

Chinese Journal of Organic Chemistry >
Palladium-Catalyzed Dehydrative Cross Couplings of Stabilized Phosphorus Ylides with Allylic Alcohols
Received date: 2018-12-31
Revised date: 2019-01-27
Online published: 2019-02-22
Supported by
Project supported by the Scientific Research Project of Henan Province (No. 19B150018), the Nanhu Scholars Program for Young Scholars of Xinyang Normal University and the Young Core Instructor Program of Xinyang Normal University (No. 2018GGJS-05).
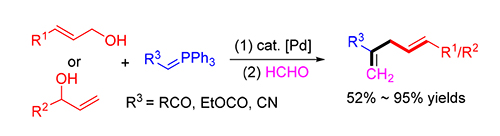
A dehydrative cross coupling of ketone-stabilized phosphorus ylides with the readily available allylic alcohols followed by an one-pot Wittig reaction is developed. A range of functional 1,4-dienes could be obtained in 52%~95% isolated yields in the presence of 5 mol% Pd(PPh3) 4 and 20 mol% B(OH) 3. The same method can be extended to ester or nitrile-stabi-lized phosphorus ylides, affording the corresponding 1,4-dienes in moderate yields.
Ma Xiantao , Yu Jing , Ma Ruitian , Yan Ran , Zhang Zhenlei . Palladium-Catalyzed Dehydrative Cross Couplings of Stabilized Phosphorus Ylides with Allylic Alcohols[J]. Chinese Journal of Organic Chemistry, 2019 , 39(3) : 830 -835 . DOI: 10.6023/cjoc201812051
[1] (a) Jie, M. S. F. L. K.; Pasha, M. K.; Syed-Rahmatulla, M. S. K. Nat. Prod. Rep. 1997, 14, 163.
(b) Fürstner, A.; Nevado, C.; Waser, M.; Tremblay, M.; Chevrier, C.; Teplý, F.; Aïssa, C.; Moulin, E.; Müller, O. J. Am. Chem. Soc. 2007, 129, 9150.
(c) Wilson, M. C.; Nam, S.-J.; Gulder, T. A. M.; Kauffman, C. A.; Jensen, P. R.; Fenical, W.; Moore, B. S. J. Am. Chem. Soc. 2011, 133, 1971.
[2] (a) Macklin, T. K.; Micalizio, G. C. Nat. Chem. 2010, 2, 638.
(b) Sharma, R. K.; RajanBabu, T. V. J. Am. Chem. Soc. 2010, 132, 3295.
(c) Trost, B. M.; Luan, X. J. Am. Chem. Soc. 2011, 133, 1706.
(d) McCammant, M. S.; Liao, L.; Sigman, M. S. J. Am. Chem. Soc. 2013, 135, 4167.
(e) Jin, W.; Yang, Q.; Wu, P.; Chen, J.; Yu, Z. Adv. Synth. Catal. 2014, 360, 2097.
[3] (a) Miyaura, N.; Yano, T.; Suzuki, A. Tetrahedron Lett. 1980, 21, 2865.
(b) Kabalka, G. W.; Al-Masum, M. Org. Lett. 2006, 8, 11.
(c) Lee, Y.; Akiyama, K.; Gillingham, D. G.; Brown, M. K.; Hoveyda, A. H. J. Am. Chem. Soc. 2008, 130, 446.
(d) Akiyama, K.; Gao, F.; Hoveyda, A. H. Angew. Chem., Int. Ed. 2010, 49, 419.
(e) Gao, F.; Lee, K. P.; McGrath, Y.; Hoveyda, A. H. J. Am. Chem. Soc. 2010, 132, 14315.
(f) Gao, F.; Carr, J. L.; Hoveyda, A. H. Angew. Chem., Int. Ed. 2012, 51, 6613.
(g) Huang, Y.; Fañanás-Mastral, M.; Minnaard, A. J.; Feringa, B. L. Chem. Commun. 2013, 49, 3309.
(h) Hamilton, J. Y.; Sarlah, D.; Carreira, E. M. J. Am. Chem. Soc. 2013, 135, 994.
(i) Gao, F.; Carr, J. L.; Hoveyda, A. H. J. Am. Chem. Soc. 2014, 136, 2149.
(j) Sidera, M.; Fletcher, S. P. Chem. Commun. 2015, 51, 5044.
(k) Zhurkin, F. E.; Hu, X. J. Org. Chem. 2016, 81, 5795.
(l) Yang, B.; Wang, Z.-X. J. Org. Chem. 2017, 82, 4542.
[4] (a) Cornella, J.; Zarate, C.; Martin, R. Chem. Soc. Rev. 2014, 43, 8081.
(b) Matsubara, R.; Jamison, T. F. J. Am. Chem. Soc. 2010, 132, 6880.
(c) Matsubara, R.; Jamison, T. F. Chem. Asian J. 2011, 6, 1860.
