

Chinese Journal of Organic Chemistry >
Ligand-Switched Pd-Catalyzed Divergent Transformations of Vinyl Cyclic Carbamates and Isocyanates
Received date: 2022-05-22
Revised date: 2022-07-25
Online published: 2022-08-18
Supported by
National Natural Science Foundation of China(21822103); National Natural Science Foundation of China(21820102003); National Natural Science Foundation of China(91956201); Program of Introducing Talents of Discipline to Universities of China (111 Program); Program of Introducing Talents of Discipline to Universities of China(B17019); Natural Science Foundation of Hubei Province(2017AHB047)
Synthesis of urea compounds is highly significant for pharmaceutical chemistry, agricultural chemistry, matterials science and organic synthesis. By ultilizing ligands to switch the chemoselectivity, two transformations of vinyl cyclic carbamates and isocyanates were developed to afford linear and cyclic ureas in a controlled manner. When phosphoramidite was used as the monodentate P-ligand, a wide range of (E)-1,3-diene-substituted linear ureas were produced in high yields and selectivity (21 examples, 51%~95% yields). While, using bidentate biphosphine ligand to replace the phosphoramidite ligand, a variety of highly functionalized vinyl-substitued cyclic ureas, tetrahydro-2-pyrimidinones, were delivered instead starting from the same chemical feedstocks (8 examples, 72%~94% yields). To illusrate the switched chemoselectivity, two possible reaction mechanisms were proposed based on the experiment results and proceeding literatures. The keys of these two reactions, dienylation and annulation, are the β-hydride elimination and the N-allylic alkylation of ally-Pd species, respectively. It is supposed that the basicity of the ligand will affect the reactivity of π-ally-Pd intermediates toward electrophilic ring closing. It is believed that this research not only enriches the application of vinyl carbamate reagents in organic synthesis, but also provides a new protocol to access significant nitrogen-containing molecules.
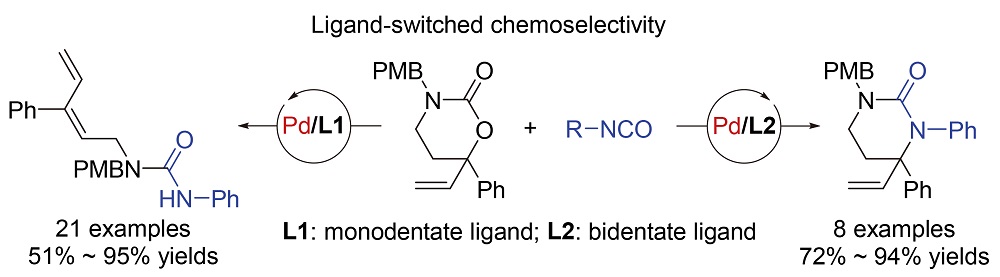
Key words: ligand-switched; palladium catalysis; urea; vinyl carbamate; isocyanate
Wei Xiong , Bin Shi , Xuan Jiang , Liangqiu Lu , Wenjing Xiao . Ligand-Switched Pd-Catalyzed Divergent Transformations of Vinyl Cyclic Carbamates and Isocyanates[J]. Chinese Journal of Organic Chemistry, 2023 , 43(1) : 265 -273 . DOI: 10.6023/cjoc202205038
| [1] | (a) Guo, W.; Gómez, J. E.; Cristòfol, à.; Xie, J.; Kleij, A. W. Angew. Chem., Int. Ed. 2018, 57, 13735. |
| [1] | (b) Zuo, L.; Liu, T.; Chang, X.; Guo, W. Molecules 2019, 24, 3930. |
| [1] | (c) Li, T.-R.; Wang, Y.-N.; Xiao, W.-J.; Lu, L.-Q. Tetrahedron Lett. 2018, 59, 1521. |
| [2] | Knight, J. G.; Ainge, A. M.; Harwood, S. J.; Maughan, H. I.; Armour, D. R.; Hollinshead, D. M.; Jaxa-Chamiec, A. A. J. Am. Chem. Soc. 2000, 122, 2944. |
| [3] | Wang, C.; Tunge, J. A. Org. Lett. 2006, 8, 3211. |
| [4] | Ohmatsu, K.; Imagawa, N.; Ooi, T. Nat. Chem. 2014, 6, 47. |
| [5] | (a) Guo, C.; Fleige, M.; Janssen-Müller, D.; Daniliuc, C. G.; Glorious, F. J. Am. Chem. Soc. 2016, 138, 7840. |
| [5] | (b) Leth, L. A.; Glaus, F.; Meazza, M.; Fu, L.; Th?gersen, M. K.; Bitsch, E. A.; J?rgensen, K. A. Angew. Chem., Int. Ed. 2016, 55, 15272. |
