

Chinese Journal of Organic Chemistry >
Review of Total Synthesis of Valienamine
Received date: 2016-05-11
Revised date: 2016-07-08
Online published: 2016-08-10
Supported by
Project supported by the National Science Foundation of China (Nos. 21376213, 21476194), the Doctoral Program of Higher Education of China (No. 20120101110062), and the Postdoctoral Advanced Programs of Zhejiang Province (No. BSH1502096).
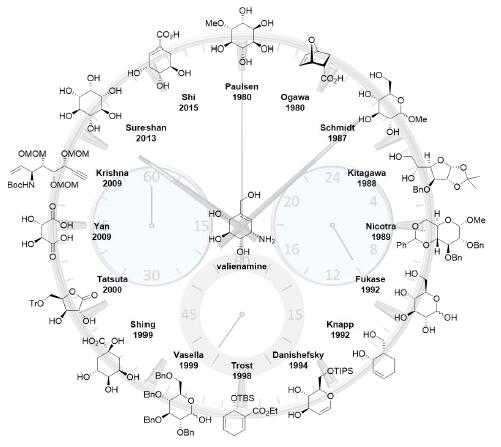
Valienamine is an α-glucosidase inhibitor belonging to C7N aminocyclitols that was isolated from the microbial degradation of validoxylamine A with Pseudomonas denitrificans. Moreover, valienamine is also an essential core unit in many kinds of pseudo-oligosaccharidic α-D-glucosidase inhibitors such as validamycins and acarbose. Due to the unique structural feature and interesting biological property, since the first isolation in 1972, a great deal of effort has been devoted to the development of various approaches for the efficient synthesis of valienamine. This review summarizes the 23 synthetic approaches towards valienamine reported in the last thirty years.
Ji Li , Wu Guofeng , Ye Weidong , Chen Xinzhi . Review of Total Synthesis of Valienamine[J]. Chinese Journal of Organic Chemistry, 2016 , 36(12) : 2771 -2785 . DOI: 10.6023/cjoc201605015
[1] Delgado, A. Eur. J. Org. Chem. 2008, 3893.
[2] Diaz, L.; Delgado, A. Curr. Med. Chem. 2010, 17, 2393.
[3] Arjona, O.; Gómez, A. M.; López, J. C.; Plumet, J. Chem. Rev. 2007, 107, 1919.
[4] Mahmud, T. Nat. Prod. Rep. 2003, 20, 137.
[5] Kameda, Y.; Horii, S. J. Chem. Soc., Chem. Commun. 1972, 1972, 746.
[6] Kamiya, K.; Wada, Y.; Horii, S.; Nishikawa, M. J. Antibiot. 1971, 24, 317.
[7] Fukase, H.; Horii, S. J. Org. Chem. 1992, 57, 3651.
[8] Chen, X. L.; Zheng, Y. G.; Shen, Y. C. Curr. Med. Chem. 2006, 13, 109.
[9] Chen, X. L.; Fan, Y. X.; Zheng, Y. G.; Shen, Y. C. Chem. Rev. 2003, 103, 1955.
[10] Fukuhara, K.; Murai, H.; Murao, S. Agric. Biol. Chem. 1982, 46, 2021.
[11] Truscheit, E.; Frommer, W.; Junge, B.; Müller, L.; Schmidt, D. D.; Wingender, W. Angew. Chem., Int. Ed. Engl. 1981, 20, 744.
[12] Kameda, Y.; Asano, N.; Yoshikawa, M.; Matsui, K. J. Antibiot. 1980, 33, 1575.
[13] Kameda, Y.; Asano, N.; Yoshikawa, M.; Takeuchi, M.; Yama-guchi, T.; Matsui, K.; Horii, S.; Fukase, H. J. Antibiot. 1984, 37, 1301.
[14] Horii, S.; Fukase, H.; Kameda, Y. Carbohydr. Res. 1985, 140, 185.
[15] Ji, L.; Zhang, D.-F.; Zhao, Q.; Hu, S.-M.; Qian, C.; Chen, X.-Z. Tetrahedron 2013, 69, 7031.
[16] Tanaka, K. S.; Winters, G. C.; Batchelor, R. J.; Einstein, F. W.; Bennet, A. J. J. Am. Chem. Soc. 2001, 123, 998.
[17] Wang, Y.; Bennet, A. J. Org. Biomol. Chem. 2007, 5, 1731.
[18] Horii, S.; Iwasa, T.; Kameda, Y. J. Antibiot. 1971, 24, 57.
[19] Berecibar, A.; Grandjean, C.; Siriwardena, A. Chem. Rev. 1999, 99, 779.
