

Chinese Journal of Organic Chemistry >
Synthesis of Wogonin, a Natural Product with Anticancer Activity
Received date: 2016-04-27
Revised date: 2016-06-21
Online published: 2016-08-10
Supported by
Project supported by the Professor of Chang Jiang Scholars Program, the National Natural Science Foundation of China (Nos. 81520108030, 81573318, 81373301, 1302658), the Shanghai Leading Academic Discipline Project (No. B906), the Shanghai Engineering Research Center for the Preparation of Bioactive Natural Products (No. 10DZ2251300).
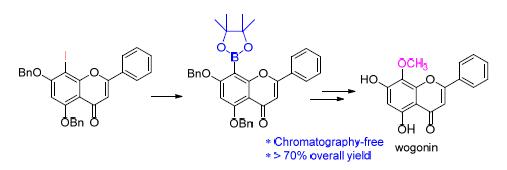
Wogonin (5,7-dihydroxy-8-methoxyflavone) is an active component isolated from the Traditional Chinese Medicine (TCM) Huang Qin, which exhibits significant anti-tumor activity via a unique mechanism. The reported chemical synthesis of wogonin suffered either from low overall yield, harsh reaction conditions or from noncommercially available starting materials. In this paper, a new synthesis of wogonin was achieved via Claisen-Schmidt condensation, cyclization, iodination at C-8 position of flavone, borylation of sterically hindered aryl iodides, oxidation of boronic esters, methylation and debenzylation reaction using 2,4-dibenzyloxy-6-hydroxy phenylacetone and benzaldehyde as the starting materials, which provided gram-scale wogonin in >70% overall yield. The total synthesis employs mild reaction conditions and conventional flash column chromatography is not needed for purification, which makes it suitable for large-scale production. The efficient and flexible entry should offer opportunities for further modification and chemical biology investigations.
Key words: anticancer; wogonin; borylation; synthesis
Yuan Hu , Ye Ji , Chen Hao , Zhao Zeng , Li Yuzhu , Zhang Weidong , Sun Qingyan . Synthesis of Wogonin, a Natural Product with Anticancer Activity[J]. Chinese Journal of Organic Chemistry, 2016 , 36(12) : 2960 -2964 . DOI: 10.6023/cjoc201604057
[1] (a) Chirumbolo, S. Toxicology 2013, 314, 60.
(b) Yang, L.; You, Q. D.; Yang, Y.; Guo, Q. L. J. China Pharm. Univ. 2009, 40, 576(in Chinese).(杨莉, 尤启冬, 杨勇, 郭青龙, 中国药科大学学报, 2009, 40, 576.)
(c) Ren, X. D.; Fu, W.; Zhang, X. Y.; Hu, P.; Li, Z. Y. Chin. New Drugs J. 2011, 20, 777(in Chinese).(任晓东, 符伟, 张晓芸, 胡珀, 汪嵘, 李志裕, 中国新药杂志, 2011, 20, 777.)
[2] Shah, R. C.; Mehta, C. R.; Wheeler, T. S. J. Chem. Soc. 1938, 1555.
[3] Rivailie, P.; Mentzer, C. C. R. Acad. Sci., Ser. Ⅱc:Chim. 1969, 268, 2213.
[4] Löwe, W.; Maske, P. V.; Müller, W. Arch. Pharm. 1994, 327, 255.
[5] (a) Kampouris, E. M. J. Chem. Soc. C 1968, 2125.
(b) Tisdale, E. J.; Vong, B. G.; Li, H. M.; Kim, S. H.; Chowdhury, C.; Theodorakis, E. A. Tetrahedron 2003, 59, 6873.
[6] Rao, K. V.; Rao, K. V.; Seshadri, T. R. Proc. Ind. Acad. Sci. A 1947, 26, 13.
[7] Huang, W. H.; Chien, P. Y.; Yang, C. H.; Lee, A. R. Chem. Pharm. Bull. 2003, 51, 339.
[8] Li, Z. Y.; Cao, X.; Wang, X.; Guo, Q. L.; You, Q. D. Org. Prep. Proced. Int. 2009, 41, 327.
[9] Sharma, P. K.; He, M.; Jurayj, J.; Gou, D. M.; Lombardy, R.; Romanczyk, L. J., Jr.; Schroeter, H. Molecules 2010, 15, 5595.
[10] Joncour, A.; Decor, A.; Thoret, S.; Chiaroni, A.; Baudoin, O. Angew. Chem., Int. Ed. 2006, 45, 4149.
[11] Ishiyama, T.; Murata, M.; Miyaura, N. J. Org. Chem. 1995, 60, 7508.
[12] Li, B.; Li, J.; Fu, Y.; Bo, Z. J. Am. Chem. Soc. 2004, 126, 3430.
[13] Murata, M.; Oyama, T.; Watanabe, S.; Masuda, Y. J. Org. Chem. 2000, 65, 164.
[14] Takagi, A.; Ikawa, T.; Kurita, Y.; Saito, K.; Azechi, K.; Egi, M.; Itoh, Y.; Tokiwa, H.; Kita, Y.; Akai, S. Tetrahedron 2013, 69, 4338.
[15] Yaglioglu, A. S.; Demirtas, I.; Goren, N. Phytochem. Lett. 2014, 8, 213.
[16] Drewes, S. E.; van Vuuren, S. F. Phytochemistry 2008, 69, 1745.
[17] (a) Kerr, P. J.; Pyke, S. M.; Ward, A. D. Aust. J. Chem. 2008, 61, 350.
(b) Caldwell, S. T.; Petersson, H. M.; Farrugia, L. J.; Mullen, W.; Crozierb, A.; Hartley, R. C. Tetrahedron 2006, 62, 7257.
/
| 〈 |
|
〉 |