

Chinese Journal of Organic Chemistry >
Tandem Cyclization Reaction between Optically Active γ-Nitro Ketone and Chalcone towards the Synthesis of Chiral Cyclohexane Skeletons Bearing Five Stereocenters
Received date: 2016-05-04
Revised date: 2016-05-31
Online published: 2016-06-01
Supported by
Project supported by the Research Grant in 2015 of the South University of Science and Technology of China (No. FRG-SUSTC1501A-57), the National Natural Science Foundation of China (No. 21302089) and the Science and Technology Development Program of Hangzhou (No. 20130533B14).
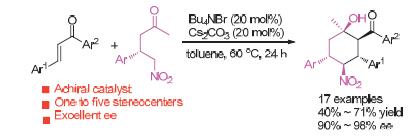
Chiral cyclohexane motif is widespread in the molecular structures of nature products and medicinal chemicals. And polysubstituted chiral cyclohexanes are important building blocks in organic synthesis. A tandem cyclization via asymmetric induction for the construction of polysubstituted chiral cyclohexane skeletons is described. In the presence of phase-transfer-catalyst, optically active γ-nitro ketone reacted with chalcone smoothly to afford cyclohexane skeletons bearing five consecutive stereocenters in 40%~71% yields with 90%~98% ee. It should be noted that the scope of chalcone is broad. Especially, the asymmetric induction is kept at a high level in each step of the tandem cyclization reaction.
Key words: nitro ketone; chalcone; tandem cyclization reaction; cyclohexane
Yu Lu , Cheng Yuyu , Li Rou , Jiao Yanhua , Li Pengfei . Tandem Cyclization Reaction between Optically Active γ-Nitro Ketone and Chalcone towards the Synthesis of Chiral Cyclohexane Skeletons Bearing Five Stereocenters[J]. Chinese Journal of Organic Chemistry, 2016 , 36(7) : 1572 -1579 . DOI: 10.6023/cjoc201605006
[1] For selected reviews, see: (a) Tietze, L. F. Chem. Rev. 1996, 96, 115.
(b) Parsons, P. J.; Penkett, C. S.; Shell, A. J. Chem. Rev. 1996, 96, 195.
(c) Pellissier, H. Tetrahedron 2006, 62, 1619.
(d) Pellissier, H. Tetrahedron 2006, 62, 2143.
(e) Nicolaou, K. C.; Edmonds, D. J.; Bulger, P. G. Angew. Chem., Int. Ed. 2006, 45, 7134.
(f) Chapman, C. J.; Frost, C. G. Synthesis 2007, 1.
(g) Enders, D.; Grondal, C.; Hüttl, M. R. M. Angew. Chem., Int. Ed. 2007, 46, 1570.
(h) Nicolaou, K. C.; Chen, J. S. Chem. Soc. Rev. 2009, 38, 2993.
(i) Grondal, C.; Jeanty, M.; Enders, D. Nat. Chem. 2010, 2, 167.
(j) Ruiz, M.; López-Alvarado, P.; Giorgi, G., Menéndez, J. C. Chem. Soc. Rev. 2011, 40, 3445.
(k) Albrecht, ?.; Jiang, H.; Jørgensen, K. A. Angew. Chem., Int. Ed. 2011, 50, 8492.
(l) Pellissier, H. Adv. Synth. Catal. 2012, 354, 237.
(m) Pellissier, H. Chem. Rev. 2013, 113, 442.
(n) Volla, C. M. R.; Atodiresei, I.; Rueping, M. Chem. Rev. 2014, 114, 2390.
(o) Vetica, F.; de Figueiredo, R. M.; Orsini, M.; Tofani, D.; Gasperi, T. Synthesis 2015, 47, 2139.
[2] (a) Merino, P.; Marqués-López, E.; Tejero, T.; Herrera, R. P. Synthesis 2010, 1.
(b) Moyano, A.; Rios, R. Chem. Rev. 2011, 111, 4703.
(c) Pellissier, H. Tetrahedron 2012, 68, 2197.
(d) Xie, P.; Huang, Y. Eur. J. Org. Chem. 2013, 6213.
