

Chinese Journal of Organic Chemistry >
Transition Metal-Catalyzed Regio-selective Aromatic C—H Bond Oxidation for C—O Bond Formation
Received date: 2018-08-16
Revised date: 2018-10-22
Online published: 2018-10-26
Supported by
Project supported by the National Natural Science Foundation of China (No. 21801018) and the Beijing Institute of Technology Research Fund Program for Young Scholars (No. 1230011181807).
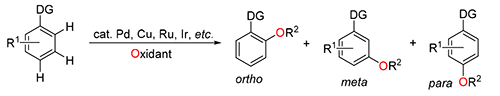
Recent years the great progress in transition metal-catalyzed direct aromatic C—H oxidation has been witnessed, which has been utilized in the preparation of various phenolic compounds. These transformations employ inter alia palladium, copper, ruthenium, iridium, etc. as the transition metal catalysts, and hypervalent iodine, persulfate, or oxygen as the oxidants. There have been several reviews in which the C—H oxidations with specific transition metal or oxidant was discussed. This review focuses specifically on transition metal-catalyzed aromatic C—H oxidations with ortho-, meta-, or para-selectivity, and rationalizes the possible generation mechanism of regio-selectivities, which might be controlled by the directing group via chelation-assistance, the ligand, or intrinsic properties of the substrate. The discussion section indicated the existing problems of transition metal-catalyzed aromatic C—H oxidations, as well as the possible limiting factors for the development and application of this strategy.
Key words: transition metal; arene; C—H oxidation; regio-selectivity; directing group
Yang Fanzhi , Zhang Han , Liu Xuri , Wang Bo , Ackermann Lutz . Transition Metal-Catalyzed Regio-selective Aromatic C—H Bond Oxidation for C—O Bond Formation[J]. Chinese Journal of Organic Chemistry, 2019 , 39(1) : 59 -73 . DOI: 10.6023/cjoc201808017
[1] (a) Rappoport, Z. The Chemistry of Phenols, Wiley-VCH, Weinheim, 2003.
(b) Fiegel, H.; Voges, H. W.; Hamamoto, T.; Umemura, S.; Iwata, T.; Miki, H.; Fujita, Y.; Buysch, H. J.; Garbe, D.; Paulus, W. Phenol Derivatives in Ullmann's Encyclopedia of Industrial Chemistry, Wiley-VCH, New York, 2002.
(c) Hartwig, J. F. In Handbook of Organopalladium Chemistry for Organic Synthesis, Vol. 1, Ed.:Negishi, E.-I., Wiley-Interscience, New York, 2002, p. 1097.
(d) Tyman, J. H. P. Synthetic and Natural Phenols, Elsevier, New York, 1996.
(e) Liao, S.; Zhang, G.; Cao, H.; Chen, B.; Li, W.; Wu, X.; Feng, Y. Chin. J. Org. Chem. 2018, 38, 1549.
(f) Lin, W.; Cai, Q.; Zheng, C.; Huang, Z.; Shi, D. Chin. J. Org. Chem. 2017, 37, 2094.
(g) Li, Z.; Kang, S.; Chen, L.; Wang, Y.; Li, J. Chin. J. Org. Chem. 2016, 36, 1143.
[2] Lyons, T. W.; Sanford, M. S. Chem. Rev. 2010, 110, 1147.
[3] Enthaler, S.; Company, A. Chem. Soc. Rev. 2011, 40, 4912.
[4] Thirunavukkarasu, V. S.; Kozhushkov, S. I.; Ackermann, L. Chem. Commun. 2014, 50, 29.
[5] Newhouse, T.; Baran, P. S. Angew. Chem. Int. Ed. 2011, 50, 3362.
[6] Liang, Y.-F.; Jiao, N. Acc. Chem. Res. 2017, 50, 1640.
[7] Mandal, S.; Bera, T.; Dubey, G.; Saha, J.; Laha, J. K. ACS Catal. 2018, 8, 5085.
[8] (a) Huang, Z.; Dong, G. Acc. Chem. Res. 2017, 50, 465.
(b) Hartwig, J. F. Acc. Chem. Res. 2017, 50, 549.
