

Chinese Journal of Organic Chemistry >
Visible Light Promoted C-H Functionalization Reactions of Tertiary Amines
Received date: 2016-07-27
Revised date: 2016-09-01
Online published: 2016-10-09
Supported by
Project supported by the National Natural Science Foundation of China(No. 31402109).
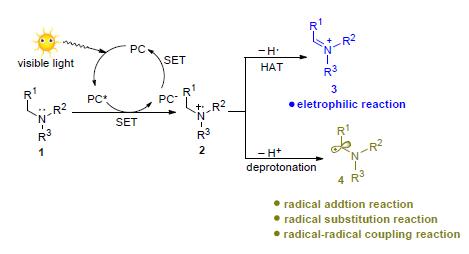
The tertiary amine motif is an important structure component of multitudinous natural products and drug molecules.Visible light promoted C-H functionalization of tertiary amines has recently received much attention and facilitated the synthesis of alkaloids and drug molecules.Based on photoredox catalysis,visible light is able to induce the formation of iminium cations or α-amino carbon radicals through single electron transfer process which can participate in multitudinous type of organic reactions and complete C-H functionalization of tertiary amines.In this review,the research progress of photoredox catalytic C-H func-tionalization was categorized and summarized.
Liu Wei , Zheng Xinyu , Zeng Jianguo , Cheng Pi . Visible Light Promoted C-H Functionalization Reactions of Tertiary Amines[J]. Chinese Journal of Organic Chemistry, 2017 , 37(1) : 1 -19 . DOI: 10.6023/cjoc201607040
[1] (a) Chen, J.-R.; Hu, X.-Q.; Lu, L.-Q.; Xiao, W.-J. Chem. Soc. Rev. 2016, 45, 2044.
(b) Morris, S. A.; Wang, J.; Zhang, N. Acc. Chem. Res. 2016, 49, 1957.
(c) Wei, G.; Basheer, C.; Tan, C.-H.; Jiang, Z. Tetrahedron Lett. 2016, 57, 3801.
(d) Beatty, J. W.; Stephenson, C. R. J. Acc. Chem. Res. 2015, 48, 1474.
(e) Nicewicz, D. A.; Nguyen, T. M. ACS Catal. 2014, 4, 355.
(f) Hari, D. P.; Konig, B. Chem. Commun. 2014, 50, 6688.
(g) Douglas, J. J.; Nguyen, J. D.; Cole, K. P.; Stephenson C. R. J. Aldrichim. Acta 2014, 47, 15.
(h) Prier, C. K.; Rankic, D. A.; McMillan, D. W. C. Chem. Rev. 2013, 113, 5322.
(i) Xuan, J.; Xiao, W.-J. Angew. Chem., Int. Ed. 2012, 51, 6828.
(j) Zuo, X.; Wu, W.; Su, W. Acta Chim. Sinica 2015, 73, 1298(in Chinese).(左璇, 吴文亮, 苏伟平, 化学学报, 2015, 73, 1298.)
(k) Sun, X.; Yu, S. Chin. J. Org. Chem. 2016, 36, 239(in Chinese).(孙晓阳, 俞寿云, 有机化学, 2016, 36, 239.)
(l) Tan, R.; Xiao. W. Acta Chim. Sinica 2015, 73, 85(in Chinese).(谭芬, 肖文精, 化学学报, 2015, 73, 85.)
[2] (a) Zenk, M. H. Pure Appl. Chem. 1994, 66, 2023.
(b) Hagel, J. M.; Facchini, P. J. Plant Cell Physiol. 2013, 54,647.
[3] (a) Hedstrand, D. M.; Kruizinga, W. M.; Kellogg, R. M. Tetrahedron Lett. 1978, 19, 1255.
(b) van Bergen, T. J.; Hedstrand, D. M.; Kruizinga, W. H.; Kellogg, R. M. J. Org. Chem. 1979, 44, 4953.
[4] (a) Tang, Q.; Liu, X.; Liu, S.; Xie, H.; Liu, W.; Zeng, J.; Cheng, P. RSC Adv. 2015, 5, 89009.
(b) Schnermann, M. J.; Overman, L. E. Angew. Chem., Int. Ed. 2012, 51, 9576.
(c) Pratsch, G.; Lackner, G. L.; Overman, L. E. J. Org. Chem. 2015, 82, 6025.
