

Chinese Journal of Organic Chemistry >
Strategies for Construction of Cyclopropanes in Natural Products
Received date: 2018-05-31
Revised date: 2018-06-26
Online published: 2018-07-24
Supported by
Project supported by the National Natural Science Foundation of China (No. 21502217) and the Science and Technology Commission of Shanghai Municipality (No. 15ZR1449400).
Cyclopropane-containing natural products frequently possess excellent biological activities, and may be developed as drug leads. Although the inherent strain of the cyclopropane greatly challenges both chemical synthesis and biosynthesis, great advances have been made for the construction of the cyclopropane in natural products by chemical synthesis owing to the importance of this kind of compounds. Many enzymes responsible for cyclopropanation have also been unraveled. This review summarizes the cyclopropanation strategies in chemical synthesis and biosynthesis. The strategies used in chemical synthesis mainly consist of three classes:(i) a carbene involved mechanism, (ii) an SN2 reaction mechanism, and (iii) cycloisomerization. The strategies discovered in nature are reviewed on the basis of the carbon state involved, including (i) a carbocation, (ii) a carbanion, and (iii) a carbon radical. Chemical synthesis and biosynthesis are mutually simulative because the strategies developed in chemical synthesis may inspire enzymologists to discover and design new biochemical reactions and vice versa.
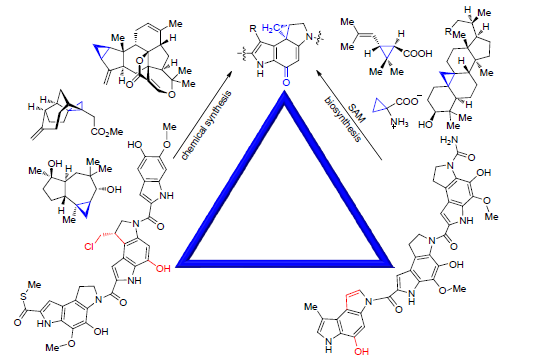
Jin Wenbing , Yuan Hua , Tang Gongli . Strategies for Construction of Cyclopropanes in Natural Products[J]. Chinese Journal of Organic Chemistry, 2018 , 38(9) : 2324 -2334 . DOI: 10.6023/cjoc201805059
[1] Freund, A. J. Prakt. Chem. 1882, 26, 367.
[2] Staudinger, H.; Ruzicka, L. Helv. Chim. Acta 1924, 7, 177.
[3] Wessjohann, L. A.; Brandt, W.; Thiemann, T. Chem. Rev. 2003, 103, 1625.
[4] Pandit, J.; Danley, D. E.; Schulte, G. K.; Mazzalupo, S.; Pauly, T. A.; Hayward, C. M.; Hamanaka, E. S.; Thompson, J. F.; Harwood, H. J. J. Biol. Chem. 2000, 275, 30610.
[5] Capitani, G.; Hohenester, E.; Feng, L.; Storici, P.; Kirsch, J. F.; Jansonius, J. N. J. Mol. Biol. 1999, 294, 745.
[6] Menderes, G.; Bonazzoli, E.; Bellone, S.; Black, J.; Predolini, F.; Pettinella, F.; Masserdotti, A.; Zammataro, L.; Altwerger, G.; Buza, N.; Hui, P.; Wong, S.; Litkouhi, B.; Ratner, E.; Silasi, D. A.; Azodi, M.; Schwartz, P. E.; Santin, A. D. Clin. Cancer Res. 2017, 23, 5836.
[7] Tietze, L. F.; Krewer, B. Anticancer Agents Med. Chem. 2009, 9, 304.
[8] Sievers, E. L.; Senter, P. D. Annu. Rev. Med. 2013, 64, 15.
[9] Wipf, P.; Reeves, J. T.; Day, B. W. Curr. Pharm. Des. 2004, 10, 1417.
[10] Hiratsuka, T.; Suzuki, H.; Kariya, R.; Seo, T.; Minami, A.; Oikawa, H. Angew. Chem., Int. Ed. 2014, 53, 5423.
[11] Silverman, R. B.; Zieske, P. A. Biochemistry 1985, 24, 2128.
[12] Wentland, M. P.; Lu, Q.; Lou, R.; Bu, Y.; Knapp, B. I.; Bidlack, J. M. Bioorg. Med. Chem. Lett. 2005, 15, 2107.
[13] Talele, T. T. J. Med. Chem. 2016, 59, 8712.
[14] Lászlò, K.; Barbara, C. Strategic Applications of Named Reactions in Organic Synthesis, Elsevier Academic Press, Burlington, 2005, p. 412.
[15] Parthasarathy, G.; Eggert, U.; Kalesse, M. Org. Lett. 2016, 18, 2320.
[16] Jin, S.; Gong, J.; Qin, Y. Angew. Chem., Int. Ed. 2015, 54, 2228.
[17] Silberrad, O. R.; Roy, C. S. J. Chem. Soc. 1906, 89, 179.
[18] Nozaki, H.; Moriuti, S.; Yamabe, M.; Noyori, R. Tetrahedron Lett. 1966, 7, 59.
[19] Moser, W. R. J. Am. Chem. Soc. 1969, 91, 1141.
[20] Paulissen, R.; Hubert, A. J.; Teyssie, P. Tetrahedron Lett. 1972, 13, 1465.
[21] Paulissen, R.; Reimlinger, H.; Hayez, E.; Hubert, A. J.; Teyssie, P. Tetrahedron Lett. 1973, 14, 2233.
[22] Hubert, A. J.; Noels, A. F.; Anciaux, A. J.; Teyssie, P. Synthesis 1976, 600.
[23] Yuan, C. C.; Du, B. A.; Yang, L.; Liu, B. J. Am. Chem. Soc. 2013, 135, 9291.