(c) Kozlowski, M. C. Acc. Chem. Res. 2017, 50, 638.
[9] For selected recent examples, see:(a) Raghuvanshi, K.; Zell, D.; Ackermann, L. Org. Lett. 2017, 19, 1278.
(b) Raghuvanshi, K.; Rauch, K.; Ackermann, L. Chem.-Eur. J. 2015, 21, 1790.
(c) Hao, X.-Q.; Chen, L.-J.; Ren, B.; Li, L.-Y.; Yang, X.-Y.; Gong, J.-F.; Niu, J.-L.; Song M.-P. Org. Lett. 2014, 16, 1104.
(d) Shi, S.; Kuang, C. J. Org. Chem. 2014, 79, 6105.
(e) Zhang, C.; Sun, P. J. Org. Chem. 2014, 79, 8457.
(f) Péron, F.; Fossey, C.; Sopkova-de Oliveira Santos, J.; Cailly, T.; Fabis, F. Chem.-Eur. J. 2014, 20, 7507.
(g) Bhadra, S.; Dzik, W. I.; Gooßen, L. J. Angew. Chem., Int. Ed. 2013, 52, 2959.
(h) Bhadra, S.; Matheis, C.; Katayev, D.; Gooßen, L. J. Angew. Chem., Int. Ed. 2013, 52, 9279.
(i) Suess, A. M.; Ertem, M. Z.; Cramer, C. J.; Stahl, S. S. J. Am. Chem. Soc. 2013, 135, 9797.
(j) Roane, J.; Daugulis, O. Org. Lett. 2013, 15, 5842.
(k) Eom, D.; Jeong, Y.; Kim, Y. R.; Lee, E.; Choi, W.; Lee, P. H. Org. Lett. 2013, 15, 5210.
(l) Zhao, J.; Wang, Y.; He, Y.; Liu, L.; Zhu, Q. Org. Lett. 2012, 14, 1078.
(m) Zhao, J.; Zhang, Q.; Liu, L.; He, Y.; Li, J.; Li, J.; Zhu, Q. Org. Lett. 2012, 14, 5362.
(n) Li, W.; Sun, P. J. Org. Chem. 2012, 77, 8362.
(o) Jiang, T.-S.; Wang, G.-W. J. Org. Chem. 2012, 77, 9504.
(p) Xiao, B.; Gong, T.-J.; Liu, Z.-J.; Liu, J.-H.; Luo, D.-F.; Xu, J.; Liu L. J. Am. Chem. Soc. 2011, 133, 9250.
(q) Anand, M.; Sunoj, R. B. Org. Lett. 2011, 13, 4802.
(r) Wei, Y.; Yoshikai, N. Org. Lett. 2011, 13, 5504.
(s) Wang, X.; Lu, Y.; Dai, H.-X.; Yu, J.-Q. J. Am. Chem. Soc. 2010, 132, 12203.
(t) Wang, G.-W.; Yuan, T.-T. J. Org. Chem. 2010, 75, 476.
[10] For selected reviews and recent examples, see:(a) Tang, S.; Liu, Y.; Lei, A. Chem 2018, 4, 27.
(b) Tang, S.; Zeng, L.; Lei, A. J. Am. Chem. Soc. 2018, 140, 13128.
(c) Ma, C.; Fang, P.; Mei, T.-S. ACS Catal. 2018, 8, 7179.
(d) Sauermann, N.; Meyer, T. H.; Qiu, Y.; Ackermann, L. ACS Catal. 2018, 8, 7086.
(e) Sauermann, N.; Meyer, T. H.; Ackermann, L. Chem.-Eur. J. 2018, 24, 16209.
(f) Jiao, K.-J.; Li, Z.-M.; Xu, X.-T.; Zhang, L.-P.; Li, Y.-Q.; Zhang, K.; Mei, T.-S. Org. Chem. Front. 2018, 5, 2244.
(g) Shao, A.; Li, N.; Gao, Y.; Zhan, J.; Chiang, C.-W.; Lei, A. Chin. J. Chem. 2018, 36, 619.
(h) Yang, Q.-L.; Fang, P.; Mei, T.-S. Chin. J. Chem. 2018, 36, 338.
