

Chinese Journal of Organic Chemistry >
Research Progress of Electrochemical Synthesis of C-Sulfonyl Compounds
Received date: 2022-05-13
Revised date: 2022-07-14
Online published: 2022-08-25
Supported by
National Natural Science Foundation of China(21961042); Natural Science Foundation of Guangxi Province(2021GXNSFBA075056); Basic Ability Improvement Project of Young and Middle-aged Teachers in Guangxi Colleges(2021KY0587); Basic Ability Improvement Project of Young and Middle-aged Teachers in Guangxi Colleges(2022KY0572); Basic Ability Improvement Project of Young and Middle-aged Teachers in Guangxi Colleges(2021KY0499); Research Project of Yulin Normal University(G2021ZK16); Open Project of Guangxi Key Laboratory of Agricultural Resources Chemistry and Biotechnology(2021KF01)
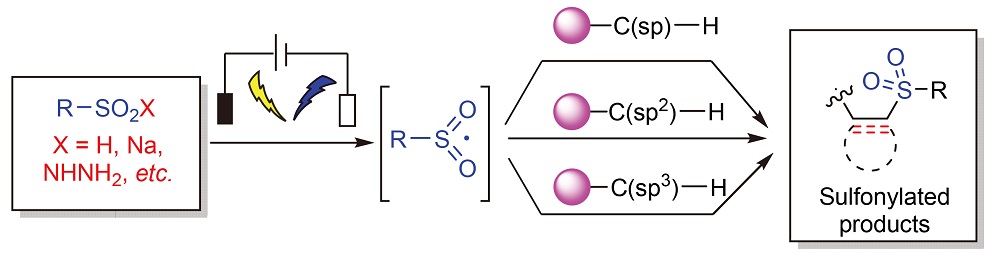
Sulfonyl compounds are important organic sulfur compounds, which are widely used in the fields of medicine, pesticides, functional materials and so on. Therefore, efficient strategies for the synthesis of sulfonyl compounds have become the focus of extensive research. Organic electrochemical synthesis is a green, mild and efficient synthesis strategy, which shows great potential in the synthesis of sulfonyl compounds. The reactions of electrochemical synthesis of C-sulfonyl compounds in recent years are introduced. The reactions of electrochemical synthesis of C(sp)-sulfonyl compounds, C(sp2)-sulfonyl compounds and C(sp3)-sulfonyl compounds are classified, summarized and discussed, and the corresponding reaction mechanism is described, so as to provide reference for the application of such reactions in organic synthesis in the future.
Key words: organic electrochemistry; sulfonyl compounds; sulfonylation
Wanjie Wei , Lei Zhan , Lei Gao , Guobao Huang , Xianli Ma . Research Progress of Electrochemical Synthesis of C-Sulfonyl Compounds[J]. Chinese Journal of Organic Chemistry, 2023 , 43(1) : 17 -35 . DOI: 10.6023/cjoc202205018
| [1] | (a) Curti, C.; Laget, M.; Carle, A. O.; Gellis, A.; Vanelle, P. Eur. J. Med. Chem. 2007, 42, 880. |
| [1] | (b) Meadows, D. C.; Sanchez, T.; Neamati, N.; North, T. W.; Hague, J. G. Bioorg. Med. Chem. 2007, 15, 1127. |
| [1] | (c) Frankel, B. A.; Bentley, M.; Kruger, R. G.; McCafferty, D. G. J. Am. Chem. Soc. 2004, 126, 3404. |
| [2] | Devendar, P.; Yang, G. F. Top. Curr. Chem. 2017, 375, 82. |
| [3] | (a) Sasabe, H.; Seino, Y.; Kimura, M.; Kido, J. Chem. Mater. 2012, 24, 1404. |
| [3] | (b) Turkoglu, G.; Cinar, M. E.; Ozturk, T. Top. Curr. Chem. 2017, 375, 84. |
| [4] | (a) Zhao, C.; Rakesh, K. P.; Ravidar, L.; Fang, W. Y.; Qin, H. L. Eur. J. Med. Chem. 2019, 162, 679. |