[24] Doering, W.; Hoffmann, A. K. J. Am. Chem. Soc. 1954, 76, 6162.
[25] Pan, S. Y.; Xuan, J.; Gao, B. L.; Zhu, A.; Ding, H. F. Angew. Chem., Int. Ed. 2015, 54, 6905.
[26] Tichenor, M. S.; Kastrinsky, D. B.; Boger, D. L. J. Am. Chem. Soc. 2004, 126, 8396.
[27] Tichenor, M. S.; Trzupek, J. D.; Kastrinsky, D. B.; Shiga, F.; Hwang, I.; Boger, D. L. J. Am. Chem. Soc. 2006, 128, 15683.
[28] Okano, K.; Tokuyama, H.; Fukuyama, T. J. Am. Chem. Soc. 2006, 128, 7136.
[29] Strickler, H.; Davis, J. B.; Ohloff, G. Helv. Chim. Acta 1976, 59, 1328.
[30] Bruneau, C. Angew. Chem., Int. Ed. 2005, 44, 2328.
[31] Mainetti, E.; Mouries, V.; Fensterbank, L.; Malacria, M.; Marco-Contelles, J. Angew. Chem., Int. Ed. 2002, 41, 2132.
[32] Lemiere, G.; Gandon, V.; Cariou, K.; Hours, A.; Fukuyama, T.; Dhimane, A. L.; Fensterbank, L.; Malacria, M. J. Am. Chem. Soc. 2009, 131, 2993.
[33] Michels, T. D.; Dowling, M. S.; Vanderwal, C. D. Angew. Chem., Int. Ed. 2012, 51, 7572.
[34] Homs, A.; Muratore, M. E.; Echavarren, A. M. Org. Lett. 2015, 17, 461.
[35] Hodgson, D. M.; Chung, Y. K.; Paris, J. M. J. Am. Chem. Soc. 2004, 126, 8664.
[36] Hodgson, D. M.; Chung, Y. K.; Nuzzo, I.; Freixas, G.; Kulikiewicz, K. K.; Cleator, E.; Paris, J. M. J. Am. Chem. Soc. 2007, 129, 4456.
[37] Hodgson, D. M.; Salik, S.; Fox, D. J. J. Org. Chem. 2010, 75, 2157.
[38] Büchner, E.; Perkel, L. Ber. Dtsch. Chem. Ges. 1903, 36, 3774.
[39] Buchner, E.; Curtius, T. Ber. Dtsch. Chem. Ges. 1885, 18, 2377.
[40] Rinehart, K. L.; Van Auken, T. V. J. Am. Chem. Soc. 1960, 82, 5251.
[41] Kirillova, M. S.; Muratore, M. E.; Dorel, R.; Echavarren, A. M. J. Am. Chem. Soc. 2016, 138, 3671.
[42] Kingsbury, J. S.; Corey, E. J. J. Am. Chem. Soc. 2005, 127, 13813.
[43] Kim, K.; Cha, J. K. Angew. Chem., Int. Ed. 2009, 48, 5334.
[44] Gaich, T.; Mulzer, J. J. Am. Chem. Soc. 2009, 131, 452.
[45] Walsh, C. T. ACS Chem. Biol. 2007, 2, 296.
[46] Thibodeaux, C. J.; Chang, W. C.; Liu, H. W. Chem. Rev. 2012, 112, 1681.
[47] Liao, R. Z.; Georgieva, P.; Yu, J. G.; Himo, F. Biochemistry 2011, 50, 1505.
[48] Zha, L.; Jiang, Y.; Henke, M. T.; Wilson, M. R.; Wang, J. X.; Kelleher, N. L.; Balskus, E. P. Nat. Chem. Biol. 2017, 13, 1063.
[49] Frederic, H.; Vaillancourt, E. Y.; Vosburg, D. A.; O'Connor, S. E.; Walsh, C. T. Nature 2005, 436, 1191.
[50] Kelly, W. L.; Boyne, M. T.; Yeh, E.; Vosburg, D. A.; Galoni, D. P.; Kelleher, N. L.; Walsh, C. T. Biochemistry 2007, 46, 359.
[51] Jiang, W.; Heemstra, J. R.; Forseth, R. R.; Neumann, C. S.; Manaviazar, S.; Schroeder, F. C.; Hale, K. J.; Walsh, C. T. Biochemistry 2011, 50, 6063.
[52] Gu, L.; Wang, B.; Kulkarni, A.; Geders, T. W.; Grindberg, R. V.; Gerwick, L.; Hakansson, K.; Wipf, P.; Smith, J. L.; Gerwick, W. H.; Sherman, D. H. Nature 2009, 459, 731.
[53] Wu, S.; Jian, X. H.; Yuan, H.; Jin, W. B.; Yin, Y.; Wang, L. Y.; Zhao, J.; Tang, G. L. ACS Chem. Biol. 2017, 12, 1603.
[54] Jin, W. B.; Wu, S.; Jian, X. H.; Yuan, H.; Tang, G. L. Nat. Commun. 2018, 9, 2771.
[55] Wang, X.; Wu, S.; Jin, W.; Xu, B.; Tang, G.; Yuan, H. Acta Biochim. Biophys. Sin. 2018, 50, 516.
[56] Jakubczyk, D.; Caputi, L.; Hatsch, A.; Nielsen, C. A.; Diefenbacher, M.; Klein, J.; Molt, A.; Schröder, H.; Cheng, J. Z.; Naesby, M.; O'Connor, S. E. Angew. Chem., Int. Ed. 2015, 54, 5117.
[57] Coelho, P. S.; Brustad, E. M.; Kannan, A.; Arnold, F. H. Science 2013, 339, 307.
/
| 〈 |
|
〉 |