(i) Jiao, K.-J.; Zhao, C.-Q.; Fang, P.; Mei, T.-S. Tetrahedron Lett. 2017, 58, 797.
(j) Yuan, Y.; Chen, Y.; Tang, S.; Huang, Z.; Lei, A. Sci. Adv. 2018, 4, eaat5312.
(k) Shrestha, A.; Lee, M.; Dunn, A. L.; Sanford, M. S. Org. Lett. 2018, 20, 204.
(l) Sauermann, N.; Meyer, T. H.; Tian, C.; Ackermann, L. J. Am. Chem. Soc. 2017, 139, 18452.
(m) Yang, Q.-L.; Li, Y.-Q.; Ma, C.; Fang, P.; Zhang, X.-J.; Mei, T.-S. J. Am. Chem. Soc. 2017, 139, 3293.
(n) Li, Y.-Q.; Yang, Q.-L.; Fang, P.; Mei, T.-S.; Zhang, D. Org. Lett. 2017, 19, 2905.
[11] A recent example:Zhang, Z.-J.; Quan, X.-J.; Ren, Z.-H.; Wang, Y.-Y.; Guan, Z.-H. Org. Lett. 2014, 16, 3292.
[12] (a) Jintoku, T.; Taniguchi, H.; Fujiwara, Y. Chem. Lett. 1987, 1865.
(b) Jintoku, T.; Takaki, K.; Fujiwara, Y.; Fuchita Y.; Hiraki, K. Bull. Chem. Soc. Jpn. 1990, 63, 438.
(c) Jintoku, T.; Nishimura, K.; Takaki, K.; Fujiwara, Y. Chem. Lett. 1990, 1687.
[13] Yamada, S.; Sakaguchi, S.; Ishii, Y. J. Mol. Catal. A Chem. 2007, 262, 48.
[14] Yoneyama, T.; Crabtree, R. H. J. Mol. Catal. A: Chem. 1996, 108, 35.
[15] Cook, A. K.; Sanford, M. S. J. Am. Chem. Soc. 2015, 137, 3109.
[16] Gary, J. B.; Cook, A. K.; Sanford, M. S. ACS Catal. 2013, 3, 700.
[17] Shibahara, F.; Kinoshita, S.; Nozaki, K. Org. Lett. 2004, 6, 2437.
[18] Chen, C.-D.; Sheng, W.-B.; Shi, G.-J.; Guo, C.-C. J. Phys. Org. Chem. 2013, 26, 23.
[19] Casella, L.; Rigoni, L. J. Chem. Soc., Chem. Commun. 1985, 1668.
[20] Réglier, M.; Amadeï, E.; Tadayoni, R.; Waegell, B. J. Chem. Soc., Chem. Commun. 1989, 447.
[21] Cruse, R. W.; Kaderli, S.; Meyer, C. J.; Zuberbühler, A. D.; Karlin, K. D. J. Am. Chem. Soc. 1988, 110, 5020.
[22] Reinaud, O.; Capdevielle, P.; Maumy, M. J. Chem. Soc., Chem. Commun. 1990, 566.
[23] Takizawa, Y.; Tateishi, A.; Sugiyama, J.; Yoshida, H.; Yoshihara, N. J. Chem. Soc., Chem. Commun. 1991, 104.
[24] Singh, B. K.; Jana, R. J. Org. Chem. 2016, 81, 831.
[25] Chen, X.; Hao, X.-S.; Goodhue, C.-E.; Yu, J.-Q. J. Am. Chem. Soc. 2006, 128, 6790.
[26] (a) Valk, J.-M.; van Belzen, R.; Boersma, J.; Spek, A. L.; van Koten, G. J. Chem. Soc., Dalton Trans. 1994, 2293.
(b) Valk, J.-M.; Boersma, J.; van Koten, G. Organometallics 1996, 15, 4366.
[27] Zhang, Y.-H.; Yu, J.-Q. J. Am. Chem. Soc. 2009, 131, 14654.
[28] Shan, G.; Yang, X.; Ma, L.; Rao, Y. Angew. Chem., Int. Ed. 2012, 51, 13070.
[29] Dick, A. R.; Hull, K. L.; Sanford, M. S. J. Am. Chem. Soc. 2004, 126, 2300.