[20] Ji, L.; Zhou, G.-Q.; Qian, C.; Chen, X.-Z. Eur. J. Org. Chem. 2014, 3622.
[21] Paulsen, H.; Heiker, F. R. Angew. Chem., Int. Ed. Engl. 1980, 19, 904.
[22] Knapp, S.; Naughton, A. B. J.; Dhar, T. G. M. Tetrahedron Lett. 1992, 33, 1025.
[23] Shing, T. K.; Li, T. Y.; Kok, S. H.-L. J. Org. Chem. 1999, 64, 1941.
[24] Kok, S. H.-L.; Lee, C. C.; Shing, T. K. M. J. Org. Chem. 2001, 66, 7184.
[25] Mondal, S.; Prathap, A.; Sureshan, K. M. J. Org. Chem. 2013, 78, 7690.
[26] Nie, L.-D.; Shi, X.-X.; Ko, K. H.; Lu, W.-D. J. Org. Chem. 2009, 74, 3970.
[27] Quan, N.; Nie, L.; Shi, X.; Zhu, R.; Lü, X. Chin. J. Chem. 2012, 30, 2759.
[28] Quan, N.; Nie, L. D.; Zhu, R. H.; Shi, X. X.; Ding, W.; Lu, X. Eur. J. Org. Chem. 2013, 6389.
[29] Ding, W.; Yu, J. P.; Shi, X. X.; Nie, L. D.; Quan, N.; Li, F. L. Tetrahedron:Asymmetry 2015, 26, 1037.
[30] Ogawa, S.; Toyokuni, T.; Suami, T. Chem. Lett. 1980, 9, 713.
[31] Toyokuni, T.; Ogawa, S.; Suami, T. Bull. Chem. Soc. Jpn. 1983, 56, 1161.
[32] Ogawa, S.; Shibata, Y.; Nose, T.; Suami, T. Bull. Chem. Soc. Jpn. 1985, 58, 3387.
[33] Trost, B. M.; Chupak, L. S.; Lubbers, T. J. Am. Chem. Soc. 1998, 120, 1732.
[34] Trost, B. M. Chem. Pharm. Bull. 2002, 50, 1.
[35] Schmidt, R. R.; Köhn, A. Angew. Chem., Int. Ed. Engl. 1987, 26, 482.
[36] Nicotra, F.; Panza, L.; Ronchetti, F.; Russo, G. Gazz. Chim. Ital. 1989, 119, 577.
[37] Park, T. K.; Danishefsky, S. J. Tetrahedron Lett. 1994, 35, 2667.
[38] Chang, Y.-K.; Lo, H.-J.; Yan, T.-H. Org. Lett. 2009, 11, 4278.
[39] Yoshikawa, M.; Cha, B. C.; Nakae, T.; Kitagawa, I. Chem. Pharm. Bull. 1988, 36, 3714.
[40] Yoshikawa, M.; Cha, B. C.; Okaichi, Y.; Takinami, Y.; Yokokawa, Y.; Kitagawa, I. Chem. Pharm. Bull. 1988, 36, 4236.
[41] Shing, T. K.; Chen, Y.; Ng, W. Synlett 2011, 1318.
[42] Shing, T. K. M.; Chen, Y.; Ng, W.-L. Tetrahedron 2011, 67, 6001.
[43] Tatsuta, K.; Mukai, H.; Takahashi, M. J. Antibiot. 2000, 53, 430.
[44] Zhou, B.; Luo, Z.; Lin, S.; Li, Y. C. Synlett 2012, 913.
[45] Plumet, J.; Gomez, A. M.; Lopez, J. C. Mini-Rev. Org. Chem. 2007, 4, 201.
[46] Kapferer, P.; Sarabia, F.; Vasella, A. Helv. Chim. Acta 1999, 82, 645.
[47] Chang, Y.-K.; Lee, B.-Y.; Kim, D. J.; Lee, G. S.; Jeon, H. B.; Kim, K. S. J. Org. Chem. 2005, 70, 3299.
[48] Cumpstey, I. Tetrahedron Lett. 2005, 46, 6257.
[49] Cumpstey, I.; Gehrke, S.; Erfan, S.; Cribiu, R. Carbohydr. Res. 2008, 343, 1675.
[50] Li, Q. R.; Kim, S. I.; Park, S. J.; Yang, H. R.; Baek, A. R.; Kim, I. S.; Jung, Y. H. Tetrahedron 2013, 69, 10384.
[51] Lo, H.-J.; Chen, C.-Y.; Zheng, W.-L.; Yeh, S.-M.; Yan, T.-H. Eur. J. Org. Chem. 2012, 2780.
[52] Krishna, P. R.; Reddy, P. S. Synlett 2009, 209.
/
| 〈 |
|
〉 |