| [4] | (b) Man, H. W.; Schafer, P.; Wong, L. M.; Patterson, R. T.; Corral, L. G.; Raymon, H.; Blease, K.; Leisten, J.; Shirley, M. A.; Tang, Y.; Babusis, D. M.; Chen, R.; Stirling, D.; Muller, G. W. J. Med. Chem. 2009, 52, 1522. |
| [5] | Julia, M.; Paris, J. M. Tetrahedron Lett. 1973, 49, 4833. |
| [6] | (a) Wang, Z.-Q.; Hou, C.; Zhong, Y.-F.; Lu, Y.-X.; Mo, Z.-Y.; Pan, Y.-M.; Tang, H.-T. Org. Lett. 2019, 21, 9841. |
| [6] | (b) He, M.-X.; Mo, Z.-Y.; Wang, Z.-Q.; Cheng, S.-Y.; Xie, R.-R.; Tang, H.-T.; Pan, Y.-M. Org. Lett. 2020, 22, 724. |
| [6] | (c) Meng, X.-J.; Zhong, P.-F.; Wang, Y.-M.; Wang, H.-S.; Tang, H.-T.; Pan, Y.-M. Adv. Synth. Catal. 2020, 362, 506. |
| [6] | (d) Li, Q.-Y.; Cheng, S.-Y.; Tang, H.-T.; Pan, Y.-M. Green Chem. 2019, 21, 5517. |
| [6] | (e) Zhong, P.-F.; Lin, H.-M.; Wang, L.-W.; Mo, Z.-Y.; Meng, X.-J.; Tang, H.-T.; Pan, Y.-M. Green Chem. 2020, 22, 6334. |
| [6] | (f) Wu, Y.; Chen, J.-Y.; Liao, H.-R.; Shu, X.-R.; Duan, L.-L.; Yang, X.-F.; He, W.-M. Green Synth. Catal. 2021, 2, 233. |
| [6] | (g) Li, Q.-Y.; Swaroop, T. R; Hou, C.; Wang, Z.-Q.; Pan, Y.-M.; Tang, H.-T. Adv. Synth. Catal. 2019, 361, 1761. |
| [6] | (h) Chen, N.; Xu, H.-C. Green Synth. Catal. 2021, 2, 165. |
| [6] | (i) He, M.-X.; Zhong, P.-F.; Liu, H.-F.; Ou, C.-H.; Pan, Y.-M.; Tang, H.-T. Green Synth. Catal. 2022, DOI: 10.1016/j.gresc.2022.03.002. |
| [6] | (j) Wang, X.-Y.; Zhong, Y.-F.; Mo, Z.-Y.; Wu, S.-H.; Xu, Y.-L.; Tang, H.-T.; Pan, Y.-M. Adv. Synth. Catal. 2021, 363, 208. |
| [6] | (k) Yang, Z.; Yu, Y.; Lai, L.; Zhou, L.; Ye, K.; Chen, F.-E. Green Synth. Catal. 2021, 2, 19. |
| [6] | (k) Mei, H.; Pajkert, R.; Wang, L.; Li, Z.; R?schenthaler, G.-V.; Han, J. Green Chem. 2020, 22, 3028. |
| [7] | (a) Todoroki, H.; Iwatsu, M.; Urabe, D.; Inoue, M. J. Org. Chem. 2014, 79, 8835. |
| [7] | (b) Kawai, H.; Yuan, Z.; Tokunaga, E.; Shibata, N. Org. Lett. 2012, 14, 5330. |
| [7] | (c) Riddell, N.; Tam, W. J. Org. Chem. 2006, 71, 1934. |
| [7] | (d) Huang, X.; Duan, D.; Zheng, W. J. Org. Chem. 2003, 68, 1958. |
| [7] | (e) Xie, M.; Wang, J.; Gu, X.; Sun, Y.; Wang, S. Org. Lett. 2006, 8, 431. |
| [7] | (f) Zhang, S.; Ye, X.; Wojtas, L.; Hao, W.; Shi, X. Green Synth. Catal. 2021, 2, 82. |
| [8] | (a) Guo, A.; Han, Ji. B.; Zhu, L.; Wei, Y.; Tang, X. Y. Org. Lett. 2019, 21, 2927. |
| [8] | (b) Jin, W.; Wu, M.; Xiong, Z.; Zhu, G. Chem. Commun. 2018, 54, 7924. |
| [8] | (c) Schwarz, J.; K?nig, B. ChemPhotoChem 2017, 1, 237. |
| [8] | (d) Wang, X.; Wu, S.; Zhong, Y.; Wang, Y.; Pan, Y.; Tang, H. Chin. Chem. Lett. 2022, DOI: 10.1016/j.cclet.2022.05.051. |
| [9] | Chen, H.; Zhang, L. Angew. Chem., Int. Ed. 2015, 54, 11775. |
| [10] | Mo, Z.-Y.; Zhang, Y.-Z.; Huang, G.-B.; Wang, X.-Y.; Pan, Y.-M.; Tang, H.-T. Adv. Synth. Catal. 2020, 362, 2160. |
| [11] | Meng, X.; Xu, H.; Cao, X.; Cai, X. M.; Luo, J.; Wang, F.; Huang, S. Org. Lett. 2020, 22, 6827. |