[30] Subba Reddy, B. V.; Umadevi, N.; Narasimhulu, G.; Yadav, J. S. Tetrahedron Lett. 2012, 53, 6091.
[31] Zhao, Q.; Poisson, T.; Pannecoucke, X.; Besset, T. Synthesis 2017, 49, 4808.
[32] Gandeepan, P.; Ackermann, L. Chem 2018, 4, 199.
[33] Chen, X.-Y.; Ozturk, S.; Sorensen, E. J. Org. Lett. 2017, 19, 6280.
[34] Desai, L. V.; Malik, H. A.; Sanford, M. S. Org. Lett. 2006, 8, 1141.
[35] Liang, Y.-F.; Wang, X.; Yuan, Y.; Liang, Y.; Li, X.; Jiao, N. ACS Catal. 2015, 5, 6148.
[36] Nguyen, T. H. L.; Gigant, N.; Delarue-Cochin, S.; Joseph, D. J. Org. Chem. 2016, 81, 1850.
[37] Qian, C.; Lin, D.; Deng, Y.; Zhang, X.-Q.; Jiang, H.; Miao, G.; Tang, X.; Zeng, W. Org. Biomol. Chem. 2014, 12, 5866.
[38] Zhang, D.; Cui, X.; Yang, F.; Zhang, Q.; Zhu, Y.; Wu, Y. Org. Chem. Front. 2015, 2, 951.
[39] Kalyani, D.; Sanford, M. S. Org. Lett. 2005, 7, 4149.
[40] Irastorza, A.; Aizpurua, J. M.; Correa, A. Org. Lett. 2016, 18, 1080.
[41] Kim, S. H.; Lee, H. S.; Kim, S. H.; Kim, J. N. Tetrahedron Lett. 2008, 49, 5863.
[42] Dong, J.; Liu, P.; Sun, P. J. Org. Chem. 2015, 80, 2925.
[43] Yamaguchi, T.; Yamaguchi, E.; Tada, N.; Itoh, A. Adv. Synth. Catal. 2015, 357, 2017.
[44] Dick, A. R.; Kampf, J. W.; Sanford, M. S. J. Am. Chem. Soc. 2005, 127, 12790.
[45] Stowers, K. J.; Sanford, M. S. Org. Lett. 2009, 11, 4584.
[46] Powers, D. C.; Geibel, M. A. L.; Klein, J. E. M. N.; Ritter, T. J. Am. Chem. Soc. 2009, 131, 17050.
[47] Powers, D. C.; Xiao, D. Y.; Geibel, M. A. L.; Ritter, T. J. Am. Chem. Soc. 2010, 132, 14530.
[48] Yan, Y.; Feng, P.; Zheng, Q.-Z.; Liang, Y.-F.; Lu, J.-F.; Cui, Y.; Jiao, N. Angew. Chem., Int. Ed. 2013, 52, 5827.
[49] Desai, L. V; Stowers, K. J.; Sanford, M. S. J. Am. Chem. Soc. 2008, 130, 13285.
[50] Wang, G.-W.; Yuan, T.-T.; Wu, X.-L. J. Org. Chem. 2008, 73, 4717.
[51] Sun, Y.-H.; Sun, T.-Y.; Wu, Y.-D.; Zhang, X.; Rao, Y. Chem. Sci. 2016, 7, 2229.
[52] Yang, X.; Sun, Y.; Chen, Z.; Rao, Y. Adv. Synth. Catal. 2014, 356, 1625.
[53] A review:Parasram, M.; Gevorgyan, V. Acc. Chem. Res. 2017, 50, 2038.
[54] (a) Chernyak, N.; Dudnik, A. S.; Huang, C.; Gevorgyan, V. J. Am. Chem. Soc. 2010, 132, 8270.
(b) Huang, C.; Chernyak, N.; Dudnik, A. S.; Gevorgyan, V. Adv. Synth. Catal. 2011, 353, 1285.
(c) Gulevich, A. V.; Melkonyan, F. S.; Sarkar, D.; Gevorgyan, V. J. Am. Chem. Soc. 2012, 134, 5528.