(d) Ye, K.-Y.; He, H.; Liu, W.-B.; Dai, L.-X.; Helmchen, G.; You, S.-L. J. Am. Chem. Soc. 2011, 133, 19006.
(e) Hamilton, J. Y.; Sarlah, D.; Carreira, E. M. J. Am. Chem. Soc. 2014, 13 6, 2006.
(f) Gumrukcu, Y.; de Bruin, B.; Reek, J. N. H. Chem.-Eur. J. 2014, 20, 10905.
[5] (a) Thadani, A. N.; Rawal, V. H. Org. Lett. 2002, 4, 4317.
(b) Chen, X.; Chen, D.; Lu, Z.; Kong, L.; Zhu, G.-G. J. Org. Chem. 2011, 76, 6338.
(c) Wen, Y.; Jiang, H.-F. Tetrahedron Lett. 2013, 54, 4034.
(d) Todd, D. P.; Thompson, B. B.; Nett, A. J.; Montgomery, J. J. Am. Chem. Soc. 2015, 137, 12788
(e) Mateos, J.; Rivera-Chao, E.; Fañanás-Mastral, M. ACS Catal. 2017, 7, 5340.
[6] For rare examples for terminal skipped dienes synthesis, see:(a) Basavaiah, D.; Kumaragurubaran, N.; Sharada, D. S. Tetrahedron Lett. 2001, 42, 85.
(b) Basavaiah, D.; Sharada, D. S.; Kumaragurubaran, N.; Reddy, R. M. J. Org. Chem. 2002, 67, 7135.
(c) Li, Y.-Q.; Wang, H.-J.; Huang, Z.-Z. J. Org. Chem. 2016, 81, 4429.
[7] For reviews, see:(a) Edmonds, M.; Abell, A. In Modern Carbonyl Olefination, Ed.:Takeda, T., Wiley-VCH, Weinheim, Germany, 2004, pp. 1~17.
(b) Ju, Y. In Modern Organic Reactions, Vol. 3, Eds.:Hu, Y.-F.; Lin, G.-Q., Chemical Industry Press, Beijing, 2008, pp. 413~460(in Chinese). (巨勇, 现代有机反应, 主编:胡跃飞, 林国强, 第3卷, 化学工业出版社, 北京, 2008, pp. 413~460.)
(c) Gu, Y.; Tian, S.-K. Top. Curr. Chem. 2012, 327, 197.
[8] (a) Liu, W.-B.; He, H.; Dai, L.-X.; You, S.-L. Chem.-Eur. J. 2010, 16, 7376.
(b) Ma, X.-T.; Wang, Y.; Dai, R.-H.; Liu, C.-R.; Tian, S.-K. J. Org. Chem. 2013, 78, 11071.
[9] For reviews, see:(a) Bandini, M. Angew. Chem., Int. Ed. 2011, 50, 994.
(b) Sundararaju, B.; Achard, M.; Bruneau, C. Chem. Soc. Rev. 2012, 41, 4467.
(c) Butta, N. A.; Zhang, W. Chem. Soc. Rev. 2015, 44, 7929.
(d) Ferraccioli, R.; Pignataro, L. Curr. Org. Chem. 2015, 19, 106. For selected recent examples:
(e) Shen, D.; Chen, Q.; Yan, P.; Zeng, X.; Zhong, G. Angew. Chem., Int. Ed. 2017, 129, 3290.
(f) Wu, F.-P.; Peng, J.-B.; Fu, L.-Y.; Qi, X.; Wu, X.-F. Org. Lett 2017, 19, 5474.
(g) Su, Y.-L.; Han, Z.-Y.; Li, Y.-H.; Gong, L.-Z. ACS Catal. 2017, 7, 7917.
(h) Jia, X.-G.; Guo, P.; Duan, J.; Shu, X.-Z. Chem. Sci. 2018, 9, 640.
[10] Ma, X.; Yu, J.; Han, C.; Zhou, Q.; Ren, M.; Li, L.; Tang, L. Adv. Synth. Catal. 2019, https://doi.org/10.1002/adsc.201801266.
[11] (a) Ma, X.-T.; Dai, R.-H.; Zhang, J.; Gu, Y.; Tian, S.-K. Adv. Synth. Catal. 2014, 356, 2984.
(b) Ma, X.; Yu, L.; Su, C.; Yang, Y.; Li, H.; Xu, Q. Adv. Synth. Catal. 2017, 359, 1649.
(c) Ma, X.; Xu, Q.; Li, H.; Su, C.; Yu, L.; Zhang, X.; Cao, H.; Han, L.-B. Green Chem. 2018, 20, 3408.
(d) Ma, X.; Su, C.; Xu, Q. Top. Curr. Chem. 2016, 374, 27.
[12] Kaszynski, P.; Friedli, A. C.; Michl, J. J. Am. Chem. Soc. 1992,114, 601.
[13] Liu, H.-J.; Wynn, H. Tetrahedron Lett. 1982, 23, 3151.
/
| 〈 |
|
〉 |