

Chinese Journal of Organic Chemistry >
Efficient Hydrolysis of Haloalkynes to α-Haloketones in Ionic Liquid
Received date: 2016-10-18
Revised date: 2016-12-30
Online published: 2017-01-20
Supported by
Project supported by the Planned Science and Technology Project of Hunan Province (No. 2015WK3003), the Scientific Research Fund of Hunan Provincial Education Department (No. 14C0405).
The haloalkynes were hydrolyzed to α-haloketones by sulfuric acid promotion in ionic liquids (ILs) with 85%~94% yields. The ILs-H2SO4 reaction system could be easily recycled (more five times) without any effect for reaction yield. At the same time, a wide scope of substrates haloalkynes were proper in this reaction system and a series of α-chloro/bromo/iodo acetophenones with different substituent (such as methyl, methoxyl, hydroxyl, nitro etc.) and aliphatic α-haloketones have been obtained in good yields.
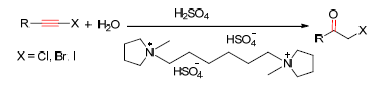
Key words: ionic liquid; haloalkynes; hydration; α-haloketones; recyclable
Fu Wenqiang , Tan Ping , Deng Wei , Xiang Jiannan . Efficient Hydrolysis of Haloalkynes to α-Haloketones in Ionic Liquid[J]. Chinese Journal of Organic Chemistry, 2017 , 37(6) : 1501 -1505 . DOI: 10.6023/cjoc201610031
[1] (a) Erian, A. W.; Sherif, S. M.; Gaber, H. M. Molecules 2003, 8, 793.
(b) Zhang, H.; Wei, Q.; Wei, S.; Qu, J.; Wang, B. Eur. J. Org. Chem. 2016, 3373.
[2] De Kimpe, N.; Verhé, R. The Chemistry of α-Haloketones, α-Haloaldehydes and α-Haloimines, Ed.: Patai, S., Wiley, Chichester, UK, 1988, pp. 1~119.
[3] (a) Ostrowski, T.; Golankiewicz, B.; De Clercq, E.; Andrei, G.; Snoeck, R. Eur. J. Med. Chem. 2009, 44,3313.
(b) Conde, S.; Pérez, D. I.; Martínez, A.; Perez, C.; Moreno, F. J. J. Med. Chem. 2003, 46, 4631.
[4] (a) Morton, H. E.; Leanna, M. R. Tetrahedron Lett. 1993, 34, 4481.
(b) Patil, R. D.; Joshi, G.; Adimurthy, S.; Ranu, B. C. Tetrahedron Lett. 2009, 50, 2529.
(c) Tanemura, K.; Suzuki, T.; Nishida, Y.; Satsumabayashi, K.; Horaguchi, T. Chem. Commun. 2004, 470.
(d) Meshram, H. M.; Reddy, P. N.; Vishnu, P.; Sadashiv, K.; Yadav, J. S. Tetrahedron Lett. 2006, 47, 991.
(e) Pravst, I.; Zupan, M.; Stavber, S. Tetrahedron 2008, 64, 5191.
(f) Kosower, E. M.; Wu, G. S. J. Org. Chem. 1963, 28, 633.
(g) Kajigaeshi, S.; Kakinami, T.; Moriwaki, M.; Fujisaki, S.; Maeno, K.; Okamoto, T. Synthesis 1988, 545.
(h) Dieter, R. K.; Nice, L. E.; Velu, S. E. Tetrahedron Lett. 1996, 37, 2377.
[5] Xie, L.; Wu, Y.; Yi, W.; Zhu, L.; Xiang, J.; He, W. J. Org. Chem. 2013, 78, 9190.
[6] Zou, H.; He, W.; Dong, Q.; Wang, R.; Yi, N.; Jiang, J.; Peng, D.; He, W. Eur. J. Org.Chem. 2016, 116.
[7] (a) Chen, Z.; Ye, D.; Ye, M.; Zhou, Z.; Li, S.; Liu, L. Tetrahedron Lett. 2014, 55, 1373.
(b) Zeng, M.; Huang, R.; Li, W.; Liu, X.; He, F.; Zhang, Y.; Xiao, F. Tetrahedron Lett. 2016, 72, 3818.
[8] (a) Olivier-Bourbigou, H.; Magna, L.; Morvan, D. Appl. Catal. A: Gen. 2010, 373, 1.
(b) Hu, Y.; Guo, Z.; Lue, B. M.; Xu, X. J. Agric. Food Chem. 2009, 57, 3845.
(c) Tzani, A.; Douka, A.; Papadopoulos, A.; Pavlatou, E. A.; Voutsas, E.; Detsi, A. ACS Sustainable Chem. Eng. 2013, 1, 1180.
[9] Wong, W.; Ho, K.; Lee, L.; Lam, K.; Zhou, Z.; Chan, T.; Wong, K. ACS Catal. 2011, 1, 116.
[10] Wei, Q. L.; Zhang, S. S.; Gao, J.; Li, W. H.; Xu, L. Z.; Yu, Z. G. Bioorg. Med. Chem. 2006, 14, 7146.
[11] Reddy Bodireddy, M.; Gangi, R. N. C. Synth. Commun. 2013, 43, 2603.
[12] Guan, X.; AlMisbaa, Z.; Huang, K. Arabian J. Chem. 2015, 8, 892.
[13] Sugiura, A.; Kepner, R. E.; Webb, A. D. J. Org. Chem. 1962, 27, 773.
[14] Jiang, Q.; Sheng, W.; Guo, C. Green Chem. 2013, 15, 2175
[15] Rosenmund, K. W. Chem. Ber. 1957, 90, 1922.
[16] Lapointe, D.; Markiewicz, T.; Whipp, C. J.; Toderian, A.; Fagnou, K. J. Org. Chem. 2011, 76,749.
[17] Judefind, W. L. J. Am. Chem. Soc. 1920, 42, 1043.
[18] Arrieta, A. Synth. Commun. 1984, 14, 939.
[19] Suzuki, M. Yakugaku Zasshi 1952, 72, 305.
[20] Antunes, H.; Fardelone, L. C.; Rodrigues, J. A. R.; Moran, P. J. S. Tetrahedron: Asymmetry 2004, 15, 2615.
/
| 〈 |
|
〉 |