

Chinese Journal of Organic Chemistry >
Synthesis of Thiophene Formyl Thiourea Derivatives and Fungicidal Activity
Received date: 2016-05-10
Revised date: 2016-06-12
Online published: 2016-07-08
Supported by
Project supported by the Special Fund for Agro-scientific Research in the Public Interest (No.201503112) and the Key Scientific Research Project of Colleges and Universities in Henan Province (No.16A210006).
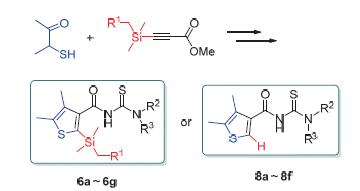
13 New thiophene formyl thiourea derivatives were synthesized via cycloaddition reaction of 3-mercaptobutan-2-one and methyl 3-(substitutedsilyl)propiolate, selective hydrolysis of ester, isothiocyanatation and amination. Their chemical structures were confirmed by 1H NMR, 13C NMR and elemental analysis. The inhibitory activity against Gaeumannomyces graminis var. tritici and Rhizoctonia solani was evaluated in vitro by the plate method. The results indicated that most of thiophene formyl thiourea derivatives showed some inhibitory activity against gaeumannomyces graminis var. tritici. Compound 1-cyclopropyl-3-(4,5-dimethylthiophene-3-carbonyl)thiourea (8c) gave the best performance and its inhibitory activity was close to the control level of silthiopham in the concentration of 10 mg/L. The structure analysis showed that the steric hindrance of functional group at the second position of the thiophene formyl thioureas had no obvious effect on the inhibitory activity against gaeumannomyces graminis var. tritici.
Cheng Yinan , Jin Wenbo , Xie Guiying , Ma Yichao , Zhao Yanqin , Li Honglian . Synthesis of Thiophene Formyl Thiourea Derivatives and Fungicidal Activity[J]. Chinese Journal of Organic Chemistry, 2016 , 36(11) : 2683 -2688 . DOI: 10.6023/cjoc201605012
[1] Yan, Z. K.; Song, B. A.; Yang, X.; Hu, D. Y.; Yang, S.; Jin, L. H.; Chen, G. M. Agrochemicals 2008, 47, 706 (in Chinese). (闫志坤, 宋宝安, 杨璇, 胡德禹, 杨松, 金林红, 陈广明, 农药, 2008, 47, 706.)
[2] Ishaaya, I.; Mendelson, Z.; Horowitz, A. R. Phytoparasitica 1993, 21, 199.
[3] Sun, Z. H.; Huang, W.; Gong, Y. Y.; Lan, J.; Liu, X. H.; Weng, J. Q.; Li, Y. S.; Tan, C. X. Chin. J. Org. Chem. 2013, 33, 2612 (in Chinese). (孙召慧, 黄伟, 贡云芸, 蓝健, 刘幸海, 翁建全, 李永曙, 谭成侠, 有机化学, 2013, 33, 2612.)
[4] Miao, H. J.; Zhang, J. W.; Yuan, H. Z.; Li, Y.; Xu, Y.; Li, H.; Yang, X. L.; Ling, Y. Chin. J. Org. Chem. 2012, 32, 915 (in Chinese). (苗宏健, 张继伟, 袁会珠, 李映, 徐焱, 李慧, 杨新玲, 凌云, 有机化学, 2012, 32, 915.)
[5] Saeed, A.; Khan, M. S.; Rafique, H.; Shahid, M.; Iqbal, J. Bioorg. Chem. 2014, 52, 1.
[6] Williams, D. J.; Dimmic, M. W.; Haakenson, W. P. J.; Wideman, A.; Shortt, B. J.; Cheeseright, T.; Crawford, M. J. WO 2009023721, 2009[Chem. Abstr. 2009, 150, 230986].
[7] Dolman, H.; Kuipers, J. EP 234622, 1987[Chem. Abstr. 1988, 109, 110245].
[8] Kim, D. S.; Chun, S. J.; Jeon, J. J.; Lee, S. W.; Joe, G. H. Pest Manage. Sci. 2004, 60, 1007.
[9] Phillion, D.; Wong, S. C.; Shortt, B. US 5486621, 1996[Chem. Abstr. 1996, 124, 253325].
[10] Yoshikawa, Y.; Tomitani, K.; Katsuta, H.; Kawashima, H.; Takahashi, T.; Inami, S.; Yanase, Y.; Takashi, A.; Shimotori, H.; Tomura, N. JP 09301974, 1997[Chem. Abstr. 1997, 128, 22908].
[11] Xie, G. Y.; Jin, W. B.; Zhao, Y. Q.; Cheng, Y. N.; Sun, B. J.; Sun, S. J.; Wang, M. Z.; Wei D. D.; Li, H. L. Chin. J. Org. Chem. 2014, 34, 1124 (in Chinese). (谢桂英, 靳文波, 赵艳芹, 程绎南, 孙炳剑, 孙淑君, 汪梅子, 位丹丹, 李洪连, 有机化学, 2014, 34, 1124.)
[12] Jin, W. B.; Xie, G. Y.; Sun, S. J.; Zhao, Y. Q.; Cheng, Y. N.; Sun, B. J.; Li, H. L. Chin. J. Org. Chem. 2014, 34, 2376 (in Chinese). (靳文波, 谢桂英, 孙淑君, 赵艳芹, 程绎南, 孙炳剑, 李洪连, 有机化学, 2014, 34, 2376.)
[13] Fevig, T. L.; Pillips, W. G.; Lau, P. H. J. Org. Chem. 2001, 66, 2493.
[14] Bures, E.; Spinazze, P. G.; Beese, G.; Hunt, I. R.; Rogers, C.; Keay, B. A. J. Org. Chem. 1997, 62, 8741.
[15] Lu, B.; Falck, J. R. Angew. Chem., Int. Ed. 2008, 47, 7508.
[16] Kuznetsov, A.; Onishi, Y.; Inamoto, Y.; Gevorgyan, V. Org. Lett. 2013, 15, 2498.
[17] Ihara, H.; Koyanagi, M.; Suginome, M. Org. Lett., 2011, 13, 2662.
[18] Cao, H.; Zhan, H. Y.; Wu, J. Y.; Zhong, H. P.; Lin, Y. G.; Zhang, H. Eur. J. Org. Chem. 2012, 2318.
[19] Cao, H.; Jiang, H. F.; Zhou, X. S.; Qi, C. R.; Lin, Y. G.; Wu, J. Y.; Liang, Q. M. Green Chem. 2012, 14, 2710.
[20] Baharfar, R.; Baghbaniana, S. M.; Hossein, N. R.; Bijanzadeh, H. R. Lett. Org. Chem. 2007, 4, 567.
[21] Reddy, L. R.; Saravanan, P.; Corey, E. J. J. Am. Chem. Soc. 2004, 126, 6230.
[22] Downey, C. W.; Craciun, S.; Neferu, A. M.; Vivelo, C. A.; Mueller, C. J.; Southall, B. C.; Corsi, S.; Etchill, E. W.; Sault, R. J. Tetrahedron Lett. 2012, 53, 5763
[23] Bohlmann, F.; Bresinsky, E. Chem. Ber. 1964, 97, 2109.
[24] Sun, B. J.; Yuan, H. X.; Xing, X. P.; Li, H. L. J. Triticeae Crops 2008, 28, 709 (in Chinese). (孙炳剑, 袁虹霞, 邢小萍, 李洪连, 麦类作物学报, 2008, 28, 709.)
/
| 〈 |
|
〉 |