(e) Yang, X.; Wang, J.; Li, P. Org. Biomol. Chem. 2014, 12, 2499.
[3] (a) Posner, G. H. Chem. Rev. 1986, 86, 831.
(b) Schultz, A. G. Acc. Chem. Res. 1990, 23, 207.
(c) Li J. K. In Name Reaction for Carbocyclic Ring Formarions, Wiley, Hoboken, NJ, 2010, p. 197~142.
(d) Goudedranche, S.; Raimondi, W.; Bugaut, X.; Constantieux, T.; Bonne, D.; Rodriguez, J. Synthesis 2013, 45, 1909.
[4] (a) Enders, D.; Hüttl, M. R. M.; Grondal, C.; Raabe, G. Nature 2006, 441, 861.
(b) Enders, D.; Hüttl, M. R. M.; Runsink, J.; Raabe, G.; Wendt, B. Angew. Chem., Int. Ed. 2007, 46, 467.
(c) Enders, D.; Hüttl, M. R. M.; Raabe, G.; Bats, J. W. Adv. Synth. Catal. 2008, 350, 267.
(d) Ishikawa, H.; Suzuki, T.; Orita, H.; Uchimaru, T.; Hayashi, Y. Chem. Eur. J. 2010, 16, 12616.
(e) Anwar, S.; Chang, H.-J.; Chen, K. Org. Lett. 2011, 13, 2200.
(f) Ma, G.; Lin, S.; Ibrahem, I.; Kubik, G.; Liu, L.; Sun, J.; Córdova, A. Adv. Synth. Catal. 2012, 354, 2865.
[5] (a) Han, B.; Xiao, Y.-C.; He, Z.-Q.; Chen, Y.-C. Org. Lett. 2009, 11, 4660.
(b) Wang, Y.; Yu, D.-F.; Liu, Y.-Z.; Wei, H.; Luo, Y.-C.; Dixon, D. J.; Xu, P.-F. Chem. Eur. J. 2010, 16, 3922.
[6] (a) Ishikawa, H.; Sawano, S.; Yasui, Y.; Shibata, Y.; Hayashi, Y. Angew. Chem., Int. Ed. 2011, 50, 3774.
(b) Hahn, R.; Raabe, G.; Enders, D. Org. Lett. 2014, 16, 3636.
[7] (a) Mao, Z.; Jia, Y.; Xu, Z.; Wang, R. Adv. Synth. Catal. 2012, 354, 1401.
(b) Enders, D.; Hahn, R.; Atodiresei, I. Adv. Synth. Catal. 2013, 355, 1126.
(c) Chauhan, P.; Mahajan, S.; Loh, C. C. J.; Raabe, G.; Enders, D. Org. Lett. 2014, 16, 2954.
(d) Chauhan, P.; Urbanietz, G.; Raabe, G.; Enders, D. Chem. Commun. 2014, 50, 6853.
(e) Zhou, R.; Wu, Q.; Guo, M.; Huang, W.; He, X.; Yang, L.; Peng, F.; He, G.; Han, B. Chem. Commun. 2015, 51, 13113.
[8] Xie, X.; Peng, C.; He, G.; Leng, H.-J.; Wang, B.; Huang, W.; Han, B. Chem. Commun. 2012, 48, 10487.
[9] (a) Varga, S.; Jakab, G.; Drahos, L.; Holczbauer, T.; Czugler, M.; Soós, T. Org. Lett. 2011, 13, 5416.
(b) Sun, J.; Jiang, C.; Zhou, Z. Eur. J. Org. Chem. 2016, 1165.
[10] Duan, J.-D.; Cheng, J.; Li, P.-F. Org. Chem. Front. 2015, 2, 1048.
[11] Yu, L.; Yang, Z.; Peng, J.-Z.; Li, P.-F. Eur. J. Org. Chem. 2016, 535.
[12] Yu, L.; Yang, Q.-J.; Li, P.-F. Eur. J. Org. Chem. 2014, 7499.
[13] Huang, H.; Jacobsen, E. N. J. Am. Chem. Soc. 2006, 128, 7170.
/
| 〈 |
|
〉 |