

Chinese Journal of Organic Chemistry >
Synthesis of Novel Ferrocenyl Thiazole and Imidazole Derivatives and Its Selective Recognition to Fe3+
Received date: 2016-11-25
Revised date: 2017-02-09
Online published: 2017-02-27
Supported by
Project supported by the Beijing Natural Science Foundation (No. 2173060) and the General Plan of Beijing Municipal Education Commission (No. KM201710028007).
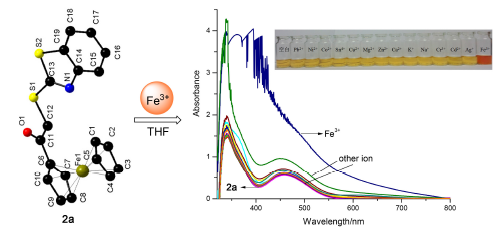
Five novel ferrocenyl thiazole and imidazole derivatives were synthesized. The structure characterization of the five products showed that ferrocene conjugative unit and thiazole or imidazole conjugative unit were linked by carbonyl methylene skeleton (COCH2). Chloroacetylferrocene (1a) was prepared by chloroacetylation of ferrocene which reacted with different heterocyclic compounds (2-mercaptobenzothiazole, 2-mercaptobenzimidazole, imidazole and benzimidazole) to give four novel compounds, respectively. 1,1'-Bis(bromoacetyl)ferrocene (1b) was prepared by acetylation and subsequent bromination of ferrocene, which then reacted with 2-mercaptobenzothiazole to produce 2e. All the new compounds were confirmed by 1H NMR, 13C NMR, ESI-MS, HRMS and IR. Three single crystal structures were obtained. Recognition properties on 14 metal ions of the five new compounds were investigated by UV-vis spectrum. The results showed that the five new compounds possessed recognition properties only to iron(III), in which 2a had maximum spectra response.
Key words: ferrocene; thiazole; imidazole; iron(III) ion recognition; tautomerism
Wu Xiaofang , Wu Pei , Li Jingyu , Lu Jing , Wang Jianchun . Synthesis of Novel Ferrocenyl Thiazole and Imidazole Derivatives and Its Selective Recognition to Fe3+[J]. Chinese Journal of Organic Chemistry, 2017 , 37(6) : 1412 -1416 . DOI: 10.6023/cjoc201611037
[1] Umesh, F.; Sanjay, A.; Anil, K. Chem. Phys. Lett. 2013, 584, 165.
[2] Choi, Y. W.; Park, G. J.; Na, Y. J.; Jo, H. Y.; Lee, S. A. You, G. R.; Kim, C. Sens. Actuators, B 2014, 194, 343.
[3] Xu, D.; Wang, M.-M.; Zeng, R.-J.; He, S.-G.; Zhang, Y.; Wang, H.; Shen, H.-T. Appl. Chem. Ind. 2013, 42, 1712 (in Chinese). (徐栋, 王鸣明, 曾瑞杰, 何森, 张玉, 王辉, 盛鸿婷, 应用化工, 2013, 42, 1712.)
[4] Bhatt, K. D.; Makwana, B. A.; Vyas, D. J.; Mishra, D. R.; Jain, V. K. J. Lumin. 2014, 146, 450.
[5] Zhang, H.; Wu, X.-X.; Wu, F.-Y. Chin. J. Inorg. Chem. 2013, 29, 2394 (in Chinese). (张华, 徐孝武, 吴芳英, 无机化学学报, 2013, 29, 2394.)
[6] Du, B.; Ding, Z.-J.; Guo, L.; Wang, P.-H.; Li, Z.-J.; Yu, J.-H. Spectrosc. Spectral Anal. 2015, 35, 470 (in Chinese). (杜斌, 丁志军, 郭磊, 王普红, 李志军, 余建华, 光谱学与光谱分析, 2015, 35, 470.)
[7] Zhang, S.-L.; Zhao, B.; Ran, L.; Qin, D.-B.; Luo, J.-W. Chem. Reag. 2014, 36, 925 (in Chinese). (张仕禄, 赵波, 冉利, 秦大斌, 罗杰伟, 化学试剂, 2014, 36, 925.)
[8] Su, N.; Yang, M.-P.; Meng, W.-F.; Yang, B.-Q. Chin. J. Org. Chem. 2015, 35, 175 (in Chinese). (苏娜, 杨美盼, 孟文斐, 杨秉勤, 有机化学, 2015, 35, 175.)
[9] Garcia-Beltran, O.; Cassels, B. K.; Perez, C.; Mena, N.; Nunez, M. T.; Martine, N. P.; Pavez, P.; Aliaga, M. E. Sensors 2014, 14, 1358.
[10] Maria, A.; Arturo, E.; Alberto, T.; Pedro, M. Org. Lett. 2011, 13, 2078.
[11] Zapata, F.; Caballero, A.; Espinosa, A.; Tárraga, A.; Molina, P. J. Org. Chem. 2009, 74, 4787.
[12] Caballero, A.; Lloveras, V.; Curiel, D. T.; Tarraga, A.; Espinosa, A.; Garcia, R.; Vidal-Gancedo, J.; Rovira, C.; Wurst, K.; Molina, P. Inorg. Chem. 2007, 46, 825.
[13] Zapata, F.; Caballero, A.; Espinosa, A.; Tarraga, A.; Molina, P. Inorg. Chem. 2009, 48, 11566.
[14] Romero, T.; Caballero, A.; Tárraga, A.; Molina, P. Org. Lett. 2009, 11, 3466.
[15] Zhuo, J.-B.; Yan, X.-Q.; Wang, X.-X.; Xie, L.-L.; Yuan, Y.-F. Chin. J. Org. Chem. 2015, 35, 1090 (in Chinese). (卓继斌, 晏希泉, 王小雪, 谢莉莉, 袁耀锋, 有机化学, 2015, 35, 1090.)
[16] Zhuo, J.-B., Wan, Q., Yan, X.-Q., Xie, L.-L., Yuan, Y.-F. Chem. J. Chin. Univ. 2015, 36, 477 (in Chinese). (卓继斌, 万 倩, 晏希泉, 谢莉莉, 袁耀锋, 高等学校化学学报, 2015, 36, 477.)
[17] Molina, P.; Tarraga, A.; Curiel, D.; Velasco, M. D. J. Organomet. Chem. 2001, 637~639, 258.
[18] Ferreira, A. P.; Ferreira da Silva, J. L.; Duarte, M. T.; Minas da Piedade, M. F.; Robalo, M. P.; Harjivan, S. G.; Marzano, C.; Gandin, V.; Marques, M. M. Organometallics 2009, 28, 5412.
[19] Tárraga, A.; Molina, P.; Curiel, D.; Velasco, M. D. Organometallics 2001, 20, 2145.
/
| 〈 |
|
〉 |