(d) Tucker, J. W.; Nguyen, J. D.; Narayanam, J. M. R.; Krabbe, S. W.; Stephenson, C. R. J. Chem. Commun. 2010, 46, 4985.
[5] Freeman, D. B.; Furst, L.; Condie, A. G.; Stephenson, C. R. J. Org. Lett. 2012, 14, 94.
[6] Condie, A. G.; Gonzalez-Gomez, J. C.; Stephenson, C. R. J. J. Am. Chem. Soc. 2010, 132, 1464.
[7] Rueping, M.; Zhua, S.; Koenigs, R. Chem. Commun. 2011, 47, 12709.
[8] Rueping, M.; Koenigs, R. M.; Poscharny, K.; Fabry, D. C.; Leonori, D.; Vila, C. Chem.-Eur. J. 2012, 18, 5170.
[9] Rueping, M.; Zhu, S.; Koenigs, R. M. Chem. Commun. 2011, 47, 8679.
[10] Rueping, M.; Vila, C.; Bootwicha, T. ACS Catal. 2013, 3, 1676.
[11] Pan, Y.; Wang, S.; Kee, C. W.; Dubuisson, E.; Yang, Y.; Loh, K. P.; Tan, C.-H. Green Chem. 2011, 13, 3341.
[12] Feng, Z.-J.; Xuan, J.; Xia, X.-D.; Ding, W.; Guo, W.; Chen, J.-R.; Zou, Y.-Q.; Lu, L.-Q.; Xiao, W.-J. Org. Biomol. Chem. 2014, 12, 2037.
[13] Xiao, T.; Li, L.; Lin, G.; Mao, Z.-W.; Zhou, L. Org. Lett. 2014, 16, 4232.
[14] DiRocco, D. A.; Rovis, T. J. Am. Chem. Soc. 2012, 134, 8094.
[15] Bergonzini, G.; Schindler, C. S.; Wallentin C.-J.; Jacobsen, E. N.; Stephenson, C. R. J. Chem. Sci. 2014, 5, 112.
[16] Cai, S.; Zhao, X.; Wang, X.; Liu, Q.; Li, Z.; Wang, D. Z. Angew. Chem., Int. Ed. 2012, 51, 8050.
[17] Zhao, Y.; Cai, S.; Li, J.; Wang, D. Z. Tetrahedron 2013, 69, 8129.
[18] Li, X.; Gu, X.; Li, Y.; Li, P. ACS Catal. 2014, 4, 1897.
[19] Beatty, J. W.; Stephenson, C. R. J. J. Am. Chem. Soc. 2014, 136, 10270.
[20] Xuan, J.; Cheng, Y.; An, J.; Lu, L.-Q.; Zhang, X.-X.; Xiao, W.-J. Chem. Commun. 2011, 47, 8337.
[21] Orgren, L. R.; Maverick, E. E.; Marvin, C. J. Org. Chem. 2015, 80, 12635.
[22] Bertrand, S.; Hoffmann, N.; Pete, J.-P. Eur. J. Org. Chem. 2000, 2227.
[23] Miyake, Y.; Nakajima, K.; Nishibayashi, Y. J. Am. Chem. Soc. 2012, 134, 3338.
[24] Zhou, H.; Lu, P.; Gu, X.; Li, P. Org. Lett. 2013, 15, 5646.
[25] Dai, X.; Cheng, D.; Guan, B.; Mao, W.; Xu. X.; Li, X. J. Org. Chem. 2014, 79, 7212.
[26] Dai, X.; Mao, R.; Guan, B.; Xu, X.; Li, X. RSC Adv. 2015, 5, 55290.
[27] Noble, A.; MacMillan, D. W. C. J. Am. Chem. Soc. 2014, 136, 11602.
[28] Kohls, P.; Jadhav, D.; Pandey, G.; Reiser, O. Org. Lett. 2012, 14, 672.
[29] Liu, X.; Ye, X.; Bures, F.; Liu, H.; Jiang, Z. Angew. Chem., Int. Ed. 2015, 54, 11443.
[30] Zhu, S.; Das, A.; Bui, L.; Zhou, H.; Curran, D. P.; Rueping, M. J. Am. Chem. Soc. 2013, 135, 1823.
