

Chinese Journal of Organic Chemistry >
An “Aggregation-Induced Emission+Excited-State Intramolecular Proton Transfer” Mechanisms-Based Benzothiazole Derived Fluorescent Probe and Its ClO– Recognition
Received date: 2022-04-22
Revised date: 2022-07-17
Online published: 2022-08-25
Supported by
National Natural Science Foundation of China(21878023); National Natural Science Foundation of China(U1608222); Program for Distinguished Professor of Liaoning Province
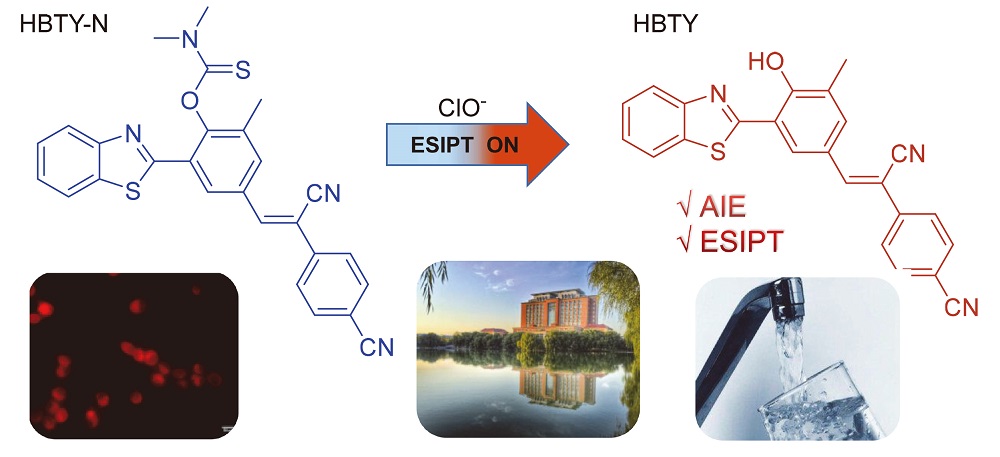
A fluorescent probe (Z)-O-(2-(benzo[d]thiazol-2-yl)-4-(2-cyano-2-(4-cyanophenyl)vinyl)-6-methylphenyl) dimeth- yl cabarmothioate (HBTY-N) based on benzothiazole derivatives was synthesized. The probe can recognize ClO– with high selectivity through fluorescence Off-On change in Tris solution (pH=7). After adding ClO– to the probe, the probe solution changed from no fluorescence to orange-red fluorescence, the Stokes shift is 235 nm, and the recognition response is fast. The detection limit of the probe HBTY-N for ClO– is 2.127×10–7 mol/L, the applicable pH range is 1~10, and it has strong anti- interference ability. Mechanistic studies show that under the action of ClO–, the probe releases the fluorophore (Z)-4-(2- (3-(benzo[d]thiazol-2-yl)-4-hydroxy-5-methylphenyl)-1-cyanovinyl)benzonitrile (HBTY) with “aggregation-induced emission (AIE)+excited-state intramolecular proton yransfer (ESIPT)” properties through the “oxidative deprotection” mechanism, and the quantum yield changes from 0 to 42.88%. In addition, the probe HBTY-N can perform fluorescence imaging of ClO– in living cells with low cytotoxicity, and can also be used for the detection of ClO– in actual water samples, which has potential application value.
Meng Liu , Yanru Huang , Xiaofei Sun , Lijun Tang . An “Aggregation-Induced Emission+Excited-State Intramolecular Proton Transfer” Mechanisms-Based Benzothiazole Derived Fluorescent Probe and Its ClO– Recognition[J]. Chinese Journal of Organic Chemistry, 2023 , 43(1) : 345 -351 . DOI: 10.6023/cjoc202204054
| [1] | Xie, X.; Wang, J.; Yan, Y.; Zhang, X.; Liu, C.; Yang, J.; Hua, J. Analyst 2018, 143, 5736. |
| [2] | Huo, B.; Du, M.; Shen, A.; Li, M.; Lai, Y.; Bai, X.; Gong, A.; Fang, L.; Yang, Y. Sens. Actuators, B 2019, 284, 23. |
| [3] | Shi, W. J.; Feng, L. X.; Wang, X.; Huang, Y.; Wei, Y. F.; Huang, Y. Y.; Ma, H. J.; Wang, W.; Xiang, M.; Gao, L. Talanta 2021, 233, 122581. |
| [4] | Xiao, H.; Li, J.; Zhao, J.; Yin, G.; Quan, Y.; Wang, J.; Wang, R. J. Mater. Chem. B 2015, 3, 1633. |
| [5] | Li, J.; Li, P.; Huo, F.; Yin, C.; Liu, T.; Chao, J.; Zhang, Y. Dyes Pigm. 2016, 130, 209. |
| [6] | Wu, L.; Yang, Q.; Liu, L.; Sedgwick, A. C.; Cresswell, A. J.; Bull, S. D.; Huang, C.; James, T. D. Chem. Commun. 2018, 54, 8522. |
| [7] | Huo, B. L.; Du, M.; Shen, A.; Li, M. W.; Lai, Y.; Bai, X.; Gong, A. J.; Yang, Y. X. Anal. Chem. 2019, 91, 10979. |
