

Chinese Journal of Organic Chemistry >
Design, Synthesis and Bioactivity of New Cyclohexanedione Inhibitors
Received date: 2017-06-21
Revised date: 2017-07-10
Online published: 2017-07-18
Supported by
Project supported by the National Natural Science Foundation of China (No. 21472065), the 111 Project (No. B17019) and the Fundamental Research Funds for the Central Universities (No. CCNU15A02013).
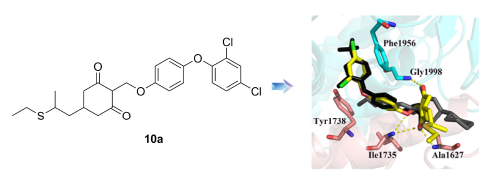
Acetyl-CoA carboxylase (ACCase, EC 6.4.1.2) was one of important herbicidal targets. However, the number of resistant weeds targeting to ACCase had risen to the third place among all of herbicidal targets. Therefore, it is of great importance to design new ACCase inhibitors. Herein, based on the crystal structures, aryloxyphenoxypropanotes (APP)-site and cyclohexanedioneoximes (CHD)-site were used to constructing APP+CHD-site, and then thirteen compounds were designed and synthesized. All the newly prepared compounds were characterized by 1H NMR, 13C NMR and HRMS. The molecular docking showed that 2-((4-(2,4-dichlorophenoxy)phenoxy)methyl)-5-(2-(ethylthio)propyl)cyclohexane-1,3-dione (10a) could occupy not only APP-site, but also CHD-site. In addition, based on the herbicidal activity, some compounds showed some inhibition rate upon tested weeds. Most importantly, 5-(2-(ethylthio)propyl)-2-((4-phenoxyphenoxy)methyl)cyclohexane-1,3-dione (10b) showed a wide range of herbicidal spectrum, which could be used to inhibit not only poaceae weeds, but also broadleaf weeds. All these results indicated that this type of compounds worth of the further investigation.
Key words: ACCase; molecular docking; organic synthesis; herbicidal activity
Li Kejian , Wu Shuangzhi , Wang Nannan , Zhu Xiaolei , Yang Guangfu . Design, Synthesis and Bioactivity of New Cyclohexanedione Inhibitors[J]. Chinese Journal of Organic Chemistry, 2017 , 37(11) : 2978 -2984 . DOI: 10.6023/cjoc201706029
[1] Prado, R.; Gonzdez-Gutierrez, J.; Menendez, J. Weed Sci. 2000, 48, 311.
[2] Burton, J. D.; Gronwald, J. W.; Somers, D. A.; Connelly, J. A.; Gengenbach, B. G.; Wyse, D. L. Biochem. Biophys. Res. Commun. 1987, 148, 1039.
[3] Babczinski, P.; Fischer, R. Pestic. Sci. 1991, 33, 455.
[4] Matthews, N.; Powles, S. B.; Preston, C. Pest Manage. Sci. 2000, 56, 441.
[5] Burton, J. D.; Gronwald, J. W.; Keith, R. A. Pestic. Biochem. Physiol. 1991, 39, 100.
[6] Itomaso, J. M.; Brown, P. H.; Stowe, A. E. Plant Physiol. 1991, 95, 1063.
[7] Nikolau, B. J.; Ohlrogge J. B.; Wurtele, E. S. Arch. Biochem. Biophys. 2003, 414, 211.
[8] Mohamed, I. A.; Li, R. Z.; You, Z. G. Weed Sci. 2012, 60, 167.
[9] Han, J. T.; Wang, J. M.; Dong, H. B.; Xu, Z. H.; Liu, B.; Wang, M. A. Chin. J. Org. Chem. 2013, 33, 596(in Chinese). (韩金涛, 王进敏, 董宏波, 徐志红, 刘斌, 王明安, 有机化学, 2013, 33, 596.)
[10] Xu, Z. H.; Dong, H. B.; Liu, B.; Kong, L. Q.; Wang, M. A. Chin. J. Org. Chem. 2015, 35, 411(in Chinese). (徐志红, 董宏波, 刘斌, 孔令青, 王明安, 有机化学, 2015, 35, 411.)
[11] Sandra, L.; Joachim, J.; Guido, M.; Michael, K.; Thomas, S.; Bernd, W. WO 200610474, 2006[Chem. Abstr. 2006, 103516].
[12] Coret, J. M. US 7329770, 2005[Chem. Abstr. 2005, 1335545].
[13] Becker, W.; Langeluddeeke, P.; Leditschke, H. US 3954442, 1974[Chem. Abstr. 1974, 70543].
[14] Loehnert, G.; Hansigk, H. D. DE 2327954, 1975[Chem. Abstr. 1975, 125068].
[15] Xu, Z. H.; Zhang, T.; Wang, S. K.; Li, J. K. Chin. J. Org. Chem. 2017, 37, 526(in Chinese). (徐志红, 张涛, 王少康, 李俊凯, 有机化学, 2017, 37, 526.)
[16] Tatao, L.; William, L. GB 2179352, 1987[Chem. Abstr. 1987, 213492].
[17] Graham, J. B.; Lindsay, E. C.; Graeme, J. F.; Wendy, A. J.; Jack, L.; Alexander, S.; Richard, B. W. and Keith, G. W. Bioorg. Med. Chem. Lett. 1997, 12, 1489.
[18] Kim, D. S.; Chun, S. J.; Jeon, J. J.; Lee, S. W.; Joe, G. H. Pest Manage. Sci. 2004, 60, 1007.
[19] John, W.; Sandra, J.; Allan, R. US 6300281, 2001[Chem. Abstr. 2001, 741533].
[20] Prem, N. M.; Michael, P.; Sammes. J. Chem. Soc., Perkin. Trans. 1988, pp. 161~168.
[21] Tsutomu, K.; Kiyoshi, K.; Mikio, S. US 4355184, 1982[Chem. Abstr. 1982, 103658].
[22] Bernard, L. Synth. Commun. 1992, 22, 2343.
[23] Morris, G. M.; Goodsell, D. S.; Halliday, R. S.; Huey, R.; Hart, W. E.; Belew, R. K.; Olson, A. J. Comput. Chem. 1998, 19, 1639.
[24] Xiong, L.; Li, H.; Jiang, L. N.; Ge, J. M.; Yang, W. C.; Zhu, X. L.; Yang, G. F. J. Agric. Food Chem. 2017, 65, 1021.
/
| 〈 |
|
〉 |