[31] Zhang, P.; Xiao, T.; Xiong, S.; Dong, X. Zhou, L. Org. Lett. 2014, 16, 3267.
[32] Singh, A.; Arora, A.; Weaver, J. D. Org. Lett. 2013, 15, 5390.
[33] Prier, C. K.; MacMillan, D. W. C. Chem. Sci. 2014, 5, 4173.
[34] Douglas, J. J.; Cole, K. P.; Stephenson, C. R. J. J. Org. Chem. 2014, 79, 11631.
[35] Terrett, J. A.; Clift, M. D.; MacMillan, D. W. C. J. Am. Chem. Soc. 2014, 136, 6858.
[36] Zou, Y.-Q.; Chen, J.-R.; Xiao, W.-J. Angew. Chem., Int. Ed. 2013, 52, 11701.
[37] Pirnot, M. T.; Rankic, D. A.; Martin, D. B. C.; MacMillan, D. W. C. Science 2013, 339, 1593.
[38] Petronijevic, F. R.; Nappi, M.; MacMillan, D. W. C. J. Am. Chem. Soc. 2013, 135, 18323.
[39] Jeffrey, J. L.; Petronijevic, F. R. MacMillan, D. W. C. J. Am. Chem. Soc. 2015, 137, 8404.
[40] Fava, E.; Millet, A.; Nakajima, M.; Loescher, S.; Rueping, M. Angew. Chem., Int. Ed. 2016, 55, 6776.
[41] Ding, W.; Lu, L.-Q.; Liu, J.; Liu, D.; Song, H.-T.; Xiao, W.-J. J. Org. Chem. 2016, 81, 7237.
[42] Skubi, K. L.; Blum, T. R.; Yoon, T. P. Chem. Rev. 2016, 116, 10035.
[43] Xuan, J.; Zeng, T.-T.; Feng, Z.-J.; Deng, Q.-H.; Chen, J.-R.; Lu, L.-Q.; Xiao, W.-J. Angew. Chem. Int. Ed. 2015, 54, 1625.
[44] Feng, Z.; Zeng, T.; Xuan, J.; Liu, Y.; Lu, L.; Xiao, W.-J. Sci. China Chem. 2016, 59, 171.
[45] Liu, Z.; Huang, Y.; Xie, H.; Liu, W.; Zeng, J.; Cheng. P. RSC. Adv. 2016, 6, 50500.
[46] Uraguchi, D.; Kinoshita, N.; Kizu, T.; Ooi, T. J. Am. Chem. Soc. 2015, 137, 13768.
[47] Kizu, T.; Uraguchi, D.; Ooi, T. J. Org. Chem. 2016, 81, 6953.
[48] Wang, C.; Qin, J.; Shen, X.; Riedel, R.; Harms, K.; Meggers, E. Angew. Chem., Int. Ed. 2016, 55, 685.
[49] Amador, A. G.; Yoon, T. P. Angew. Chem., Int. Ed. 2016, 55, 2304.
[50] (a) Zhao, L.; Li, C.-J. Angew. Chem., Int. Ed. 2008, 47, 7075.
(b) Wu, J.-C.; Song, R.-J.; Wang, Z.-G.; Huang, X.-C.; Xie, Y.-X.; Li, J.-H. Angew. Chem., Int. Ed. 2012, 51, 3453.
(c) Huo, C.; Yuan, Y.; Wu, M.; Jia, X.; Wang, X.; Chen, F.; Tang, J. Angew. Chem., Int. Ed. 2014, 53, 13544.
(d) Peng, H.; Yu, J.-T.; Jiang, Y.; Yang, H.; Cheng, J. J. Org. Chem. 2014, 79, 9847.
(e) Zhu, Z.-Q.; Bai, P.; Huang, Z.-Z. Org. Lett. 2014, 16, 4881.
[51] Liu, W.; Liu, S.; Xie, H.; Qing, Z.; Zeng, J. Cheng, P. RSC Adv. 2015, 5, 17383.
[52] Zhu, S.; Rueping, M.; Chem. Commun. 2012, 48, 11960.
[53] Zou, Y.-Q.; Lu, L.-Q.; Fu, L.; Chang, N.-J.; Rong, J.; Chen, J.-R. Xiao, W.-J. Angew. Chem., Int. Ed. 2011, 50, 7171.
/
| 〈 |
|
〉 |