

Chinese Journal of Organic Chemistry >
Synthesis and Antitumor Activity of Heterozygous Isatin-Quinazoline Compounds
Received date: 2017-01-18
Revised date: 2017-02-20
Online published: 2017-03-08
Supported by
Project supported by the National Natural Science Foundation of China (No.21272144) s
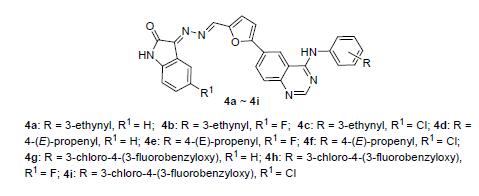
Nine new heterozygous isatin-quinazoline compounds were synthesized from cheap and easily available ortho nitrobenzaldehyde as the starting material. The chemical structures of the synthesized compounds were characterized by NMR, IR and HRMS. The structure of (E)-3-(((E)-(5-(4-((3-ethynylphenyl)amino)quinazolin-6-yl)furan-2-yl)methylene)hydrazono)-indolin-2-one (4a) was further determined by crystallization and X-ray diffraction, and the data revealed that its cis-trans isomerism was (E, E). The antitumor activity of these new compounds was evaluated in vitro by methyl thiazolyl tetrazolium (MTT) assay in human colorectal carcinoma cells SW480, human non-small cell lung cancer cells A549 and NCI- H1975, and human epidermoid squamous carcinoma cells A431. The preliminary data demonstrated that most of the synthetic compounds had moderate to potent inhibitory activity against these four tumor cell lines. In particular, compound 4a had highly potent inhibitory activity on proliferation of four cell lines, and the activity was more potent than positive lapatinib.
Key words: isatin; quinazoline; synthesis; antitumor activity
Zhang Ying, Lü Mengjiao, Zhang Yaling, Chen Li, Wang Wei, Li Baolin . Synthesis and Antitumor Activity of Heterozygous Isatin-Quinazoline Compounds[J]. Chinese Journal of Organic Chemistry, 2017 , 37(7) : 1787 -1793 . DOI: 10.6023/cjoc201701037
[1] Lipunova, G. N.; Nosova, E. V.; Charushin, V. N.; Chupakhin, O. N. Russ. Chem. Rev. 2016, 85, 759.
[2] Gordana, V.; Jeffrey, C. Oncologist 2003, 8, 531.
[3] Ioannou, N.; Seddon, A. M.; Dalgleish, A.; Mackintosh, D.; Solca, F.; Modjtahedi, H. Int. J. Oncol. 2016, 48, 908.
[4] Ravaud, A.; Hawkins, R.; Gardner, J. P.; Vonder Maase, H.; Zantl, N.; Harper, P.; Rolland, F.; Audhuy, B.; Machiels, J. P.; Petavy, F.; Gore, M.; Schoffski, P.; El-Hariry, I. J. Clin. Oncol. 2008, 26, 2285.
[5] El-Ansary, A. K.; Kamal, A. M.; Abd-Hafiz, A. G. M. Chem. Pharm. Bull. 2016, 64, 1172.
[6] Wang, W.-J.; Li, J; Zhang, Y.-L.; Zhang, X.-Q.; Gu, H.-M.; Li, B.-L. Chem. Bull 2015, 78, 1017(in Chinese). (王维佳, 李江, 张娅玲, 张喜全, 顾红梅, 李宝林, 化学通报, 2015, 78, 1017.)
[7] Nahta, R.; Yuan, L. X. H.; Du, Y.; Esteva, F. J. Mol. Cancer Ther. 2007, 6, 667.
[8] Lee, D.; Long, S. A.; Murray, J. H.; Adams, J. L.; Nuttall, M. E.; Nadeau, D. P.; Kikly, K.; Winkler, J. D.; Sung, C. M.; Ryan, M. D.; Levy, M. A.; Keller, P. M.; DeWolf, W. E. Jr. J. Med. Chem. 2001, 44, 2015.
[9] Chapman, J. G.; Magee, W. P.; Stukenbrok, H. A.; Beckius, G. E.; Milici, A. J.; Ross Tracey, W. Eur. J. Pharmacol. 2002, 456, 59.
[10] Smith, B. D.; Smith, G. L.; Hurria, A.; Hortobagyi, G. N.; Buchholz, T. A. Clin. Oncol. 2009, 27, 2758.
[11] Atkins, M.; Jones, C. A.; Kirkpatrick, P. Nat. Rev. Drug Discovery 2006, 5, 279.
[12] Wang, J.; Zhu, C.-Y.; Guo, J.-Y.; Zhang, Z.-G.; Chen, F.; Zhang, L. Guangdong Chem. Ind. 2015, 42, 106(in Chinese). (王京, 朱春媛, 国佳莹, 张泽国, 陈凡, 张磊, 广东化工, 2015, 42, 106.)
[13] Hu, C.; Solomon, V. R.; Cano, P.; Lee, H. Eur. J. Med. Chem. 2010, 45, 705.
[14] Sella, A.; Yarom, N.; Zisman, A.; Kovel, S. Oncology 2009, 76, 442.
[15] Solomon, V. R.; Hu, C.; Lee, H. Bioorg. Med. Chem. 2009, 17, 7585.
[16] Fares, M.; Eldehna, W. M.; Abou-Seri, S. M.; Abdel-Aziz, H. A.; Aly, M. H.; Tolba, M. F. Arch. Pharm. 2015, 348, 144.
[17] Ji, X.; Wang, W.-W.; Xu, G.-H.; Li, F.; Yao, S.-C. Chin. J. Pharm. 2009, 40, 801(in Chinese). (季兴, 王武伟, 许贯虹, 李飞, 姚社春, 中国医药工业杂志, 2009, 40, 801.)
[18] Liu, J; Zhang, Y.-L.; Zhang, X.-Q.; Gu, H.-M.; Li, B.-L. Chem. Bull. 2016, 79, 360(in Chinese). (刘娟, 张娅玲, 张喜全, 顾红梅, 李宝林, 化学通报, 2016, 79, 360.)
[19] McClure, M. S.; Osterhout, M. H.; Roschangar, F.; Sacchetti, M. J. WO 2002002552, 2002[Chem. Abstr. 2002, 136, 102396].
[20] Hassan, T. A. F. M.; Kadi, A. A.; Abdel-Aziz, H. A. K. US 20120252860, 2012[Chem. Abstr. 2012, 157, 548485].
[21] Dweedar, H. E.; Mahrous, H.; Ibrahim, H. S.; Abdel-Aziz, H. A. Eur. J. Med. Chem. 2014, 78, 275.
[22] Nikalje, A. P.; Ansari, A.; Bari, S.; Ugale, V. Arch. Pharm. 2015, 348, 433.
/
| 〈 |
|
〉 |