

Chinese Journal of Organic Chemistry >
Design, Synthesis and Repellent Activity of (E)-β-Farnesene Analogues Containing Benzene Ring with Different Substitutions
Received date: 2016-03-14
Revised date: 2016-04-07
Online published: 2016-04-20
Supported by
Project supported by the National Natural Science Foundation of China (No. 21132003).
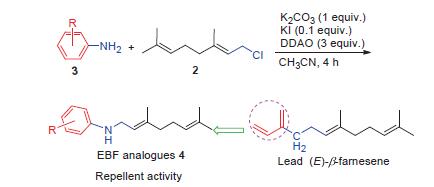
In order to discover novel compounds with high-activity to control aphid, aphid alarm pheromone (E)-β-farnesene (EBF) was chosen as lead compound and a series of substituted EBF analogues were designed by replacing unstable conjugated double bond of EBF with different substitutions benzene ring. An N-alkylation route was optimized by using aniline and (E)-1-chloro-3,7-dimethyl-2,6-octadiene. Nineteen EBF analogues were synthesized with yields of 39%~83% in this optimization reaction condition. Their structures were confirmed by 1H NMR, 13C NMR and HRMS analysis. Bioassay showed that the title compounds demonstrated repellent activity against Myzus persicae (Sulzer). In particular, 4d, 4h and 4k exhibited excellent repellent activity of 56.3%, 58.3% and 50.3% respectively. The performed structure-activity relationship indicated that introduction of halogen group at the 4 position of phenyl ring displayed relatively higher activity.
Zhang Jingpeng , Qin Yaoguo , Li Wenhao , Ling Yun , Yang Libo , Song Dunlun , Yang Xinling . Design, Synthesis and Repellent Activity of (E)-β-Farnesene Analogues Containing Benzene Ring with Different Substitutions[J]. Chinese Journal of Organic Chemistry, 2016 , 36(8) : 1883 -1889 . DOI: 10.6023/cjoc201603021
[1] Wang, G.-P.; Yu, X.-D.; Fan, J.; Wang, C.-S.; Xia, L.-Q. J. Integr. Plant. Biol. 2015, 57, 770.
[2] Bruce, T. J. A.; Birkett, M. A.; Blande, J.; Hooper, A. M.; Martin, J. L.; Khambay, B.; Prosser, I.; Smart, L. E.; Wadhams, L. J. Pest. Manag. Sci. 2005, 61, 1115.
[3] Kang, T.-N.; Ling, Y.; Rui, C.-H.; Yang, X.-L.; Fan, X.-L.; Chen, F.-H. Chin. J. Org. Chem. 2008, 28, 617 (in Chinese).
(康铁牛, 凌云, 芮昌辉, 杨新玲, 范贤林, 陈馥衡, 有机化学, 2008, 28, 617.)
[4] Dahl, M. L. Dtsch. Ent. Z. 1971, 18, 121.
[5] Kislow, C. J.; Edward, L. J. Nature 1972, 235, 108.
[6] Bowers, W. S.; Nault, L. R.; Webb, R. E.; Dutky, S. R. Science 1972, 177, 1121.
[7] Mauchamp, B.; Picker, J. J. Agronomie 1987, 7, 523.
[8] Kunert, G.; Otto, S.; Rose, U. R.; Gershenzon, J.; Weisser, W. W. Ecol. Lett. 2005, 8, 596.
[9] Joachim, C.; Weisser, W. W. J. Chem. Ecol. 2015, 47, 267.
[10] Francis, F.; Lognay, G.; Haubruge, E. J. Chem. Ecol. 2004, 30, 741.
[11] Hatano, E.; Kunert, G.; Weisser, W. W. Eur. J. Entomol. 2008, 105, 797.
[12] Heuskin, S.; Lorge, S.; Godin, B.; Leroy, P.; Frere, I.; Verheggen, F. J.; Haubruge, E.; Wathelet, J. P.; Mestdagh, M.; Hance, T.; Lognay, G.; Pest. Manage. Sci. 2012, 68, 127.
