

Chinese Journal of Organic Chemistry >
Synthesis and Anticonvulsant Activity Evaluation of 6-Substituted-pyrido[3,2-e] [1,2,4]triazolo[4,3-a]pyrazine Derivatives
Received date: 2017-12-18
Revised date: 2018-02-02
Online published: 2018-04-12
Supported by
Project supported by the National Natural Scicence Foundation of China (Nos. 21662036, 81660837).
In this paper, a series of 6-substituted-pyrido[3,2-e] [1,2,4]triazolo[4,3-a]pyrazine derivatives have been synthesized. Their anticonvulsant activity and neurotoxicity in mice were evaluated by maximal electroshock (MES) and rotarod test, respectively. The structures were confirmed by 1H NMR, 13C NMR, MS and HRMS. The experimental results show that 6-phenoxypyrido[3,2-e] [1,2,4]triazolo[4,3-a]pyrazine (5g) was safer than the reference drug, carbamazepine, with ED50value of 93.9 mg·kg-1and protective index (PI) value of 24.3, which was a potential anti-epilepsy candidate compound.
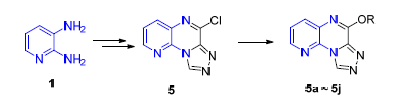
Key words: pyrido[2,3-b]pyrazine; triazole; synthesis; anticonvulsant; the maximal electroshock
Li Jiali , Hu Tao , Zhang Hongjian , Gong Guohua , Quan Zhe-Shan . Synthesis and Anticonvulsant Activity Evaluation of 6-Substituted-pyrido[3,2-e] [1,2,4]triazolo[4,3-a]pyrazine Derivatives[J]. Chinese Journal of Organic Chemistry, 2018 , 38(7) : 1673 -1679 . DOI: 10.6023/cjoc201712026
[1] Sirven, J. I.; Noe, K.; Hoerth, M.; Drazkowski, J. Mayo Clin. Proc. 2012, 87, 879.
[2] Wang, P.; Ren, R. N. Int. J. Intern. Med. 2007, 34(2), 91 (in Chinese).
(王萍, 任榕娜, 国际内科学杂志, 2007, 34(2), 91.)
[3] Genc, M.; Karagoz, G. Z.; Tekin, S.; Sandal, S.; Sirajuddin, M.; Hadda, T. B.; Sekerci, M. Acta Chim. Slov. 2016, 63(4), 726.
[4] Milošev, M. Z.; Jakovljevi?, K.; Joksovi?, M. D.; Stanojkovi?, T.; Mati?, I. Z.; Perovi?, M.; Teši?, V.; Kanazir, S.; Mladenovi?, M.; Rodi?, M. V.; Leovac, V. M.; Trifunovi?, S.; Markovi?, V. Chem. Biol. Drug Des. 2017, 89(6), 943.
[5] Mekheimer, R. A.; Sayed, A. A.; Ahmed, E. A.; Sadek, K. U. Arch. Pharm. (Weinheim) 2015, 348(9), 650.
[6] El-Sayed, N. N.; Abdelaziz, M. A.; Wardakhan, W. W. Mohareb, R. M. Steroids 2016, 107, 98.
[7] Wittine, K.; Stipkovi?, B. M.; Makuc, D.; Plavec, J.; Kraljevi?, P. S.; Sedi?, M.; Paveli?, K.; Leyssen, P.; Neyts, J.; Balzarini, J.; Mintas, M. Bioorg. Med. Chem. 2012, 20(11), 3675.
[8] Benci, K.; Suhina, T.; Mandi?, L.; Paveli?, S. K.; Paravi?, A. T.; Paveli?, K.; Balzarini, J.; Wittine, K.; Mintas, M. Antivir. Chem. Chemother. 2011, 21(6), 221.
[9] Akbari, D. K.; Nasuhi, P. F.; Hatami, N. M. Iran. J. Pharm. Res. 2015, 14(3), 693.
[10] Xiao, H.; Li, P.; Hu, J.; Li, R.; Wu, L.; Guo, D. Appl. Biochem. Biotechnol. 2014, 172(4), 2188.
[11] Flieger, J.; Kowalska, A.; Pizoń, M.; Plech, T.; ?uszczki, J. J. Sep. Sci. 2015, 38(12), 2149.
[12] Plech, T.; Kaproń, B.; Luszczki, J. J.; Paneth, A.; Siwek, A.; Ko?aczkowski, M.; ?o?nierek, M.; Nowak, G. Eur. J. Med. Chem. 2014, 86, 690.
[13] Chen, J.; Sun, X. Y.; Chai, K. Y.; Lee, J. S.; Song, M. S.; Quan, Z. S. Bioorg. Med. Chem. 2007, 15(21), 6775.
[14] Zhang, H. J.; Jin, P.; Wang, S. B.; Li, F. N.; Guan, L. P.; Quan, Z. S. Arch. Pharm. (Weinheim) 2015, 348(8), 564.
[15] Bian, M.; Deng, X. Q.; Gong, G. H.; Wei, C. X.; Quan, Z. S. J. Enzyme Inhib. Med. Chem. 2013, 28(4), 792.
[16] Xie, D.; Lu, J.; Xie, J.; Cui, J.; Li, T. F.; Wang, Y. C.; Chen, Y.; Gong, N.; Li, X. Y.; Fu, L.; Wang, Y. X. Eur. J. Med. Chem. 2016, 117, 19.
[17] Han, Z.; Hao, X.; Ma, B.; Zhu, C. Eur. J. Med. Chem. 2016, 121, 308.
[18] Unciti-Broceta, A.; Pineda de las Infantas, M. J.; Gallo, M. A.; Espinosa, A. Chemistry 2007, 13(6), 1754.
[19] Zhang, H. M.; Zhang, H. J.; Tian, Y. S.; Quan, Z. S. Chin. J. Org. Chem. 2017, 37(9), 2322 (in Chinese).
(张海明, 张洪健, 田玉顺, 全哲山, 有机化学, 2017, 37(9), 2322.)
[20] Liu, Q. L.; Fang, P. J.; Zhao, Z. L.; Zhang, H. Z.; Zhou, C. H. J. Org. Chem. 2017, 12, 3146 (in Chinese).
(刘庆龙, 房鹏金, 赵志龙, 张慧珍, 周成合, 有机化学, 2017, 12, 3146.)
[21] Güngör, T.; Chen, Y.; Golla, R.; Ma, Z.; Corte, J. R.; Northrop, J. P.; Bin, B.; Dickson, J. K.; Stouch, T.; Zhou, R.; Johnson, S. E.; Seethala, R.; Feyen, J. H. J. Med. Chem. 2006, 49, 2440.
[22] Alagarsamy, V.; Dhanabal, K.; Parthiban, P.; Anjana, G.; Deepa, G.; Murugesan, B.; Rajkumar, S.; Beevi, A. J. J. Pharm. Pharmacol. 2007, 59, 669.
[23] Krall, R. L.; Penry, J. K.; White, B. G. Epilepsia 1978, 19, 409.
[24] Porter, R. J.; Cereghino, J. J.; Gladding, G. D. Cleveland Clin. Q. 1984, 51, 293.
/
| 〈 |
|
〉 |