[55] (a) Huang, C.; Ghavtadze, N.; Chattopadhyay, B.; Gevorgyan, V. J. Am. Chem. Soc. 2011, 133, 17630.
(b) Huang, C.; Ghavtadze, N.; Godoi, B.; Gevorgyan, V. Chem.-Eur. J. 2012, 18, 9789.
[56] Yang, Y.; Lin, Y.; Rao, Y. Org. Lett. 2012, 14, 2874.
[57] Ackermann, L.; Vicente, R.; Potukuchi, H. K.; Pirovano, V. Org. Lett. 2010, 12, 5032.
[58] Thirunavukkarasu, V. S.; Hubrich, J.; Ackermann, L. Org. Lett. 2012, 14, 4210.
[59] Yang, F.; Ackermann, L. Org. Lett. 2013, 15, 718.
[60] Thirunavukkarasu, V. S.; Ackermann, L. Org. Lett. 2012, 14, 6206.
[61] Shan, G.; Han, X.; Lin, Y.; Yu, S.; Rao, Y. Org. Biomol. Chem. 2013, 11, 2318.
[62] Kim, K.; Choe, H.; Jeong, Y.; Lee, J. H.; Hong, S. Org. Lett. 2015, 17, 2550.
[63] Yang, F.; Rauch, K.; Kettelhoit, K.; Ackermann, L. Angew. Chem., Int. Ed. 2014, 53, 11285.
[64] Shome, S.; Singh, S. P. Tetrahedron Lett. 2017, 58, 3743.
[65] Ackermann, L.; Wang, L.; Wolfram, R.; Lygin, A. V. Org. Lett. 2012, 14, 728.
[66] Yang, X.; Shan, G.; Rao, Y. Org. Lett. 2013, 15, 2334.
[67] Liu, W.; Ackermann, L. Org. Lett. 2013, 15, 3484.
[68] Turlington, C. R.; Morris, J.; White, P. S.; Brennessel, W. W.; Jones, W. D.; Brookhart, M.; Templeton, J. L. Organometallics 2014, 33, 4442.
[69] Wu, Q.; Yan, D.; Chen, Y.; Wang, T.; Xiong, F.; Wei, W.; Lu, Y.; Sun, W.-Y.; Li, J. J.; Zhao, J. Nat. Commun. 2017, 8, 14227.
[70] Cook, A. K.; Emmert, M. H.; Sanford, M. S. Org. Lett. 2013, 15, 5428.
[71] For selected recent views on template-directed meta-selective C(sp2)-H functionalizations, see:(a) Mihai, M. T.; Genov, G. R.; Phipps, R. J. Chem. Soc. Rev. 2017, 47, 149.
(b) Ping, L.; Chung, D. S.; Bouffard, J.; Lee, S.-G. Chem. Soc. Rev. 2017, 46, 4299.
(c) Mihai, M. T.; Phipps, R. J. Synlett 2017, 28, 1011.
(d) Dey, A.; Agasti, S.; Maiti, D. Org. Biomol. Chem. 2016, 14, 5440.
(e) Yang, G.; Butt, N.; Zhang, W. Chin. J. Catal. 2016, 37, 98.
(f) Yang, G.; Butt, N.; Zhang, W. Chin. J. Catal. 2016, 37, 98.
(g) Ackermann, L.; Li, J. Nat. Chem. 2015, 7, 686.
(h) Yang, J. Org. Biomol. Chem. 2015, 13, 1930.
[72] (a) Bera, M.; Sahoo, S. K.; Maiti, D. ACS Catal. 2016, 6, 3575.
(b) Maji, A.; Bhaskararao, B.; Singha, S.; Sunoj, R. B.; Maiti, D. Chem. Sci. 2016, 7, 3147.
[73] Bag, S.; Patra, T.; Modak, A.; Deb, A.; Maity, S.; Dutta, U.; Dey, A.; Kancherla, R.; Maji, A.; Hazra, A.; Bera, M.; Maiti, D. J. Am. Chem. Soc. 2015, 137, 11888.
[74] A review on template-directed para-selective C(sp2)-H functionalizations:Dey, A.; Maity, S.; Maiti, D. Chem. Commun. 2016, 52, 12398.
/
| 〈 |
|
〉 |