[13] Joachim, C.; Hatano, E.; David, A.; Kunert, M.; Linse, C.; Weisser, W. W. J. Chem. Ecol. 2013, 39, 773.
[14] Griffiths, D. C.; Pickett, J. A. Entomol. Exp. Appl. 1980, 27, 199.
[15] Dawson, G. W.; Giffiths, D. C.; Pickett, J. A.; Plumb, R. T.; Woodcock, C. M.; Zhang, Z.-N. Pestic. Sci. 1988, 22, l7.
[16] Bowers, W. S.; Nishino, C.; Montgomery, M. E.; Nault, L. R. Insects Physiol. 1977, 23, 697.
[17] Sun, L.; Ling, Y.; Wang. C.; Sun, Y.-F.; Rui, C.-H.; Yang, X.-L. Chin. J. Org. Chem. 2011, 31, 2061 (in Chinese).
(孙亮, 凌云, 王灿, 孙玉凤, 芮昌辉, 杨新玲, 有机化学, 2011, 31, 2061.)
[18] Qin, Y.-G.; Qu, Y.-Y.; Zhang, J.-P.; Tan, X.-Q.; Song, L.-F.; Li, W.-H.; Song, D.-L.; Yang, X.-L. Chin. J. Org. Chem. 2015, 35, 455 (in Chinese).
(秦耀果, 曲焱焱, 张景朋, 谭晓庆, 宋丽芳, 李文浩, 宋敦伦, 杨新玲, 有机化学, 2015, 35, 455.)
[19] Sun, Y.-F.; Li, Y.-Q.; Ling, Y.; Yu, H.-L.; Yang, S.-X.; Yang, X.-L. Chin. J. Org. Chem. 2011, 31, 1425 (in Chinese).
(孙玉凤, 李永强, 凌云, 宇红莲, 杨绍祥, 杨新玲, 有机化学, 2011, 31, 1425.)
[20] Sun, Y.-F.; Filomena, D.; Qiao, H.-L.; Immacolata, I.; Yang, S.-X.; Ling, Y.; Lea, R.; Donatella, B.; Patrizia, F.; Yang, X.-L.; Paolo, P. PLoS One 2012, 7, e32759.
[21] Sun, Y.-F.; Qiao, H.-L.; Ling, Y.; Yang, S.-X.; Rui, C.-H.; Paolo Pelosi; Yang, X.-L. J. Agric. Food Chem. 2011, 59, 2456.
[22] Ohshima, T,; Miyamoto, Y,; Ipposhi, J.; Nakahara, Y.; Utsunomiya, M.; Mashima, K. J. Am. Chem. Soc. 2009, 131, 14317.
[23] Zhao, Y.-S.; Foo, S. W.; Saito, S. Angew. Chem., Int. Ed. 2011, 50, 3006.
[24] Kazuhide, T.; Tsuneaki, Y.; Susumu, A.; Hidenori, K.; Takanao, T.; Hidemasa, T.; Akira, M.; Ryoji, N.; Sei, O. J. Am. Chem. Soc. 1984, 106, 5208.
[25] Shi, H.; Lu, F.; Xiong, J.-J.; Xu, Y.-X.; Wang, J. Fine Chem. Intermed. 2008, 38, 8 (in Chinese).
(师华, 陆峰, 熊家锦, 徐永祥, 王菁, 精细化工中间体, 2008, 38, 8.)
[26] Peat, A. J.; Buchwald, S. L. J. Am. Chem. Soc. 1996, 118, 1028.
[27] Romera, J. L.; Cid, J. M.; Trabanco, A. A. Tetrahedron Lett. 2004, 45, 8797.
[28] Siswanto, C.; Rathman, J. F. J. Colloid Interface Sci. 1997, 196, 99.
[29] Pinchin, R.; De Oliveira Filho, A. M.; Figueiredo, M. J.; Muller, C. A.; Gilbert, B.; Szumlewicz, A. P.; Benson, W. W. J. Econ. Entomol. 1978, 71, 950.
[30] Sun, L. Ph.D. Dissertation, China Agricultural University, Beijing, 2013 (in Chinese).
/
| 〈 |
|
〉 |