

Chinese Journal of Organic Chemistry >
Total Synthesis of Cudratricusxanthone B
Received date: 2017-08-30
Revised date: 2017-09-21
Online published: 2017-10-24
Supported by
Project supported by the National Natural Science Foundation of China (Nos. 21472025, 21172044, 81222045, 21372049).
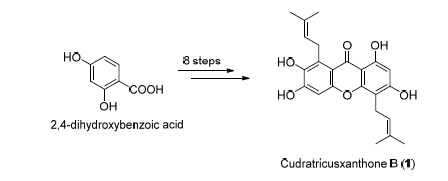
The efficient total synthesis of cudratricusxanthone B (1), a biologically interesting natural isoprenylated xanthone, has been achieved for the first time starting from commercially available 2,4-dihydroxybenzoic acid via a linear reaction sequence of 8 steps with the overall yield of 3.1%, wherein Claisen rearrangement and demethylation with AlCl3/pyridine are used as key reactions. This work definitely laid the foundation for the further pharmacological study of this natural compound. Meanwhile, another xanthone (2) has been synthesized, which proved the reported structure of the natural xanthone staudtiixanthone D to be wrong.
Zhou Pengfei , Hou Aijun , Wang Yang . Total Synthesis of Cudratricusxanthone B[J]. Chinese Journal of Organic Chemistry, 2018 , 38(1) : 156 -161 . DOI: 10.6023/cjoc201708060
[1] Zou, Y. S.; Hou, A. J.; Zhu, G. F.; Chen, Y. F.; Sun, H. D.; Zhao, Q. S. Bioorg. Med. Chem. 2004, 12, 1947.
[2] Zhang, Z.; Wu, H. J.; Pi, E. H.; Zhang, T.; Yuan, W. Y.; Hou, A. J.; Liu Q. H. World Clin. Drugs 2009, 30, 601(in Chinese).(张志, 吴海健, 皮恩浩, 张瑱, 袁文越, 侯爱君, 刘全海, 世界临床药物, 2009, 30, 601.)
[3] Ngoupayo, J.; Tabopda, T. K.; Ali, M. S. Bioorg. Med. Chem. 2009, 17, 5688.
[4] (a) Masters, K. S.; Brase, S. Chem. Rev. 2012, 112, 3717.
(b) Sousa; M. E.; Pinto; M. M. M. Curr. Med. Chem. 2005, 12, 2447.
(c) Iikubo, K.; Ishikawa, Y.; Ando, N.; Umezawa, K.; Nishiyam, S. Tetrahedron Lett. 2002, 43, 291.
(d) Hintermann, L.; Masuo, R.; Suzuki, K. Org. Lett. 2008, 10, 4859.
(e) Lin, S.; Koh, J. J.; Aung, T. T.; Lim, F.; Li, J.; Zou, H.; Wang, L.; Lakshminarayanan, R.; Verma, C.; Wang, Y.; Tan, D. T. H.; Cao, D.; Beuerman, R. W.; Ren, Li.; Liu, S. J. Med. Chem. 2017, 60, 1362.
(f) Lewis, J. R. Proc. Chem. Soc. 1963, 373.
(g) Yamazaki, S. Org. Lett. 1999, 1, 2129.
[5] Keana, J. F. W.; Guzikowski, A. P.; Nogales, D. F.; Cai, S. X. US 5476933, 1995[Chem. Abstr. 1995, 124, 232271].
[6] Kumar, S.; Reddy L, C. S.; Kumar, Y.; Kumar, A.; Singh, B. K.; Kumar, V.; Malhotra, S.; Pandey, M. K.; Jain, R.; Thimmulappa, R.; Sharma, S. K.; Prasad, A. K.; Biswal, S.; Van der Eycken, E.; DePass, A. L.; Malhotra, S. V.; Ghosh, B.; Parmar, V. S. Arch. Pharm. Chem. Life Sci. 2012, 345, 368.
[7] Oida, S.; Ohashi, Y.; Ohki, E. Chem. Pharm. Bull. 1973, 21, 528.
[8] Wu, Z.; Wei, G.; Lian, G.; Yu, B. J. Org. Chem. 2010, 75, 5725.
[9] Tisdale, E. J.; Slobodov, I.; Theodorakis, E. A. Org. Biomol. Chem. 2003, 1, 4418.
[10] Sun, H.; Chen, F.; Wang, X.; Liu, Z.; Yang, Q.; Zhang, X.; Zhu, J.; Qiang, L.; Guo, Q.; You, Q. Eur. J. Med. Chem. 2012, 51, 110.
[11] Kapingu, M. C.; Magadula, J. J. Nat. Prod. Commun. 2008, 3(9), 1501.
[12] Gokaraju, G. R.; Gokaraju, R. R.; Golakoti, T.; Somepalli, V.; Bhupathiraju, K. WO 2009093259, 2009[Chem. Abstr. 1983, 151, 198136].
[13] Fache, M.; Darroman, E.; Besse, V.; Auvergne, R.; Caillol, S.; Boutevin, B. Green Chem. 2014, 16, 1987.
[14] Shinoda, J.; Sato, D.; Kawagoye, M. Yakugaku Zasshi 1932, 52, 766.
[15] Hardegger, E.; Steiner, K.; Widmer, E.; Corrodi, H.; Schmidt, T.; Knoepfel, H. P.; Rieder, W.; Meyer, H. J.; Kugler, F.; Gempeler, H. Helv. Chim. Acta 1964, 47, 1996.
[16] Evans, J. C.; Klix, R. C.; Bach, R. D. J. Org. Chem. 1988, 53, 5519.
[17] Ueno, A. Yakugaku Zasshi 1962, 82, 1482.
/
| 〈 |
|
〉 |