, T.; Ma, R. D.; Liu, L.; Zhan, Z. P. Green Chem. 2010, 12, 1576.
[14] Huang, W.; Shen, Q.; Wang, J.; Zhou, X. J. Org. Chem. 2008, 73, 1586.
[15] Huang, W.; Zheng, P.; Zhang, Z.; Liu, R.; Chen, Z.; Zhou, X. J. Org. Chem. 2008, 73, 6845.
[16] Chatterjee, P. N.; Roy, S. J. Org. Chem. 2010, 75, 4413.
[17] Zhao, W.; Carreira, E. M. Org. Lett. 2003, 5, 4153.
[18] Yuan, F. Q.; Han, F. S. Adv. Synth. Catal. 2013, 355, 537.
[19] Zhang, L.; Zhu, Y. X.; Yin, G.; Lu, P.; Wang, Y. J. Org. Chem. 2012, 77, 9510.
[20] Hao, L.; Pan, Y.; Wang, T.; Lin, M.; Chen, L.; Zhan, Z. Adv. Synth. Catal. 2010, 352, 3215.
[21] Song, J.-N.; Fang, Z.; Liu, Y.; Li, R.; Xu, L.; Barry, B.-D.; Liu, Q.; Bi, X.; Liao, P. Synlett 2011, 2551.
[22] Li, Q.; Wang, Y.; Fang, Z.; Liao, P.; Barry, B.-D.; Che, G.; Bi, X. Synthesis 2013, 45, 609.
[23] Liu, Y.; Barry, B.-D.; Yu, H.; Liu, J.; Liao, P.; Bi, X. Org. Lett. 2013, 15, 2608.
[24] Ji, K.-G.; Shu, X.-Z.; Zhao, S.-C.; Zhu, H.-T.; Niu, Y.-N.; Liu, X. Y.; Liang, Y.-M. Org. Lett. 2009, 11, 3206.
[25] Haven, T.; Kubik, G.; Haubenreisser, S.; Niggemann, M. Angew. Chem., Int. Ed. 2013, 52, 4016.
[26] Shi, M.; Yao, L.-F. Chem.-Eur. J. 2008, 14, 8725.
[27] Yao, L.-F.; Wei, Y.; Shi, M. J. Org. Chem. 2009, 74, 9466.
[28] Huang, G. B.; Wang, X.; Pan, Y. M.; Wang, H. S.; Yao, G. Y.; Zhang, Y. J. Org. Chem. 2013, 78, 2742.
[29] Yin, G.; Zhu, Y.; Lu, P.; Wang, Y. J. Org. Chem. 2011, 76, 8922.
[30] (a) Yin, G.; Zhu, Y.; Wang, N.; Lu, P.; Wang, Y. Tetrahedron 2013, 69, 8353.
(b) Similar reference, see: Shao, Y.; Zhu, K.; Qin, Z.; Li, E.; Li, Y. J. Org. Chem. 2013, 78, 5731.
[31] Wang, S. Y.; Zhu, Y. X.; Wang, Y. G.; Lu, P. Org. Lett. 2009, 11, 2615.
[32] Zhu, Y.; Yin, G.; Hong, D.; Lu, P.; Wang, Y. Org. Lett. 2011, 13, 1024.
[33] Zhu, Y. X.; Wen, S.; Yin, G. W.; Hong, D.; Lu, P.; Wang, Y. G. Org. Lett. 2011, 13, 3553.
[34] Yin, G. W.; Zhu, Y. X.; Zhang, L.; Lu, P.; Wang, Y. G. Org. Lett. 2011, 13, 940.
[35] Mothe, S. R.; Kothandaraman, P.; Lauw, S. J. L.; Chin, S. M. W.; Chan, P. W. H. Chem.-Eur. J. 2012, 18, 6133.
[36] Mothe, S. R.; Novianti, M. L.; Ayers, B. J.; Chan, P. W. H. Org. Lett. 2014, 16, 4110.
[37] (a) Meyer, K. H.; Schuster, K. Ber. 1922, 55B, 819.
(b) Swaminathan, S.; Narayanan, K. V. Chem. Rev. 1971, 71, 429.
(c) Engel, D. A.; Dudley, G. B. Org. Biomol. Chem. 2009, 7, 4149.
[38] Aponick, A.; Li, C.-Y.; Palmes, J. A. Org. Lett. 2009, 11, 121.
[39] Wang, L.; Xie, X.; Liu, Y. Org. Lett. 2012, 14, 5848.
[40] Knight, D. W. WO 2006100479, 2006[Chem. Abstr. 2006, 145, 377330].
[41] Aponick, A.; Li, C.-Y.; Malinge, J.; Marques, E. F. Org. Lett. 2009, 11, 4624.
[42] Egi, M.; Azechi, K.; Akai, S. Org. Lett. 2009, 11, 5002.
[43] Chen, S.; Wang, J. J. Org. Chem. 2007, 72, 4993.
[44] Ji, K. G.; Zhu, H. T.; Yang, F.; Shu, X. Z.; Zhao, S. C.; Liu, X. Y. Shaukat, A.; Liang, Y. M. Chem.-Eur. J. 2010, 16, 6151.
[45] Zhu, H. T.; Ji, K. G.; Yang, F.; Wang, L. J.; Zhao, S. C.; Ali, S.; Liu, X. Y.; Liang, Y. M. Org. Lett. 2011, 13, 684.
[46] Yang, F.; Jin, T.; Bao, M.; Yamamoto, Y. Tetrahedron Lett. 2011, 52, 936.
[47] Yang, F.; Jin, T.; Bao, M.; Yamamoto, Y. Chem. Commun. 2011, 47, 4541.
[48] Choi, J.; Lee, G. H.; Kim, I. Synlett 2008, 1243.
/
| 〈 |
|
〉 |