| [8] | Yang, M. P.; Su, N.; Li, Y. X.; Wang, L.; Ma, L. F.; Zhang, Y.; Li, J.; Yang, B. Q.; Kang, L. L. Chin. J. Org. Chem. 2017, 38, 636. (in Chinese) |
| [8] | (杨美盼, 苏娜, 李玉香, 王莉, 马利锋, 张媛, 李靖, 杨秉勤, 康龙丽, 有机化学, 2017, 38, 636.) |
| [9] | Ma, T.; Zhang, Y.; Fu, K.; Li, Z.; Yuan, C.; Ma, W. Bioorg. Chem. 2022, 123, 105798. |
| [10] | Singh, G.; Devi, A.; Gupta, S.; Yadav, R.; Sehgal, R. J. Mol. Struct. 2022, 1262, 132967. |
| [11] | VanDenburgh, K. L.; Liu, Y.; Sadhukhan, T.; Benson, C. R.; Cox, N. M.; Erbas-Cakmak, S.; Qiao, B.; Gao, X.; Pink, M.; Raghava- chari, K.; Flood, A. H. Org. Biomol. Chem. 2020, 18, 431. |
| [12] | Sun, X. Y.; Liu, T.; Sun, J.; Wang, X. J. RSC Adv. 2020, 10, 10826. |
| [13] | Deng, F.; Sun, D.; Yang, S.; Huang, W.; Huang, C.; Xu, Z.; Liu, L. Spectrochim. Acta, Part A 2022, 268, 120662. |
| [14] | Tian, Y.; Li, M.; Liu, Y. Molecules 2020, 25, 4465. |
| [15] | Liu, J.; Xiong, Y.; Huang, Y.; Zhu, X.; Liu, Y.; Zhang, L.; Yan, J. New J. Chem. 2021, 45, 18453. |
| [16] | Mei, H.; Gu, X.; Wang, M.; Cai, Y.; Xu, K. J. Photochem. Photobiol., A 2021, 418, 113335. |
| [17] | Wu, Q.; Li, Y.; Li, Y.; Wang, D.; Tang, B. Z. Mater. Chem. Front. 2021, 5, 3489. |
| [18] | Liu, S.; Feng, G.; Tang, B. Z.; Liu, B. Chem. Sci. 2021, 12, 6488. |
| [19] | Shi, J.; Deng, Q.; Wan, C.; Zheng, M.; Huang, F.; Tang, B. Chem. Sci. 2017, 8, 6188. |
| [20] | Anthony, K.; Brown, R. G.; Hepworth, J. D.; Hodgson, K. W.; May, B. J. Chem. Soc., Perkin Trans. 1 1984, 2111. |
| [21] | Cai, Y.; Fang, J.; Wang, B.; Zhang, F.; Shao, G.; Liu, Y. Sens. Actuators, B 2019, 292, 156. |
| [22] | Chen, L.; Jiang, H.; Li, N.; Meng, Q.; Li, Z.; Han, Q.; Liu, X. Spectrochim. Acta, Part A 2022, 268, 120704. |
| [23] | Pan, Y. X.; Ban, L. F.; Li, J. J.; Liu, M.; Tang, L. J.; Yan, X. M. Dyes Pigm. 2022, 203, 110305. |
| [24] | Xu, D.; Tang, L.; Tian, M.; He, P.; Yan, X. Tetrahedron Lett. 2017, 58, 3654. |
| [25] | Tian, Y.; Zhou, D. Y.; Jiang, W. L.; She, Z. P.; Li, Y.; Li, C. Y. Talanta 2021, 223, 121720. |
| [26] | Wang, W. X.; Jiang, W. L.; Liu, Y.; Li, Y.; Zhang, J.; Li, C. Y. Sens. Actuators, B 2020, 320, 128296. |
| [27] | Zhou, K.; Ren, M.; Wang, L.; Li, Z.; Lin, W. Anal. Methods 2018, 10, 2546. |
| [28] | Tang, H.; Qiang, X.; Gao, Y.; Teng, H.; Chen, X.; Zhang, Y.; Tian, J.; Qin, B.; Guo, Y. Dyes Pigm. 2021, 188, 109219. |
| [29] | Jiao, X.; Li, Y.; Niu, J.; Xie, X.; Wang, X.; Tang, B. Anal. Chem. 2018, 90, 533. |
| [30] | Liu, M.; Bai, Y.; He, Y.: Zhou, J.; Ge, Y. L.; Zhou, J. G.; Song, G. W. Microchim. Acta 2021, 188, 15. |
| [31] | Han, Z.; Dong, L.; Sun, F.; Long, L.; Jiang, S.; Dai, X.; Zhang, M. Anal. Biochem. 2020, 602, 113795. |
| [32] | Tang, X.; Zhu, Z.; Wang, Z.; Tang, Y.; Wang, L.; Liu, L. Spectrochim. Acta, Part A 2020, 228, 117845. |
| [33] | Wang, W.; Ning, J. Y.; Liu, J. T.; Miao, J. Y.; Zhao, B. X. Dyes Pigm. 2019, 171, 107708. |
| [34] | Yang, Y.; Cheng, S.; Dong, W. Luminescence 2021, 36, 1377. |
/
| 〈 |
|
〉 |