| [12] | Zhong, Q.; Zhao, Y.; Sheng, S.; Chen, J. Synth. Commun. 2020, 50, 161. |
| [13] | Terent’ev, A. O.; Mulina, O. M.; Pirgach, D. A.; Ilovaisky, A. I.; Syroeshkin, M. A.; Kapustina, N. I.; Nikishin, G. I. Tetrahedron 2017, 73, 6871. |
| [14] | Kim, H. S.; Lee, S. Eur. J. Org. Chem. 2019, 41, 6951. |
| [15] | Kong, X.; Yu, K.; Chen, Q.; Xu, B. Asian J. Org. Chem. 2020, 9, 1760. |
| [16] | Zhang, X.; Lu, D.; Wang, Z. Eur. J. Org. Chem. 2021, 30, 4284. |
| [17] | Yuan, Y.; Yu, Y.; Qiao, J.; Liu, P.; Yu, B.; Zhang, W.; Liu, H.; He, M.; Huang, Z.; Lei, A. Chem. Commun. 2018, 54, 11471. |
| [18] | Zhang, Y.-Z.; Mo, Z.-Y.; Wang, H.-S.; Wen, X.-A.; Tang, H.-T.; Pan, Y.-M. Green Chem. 2019, 21, 3807. |
| [19] | Mahanty, k.; Maiti, D.; Sarkar, S. D. J. Org. Chem. 2020, 85, 369. |
| [20] | Wen, J.; Shi, W.; Zhang, F.; Liu, D.; Tang, S.; Wang, H.; Lin, X.-M.; Lei, A. Org. Lett. 2017, 19, 3131. |
| [21] | Lu, F.; Li, J.; Wang, T.; Li, Z.; Jiang, M.; Hu, X.; Pei, H.; Yuan, F.; Lu, L.; Lei, A. Asian J. Org. Chem. 2019, 8, 1838. |
| [22] | Jiang, M.; Yuan, Y.; Wang, T.; Xiong, Y.; Li, J.; Guo, H.; Lei, A. Chem. Commun. 2019, 55, 13852. |
| [23] | Xiao, H.-L.; Yang, C.-W.; Zhang, N.-T.; Zeng, C.-C.; Hu, L.-M.; Tian, H.-Y.; Daniel Little, R. Tetrahedron 2013, 69, 658. |
| [24] | Luo, Y.-C.; Pan, X.-J.; Yuan, G.-Q. Tetrahedron 2015, 71, 2119. |
| [25] | Qian, Peng.; Bi, M.; Su, J.; Zha, Z.; Wang, Z. J. Org. Chem. 2016, 81, 4876. |
| [26] | Nikl, J.; Lips, S.; Schollmeyer, D.; Franke, R.; Waldvogel, S. R. Chem.-Eur. J. 2019, 25, 6891. |
| [27] | Sun, X.; Zhang, F.; Yan, K.; Feng, W.; Sun, X.; Yang, J.; Wen, J. Adv. Synth. Catal. 2021, 363, 3485. |
| [28] | Salmas, R. E.; Seeman, P.; Aksoydan, B.; Erol, I.; Kantarcioglu, I.; Stein, M.; Yurtsever, M.; Durdagi, S. ACS Chem. Neurosci. 2017, 8, 1404. |
| [29] | Nugent, T. C.; El-Shazly, M. Adv. Synth. Catal. 2010, 352, 753. |
| [30] | Gu, Q.; Wang, X.; Liu, X.; Wu, G.; Xie, Y.; Shao, Y.; Zhao, Y.; Zeng, X. Org. Biomol. Chem. 2021, 19, 8295. |
| [31] | Yuan, Y.; Cao, Y.; Lin, Y.; Li, Y.; Huang, Z.; Lei, A. ACS Catal. 2018, 8, 10871. |
| [32] | He, T. J.; Zhong, W. Q.; Huang, J. M. Chem. Commun. 2020, 56, 2735. |
| [33] | Wei, W.-J.; Zhong, Y.-J.; Feng, Y.-F.; Gao, L.; Tang, H.-T.; Pan, Y.-M.; Ma, X.-L.; Mo, Z.-Y. Adv. Synth. Catal. 2021, 364, 726. |
| [34] | Pan, X.; Gao, J.; Yuan, G. Tetrahedron 2015, 71, 5525. |
| [35] | Zheng, M.; Yuan, X.; Cui, Y.; Qiu, J.; Li, G.; Guo, K. Org. Lett. 2018, 20, 7784. |
| [36] | Yavari, I.; Shaabanzadeh, S. Org. Lett. 2020, 22, 464. |
| [37] | Yu, M.; Wang, H.; Gao, Y.; Bu, F.; Cong, H.; Lei, A. Cell Rep. Phys. Sci. 2021, 2, 100476. |
| [38] | Luo, X.; Wang, S.; Lei, A. Adv. Synth. Catal. 2022, 364, 1016. |
/
| 〈 |
|
〉 |