| [5] | (c) Qi, Z.; Kong, L.; Li, X. Org. Lett. 2016, 18, 4392. |
| [5] | (d) Jin, J.-H.; Wang, H.; Yang, Z.-T.; Yang, W.-L.; Tang, W.; Deng, W.-P. Org. Lett. 2018, 20, 104. |
| [5] | (e) Mei, G.-J.; Li, D.; Zhou, G.-X.; Shi, Q.; Cao, Z.; Shi, F. Chem. Commun. 2017, 53, 10030. |
| [5] | (f) Mei, G.-J.; Bian, C.-Y.; Li, G.-H.; Xu, S.-L.; Zheng, W.-Q.; Shi, F. Org. Lett. 2017, 19, 3219. |
| [5] | (g) Sun, M.; Wan, X.; Zhou, S.-J.; Mei, G.-J.; Shi, F. Chem. Commun. 2019, 55, 1283. |
| [5] | (f) Ismail, S. N. F. B. S.; Yang, B.; Zhao, Y. Org. Lett. 2021, 23, 2884. |
| [6] | (a) Brown, R. W.; Zamani, F.; Gardiner, M. G.; Yu, H.-B.; Pyne, S. G.; Hyland, C. J. T. Chem. Sci. 2019, 10, 9051. |
| [6] | (b) Song, B.-C.; Xie, P.-P.; Li, Y.-Z.; Hao, J.-P.; Wang, L.; Chen, X.-Y.; Xu, Z.-L.; Quan, H.-T.; Lou, L.; Xia, Y.-Z.; Houk, K. N.; Yang, W.-B. J. Am. Chem. Soc. 2020, 142, 9982. |
| [7] | (a) Li, T.-R.; Tan, F.; Lu, L.-Q.; Wei, Y.; Wang, Y.-N.; Liu, Y.-Y.; Yang, Q.-Q.; Chen, J.-R.; Shi, D.-Q.; Xiao, W.-J. Nat. Commun. 2014, 5, 5500. |
| [7] | (b) Wei, Y.; Lu, L.-Q.; Li, T.-R.; Feng, B.; Wang, Q.; Xiao, W.-J.; Alper, H. Angew. Chem., Int. Ed. 2016, 55, 2200. |
| [7] | (c) Wang, Q.; Li, T.-R.; Lu, L.-Q.; Li, M.-M.; Zhang, K.; Xiao, W.-J. J. Am. Chem. Soc. 2016, 138, 8360. |
| [7] | (d) Li, M.-M.; Wei, Y.; Liu, J.; Chen, H.-W.; Lu, L.-Q.; Xiao, W.-J. J. Am. Chem. Soc. 2017, 139, 14707. |
| [7] | (e) Feng, B.; Lu, L.-Q.; Chen, J.-R.; Feng, G.-Q.; He, B.-Q.; Lu, B.; Xiao, W.-J. Angew. Chem., Int. Ed. 2018, 57, 5888. |
| [7] | (f) Wei, Y.; Liu, S.; Li, M.-M.; Li, Y.; Lan, Y.; Lu, L.-Q.; Xiao, W.-J. J. Am. Chem. Soc. 2019, 141, 133. |
| [7] | (g) Wang, Y.-N.; Xiong, Q.; Lu, L.-Q.; Zhang, Q.-L.; Wang, Y.; Lan, Y.; Xiao, W.-J. Angew. Chem., Int. Ed. 2019, 58, 11013. |
| [7] | (h) Zhang, M.-M.; Wang, Y.-N.; Wang, B.-C.; Chen, X.-W.; Lu, L.-Q.; Xiao, W.-J. Nat. Commun. 2019, 10, 2716. |
| [7] | (i) Xiong, W.; Xuan, J.; Zhang, M.-M.; Xiao, W.-J.; Lu, L.-Q. Chem. Commun. 2021, 57, 13566. |
| [8] | Zhou, H.-B.; Alper, H. J. Org. Chem. 2003, 68, 3439. |
| [9] | (a) Larksarp, C.; Alper, H. J. Am. Chem. Soc. 1997, 119, 3709. |
| [9] | (b) Larksarp, C.; Alper, H. J. Org. Chem. 1999, 64, 4152. |
| [9] | (c) Trost, B. M.; Fandrick, D. R. J. Am. Chem. Soc. 2003, 125, 11836. |
| [9] | (d) Dong, C.; Alper, H. Tetrahedron: Asymmetry 2004, 15, 1537. |
| [9] | (e) Shintani, R.; Park, S.; Shirozu, F.; Murakami, M.; Hayashi, T. J. Am. Chem. Soc. 2008, 130, 16174. |
| [9] | (f) Shintani, R.; Tsuji, T.; Park, S.; Hayashi, T. J. Am. Chem. Soc. 2010, 132, 7508. |
| [9] | (g) Shintani, R.; Ito, T.; Nagamoto, M.; Otomo, H.; Hayashi, T. Chem. Commun. 2012, 48, 9936. |
| [9] | (h) Khan, A.; Xing, J.-X.; Zhao, J.-M.; Kan, Y.-H.; Zhang, W.-B.; Zhang, Y.-J. Chem.-Eur. J. 2015, 21, 120. |
| [9] | (i) Khan, I.; Shah, B. H.; Zhao, C.; Xu, F.; Zhang, Y.-J. Org. Lett. 2019, 21, 9452. |
| [9] | (j) Hang, Q.-Q.; Liu, S.-J.; Sun, T.-T.; Zhang, Y.-C.; Mei, G.-J.; Shi, F. Chin. J. Chem. 2020, 38, 1612. |
| [10] | Zhang, Q.-L.; Xiong, Q.; Li, M.-M.; Xiong, W.; Shi, B.; Lan, Y.; Lu, L.-Q.; Xiao, W.-J. Angew. Chem., Int. Ed. 2020, 59, 14096. |
/
| 〈 |
|
〉 |